 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name Isocyanatobenzene | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.002.852 |
| EC Number |
|
PubChem CID | |
| UNII | |
| UN number | 2487 |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C7H5NO | |
| Molar mass | 119.123 g·mol−1 |
| Appearance | Colourless liquid |
| Density | 1.09 |
| Melting point | −30 °C (−22 °F; 243 K) |
| Boiling point | 165 °C (329 °F; 438 K) |
| Reacts with water | |
| −72.7·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: [1] | |
      | |
| Danger | |
| H226, H302, H314, H317, H330, H334, H335, H410, H411 | |
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P264+P265, P270, P271, P272, P273, P280, P284, P301+P317, P301+P330+P331, P302+P352, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P317, P319, P320, P321, P330, P333+P317, P342+P316, P362+P364, P363, P370+P378, P391, P403, P403+P233, P403+P235, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
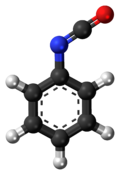
Phenyl isocyanate is an organic compound typically abbreviated PhNCO. The molecule consists of a phenyl ring attached to the isocyanate functional group. It is a colourless liquid that reacts with water. Phenyl isocyanate has a strong odor and tearing vapours, therefore it has to be handled with care.
Contents
Characteristic of other isocyanates, it reacts with amines to give ureas. [2] Similarly, reacts with alcohols to form carbamates.
It is used in addition with triethylamine to activate nitro groups to undergo (C,O) 1,3-dipolar cycloaddition (as opposed to O,O). The nitro group (RCH2NO2) is converted to oxime-like dimers in the reaction: [3]
- 4 PhNCO + 2 RCH2NO2 → 2(PhNH)2CO + 2 CO2 + (RCNO)2