
Flavan-3-ols are derivatives of flavans that possess a 2-phenyl-3,4-dihydro-2H-chromen-3-ol skeleton. These compounds include catechin, epicatechin gallate, epigallocatechin, epigallocatechin gallate, proanthocyanidins, theaflavins, thearubigins.

Polyphenols are a large family of naturally occurring organic compounds characterized by multiples of phenol units. They are abundant in plants and structurally diverse. Polyphenols include flavonoids, tannic acid, and ellagitannin, some of which have been used historically as dyes and for tanning garments.

Catechin is a flavan-3-ol, a type of natural phenol and antioxidant. It is a plant secondary metabolite. It belongs to the group of flavan-3-ols, part of the chemical family of flavonoids.
Proanthocyanidins are a class of polyphenols found in many plants, such as cranberry, blueberry, and grape seeds. Chemically, they are oligomeric flavonoids. Many are oligomers of catechin and epicatechin and their gallic acid esters. More complex polyphenols, having the same polymeric building block, form the group of tannins.
Thearubigins are polymeric polyphenols that are formed during the enzymatic oxidation and condensation of two gallocatechins with the participation of polyphenol oxidases during the fermentation reactions in black tea. Thearubigins are red in colour and are responsible for much of the staining effect of tea. Therefore, a black tea often appears red while a green or white tea has a much clearer appearance. The colour of a black tea, however, is affected by many other factors as well, such as the amount of theaflavins, another oxidized form of polyphenols.

Procyanidins are members of the proanthocyanidin class of flavonoids. They are oligomeric compounds, formed from catechin and epicatechin molecules. They yield cyanidin when depolymerized under oxidative conditions.

The phenolic content in wine refers to the phenolic compounds—natural phenol and polyphenols—in wine, which include a large group of several hundred chemical compounds that affect the taste, color and mouthfeel of wine. These compounds include phenolic acids, stilbenoids, flavonols, dihydroflavonols, anthocyanins, flavanol monomers (catechins) and flavanol polymers (proanthocyanidins). This large group of natural phenols can be broadly separated into two categories, flavonoids and non-flavonoids. Flavonoids include the anthocyanins and tannins which contribute to the color and mouthfeel of the wine. The non-flavonoids include the stilbenoids such as resveratrol and phenolic acids such as benzoic, caffeic and cinnamic acids.
Prodelphinidin is a name for the polymeric tannins composed of gallocatechin. It yields delphinidin during depolymerisation under oxidative conditions.

Procyanidin C2 is a B type proanthocyanidin trimer, a type of condensed tannin.

Procyanidin B2 is a B type proanthocyanidin. Its structure is (−)-Epicatechin-(4β→8)-(−)-epicatechin.
A type proanthocyanidins are a specific type of proanthocyanidins, which are a class of flavonoid. Proanthocyanidins fall under a wide range of names in the nutritional and scientific vernacular, including oligomeric proanthocyanidins, flavonoids, polyphenols, condensed tannins, and OPCs. Proanthocyanidins were first popularized by French scientist Jacques Masquelier.

Procyanidin B1 is a procyanidin dimer.
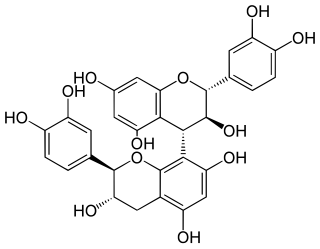
Procyanidin B3 is a B type proanthocyanidin. Procyanidin B3 is a catechin dimer.
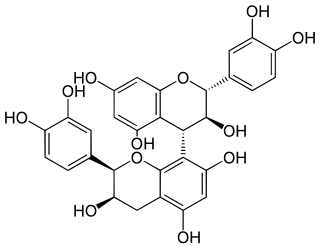
Procyanidin B4 is a B type proanthocyanidin.
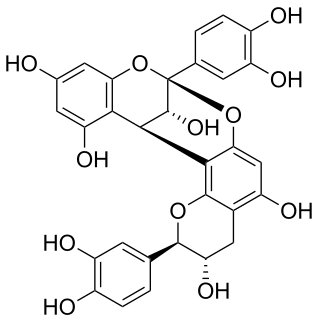
Procyanidin A1 is an A type proanthocyanidin dimer.

p-Dimethylaminocinnamaldehyde (DMACA) is an aromatic hydrocarbon. It is used in an acidic solution to detect indoles.

Procyanidin C1 (PCC1) is a B type proanthocyanidin. It is an epicatechin trimer found in grape, unripe apples, and cinnamon.

Condensed tannins are polymers formed by the condensation of flavans. They do not contain sugar residues.
B type proanthocyanidins are a specific type of proanthocyanidin, which are a class of flavanoids. They are oligomers of flavan-3-ols.

Catechin-7-O-glucoside is a flavan-3-ol glycoside formed from catechin.















