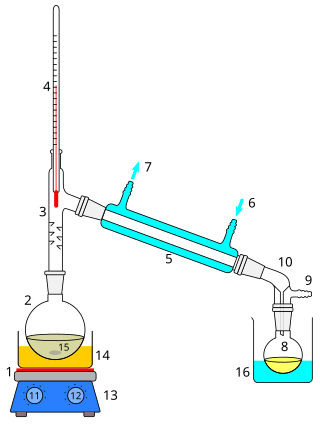
Distillation, or classical distillation, is the process of separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distillation is the heating of solid materials to produce gaseous products ; this may involve chemical changes such as destructive distillation or cracking. Distillation may result in essentially complete separation, or it may be a partial separation that increases the concentration of selected components; in either case, the process exploits differences in the relative volatility of the mixture's components. In industrial applications, distillation is a unit operation of practically universal importance, but is a physical separation process, not a chemical reaction. An installation used for distillation, especially of distilled beverages, is a distillery. Distillation includes the following applications:

Humidity is the concentration of water vapor present in the air. Water vapor, the gaseous state of water, is generally invisible to the human eye. Humidity indicates the likelihood for precipitation, dew, or fog to be present.

A cryostat is a device used to maintain low cryogenic temperatures of samples or devices mounted within the cryostat. Low temperatures may be maintained within a cryostat by using various refrigeration methods, most commonly using cryogenic fluid bath such as liquid helium. Hence it is usually assembled into a vessel, similar in construction to a vacuum flask or Dewar. Cryostats have numerous applications within science, engineering, and medicine.

In vacuum applications, a cold trap is a device that condenses all vapors except the permanent gases into a liquid or solid. The most common objective is to prevent vapors being evacuated from an experiment from entering a vacuum pump where they would condense and contaminate it. Particularly large cold traps are necessary when removing large amounts of liquid as in freeze drying.

A gas duster, also known as canned air or compressed air, is a product used for cleaning or dusting electronic equipment and other sensitive devices that cannot be cleaned using water.
A flash fire is a sudden, intense fire caused by ignition of a mixture of air and a dispersed flammable substance such as a solid, flammable or combustible liquid, or a flammable gas. It is characterized by high temperature, short duration, and a rapidly moving flame front.

In chemistry, volatility is a material quality which describes how readily a substance vaporizes. At a given temperature and pressure, a substance with high volatility is more likely to exist as a vapour, while a substance with low volatility is more likely to be a liquid or solid. Volatility can also describe the tendency of a vapor to condense into a liquid or solid; less volatile substances will more readily condense from a vapor than highly volatile ones. Differences in volatility can be observed by comparing how fast substances within a group evaporate when exposed to the atmosphere. A highly volatile substance such as rubbing alcohol will quickly evaporate, while a substance with low volatility such as vegetable oil will remain condensed. In general, solids are much less volatile than liquids, but there are some exceptions. Solids that sublimate such as dry ice or iodine can vaporize at a similar rate as some liquids under standard conditions.

A liqueur coffee is a caffeinated alcoholic drink that consists of a shot of liqueur, mixed with coffee. It is typically served in a liqueur glass, often accompanied with cream and sugar. Coffee liqueur beverages are served in different fashions and can be found throughout many countries. One of the most popular liqueur coffee beverage is commonly known as Irish coffee. Liqueur coffee beverages are largely classified as cocktails as well as digestifs which are aimed at aiding the digestive process typically after a meal.

Icyball is a name given to two early refrigerators, one made by Australian Sir Edward Hallstrom in 1923, and the other design patented by David Forbes Keith of Toronto, and manufactured by American Powel Crosley Jr., who bought the rights to the device. Both devices are unusual in design in that they did not require the use of electricity for cooling. They can run for a day on a cup of kerosene, allowing rural users lacking electricity the benefits of refrigeration.

A liquid-ring pump is a rotating positive-displacement gas pump, with liquid under centrifugal force acting as a seal.

An absorption refrigerator is a refrigerator that uses a heat source to provide the energy needed to drive the cooling process. The system uses two coolants, the first of which performs evaporative cooling and is then absorbed into the second coolant; heat is needed to reset the two coolants to their initial states. The principle can also be used to air-condition buildings using the waste heat from a gas turbine or water heater. Using waste heat from a gas turbine makes the turbine very efficient because it first produces electricity, then hot water, and finally, air-conditioning—trigeneration. Absorption refrigerators are commonly used in recreational vehicles (RVs), campers, and caravans because the heat required to power them can be provided by a propane fuel burner, by a low-voltage DC electric heater or by a mains-powered electric heater. Unlike more common vapor-compression refrigeration systems, an absorption refrigerator has no moving parts.

Vapour-compression refrigeration or vapor-compression refrigeration system (VCRS), in which the refrigerant undergoes phase changes, is one of the many refrigeration cycles and is the most widely used method for air conditioning of buildings and automobiles. It is also used in domestic and commercial refrigerators, large-scale warehouses for chilled or frozen storage of foods and meats, refrigerated trucks and railroad cars, and a host of other commercial and industrial services. Oil refineries, petrochemical and chemical processing plants, and natural gas processing plants are among the many types of industrial plants that often utilize large vapor-compression refrigeration systems. Cascade refrigeration systems may also be implemented using two compressors.

A thermal expansion valve or thermostatic expansion valve is a component in vapor-compression refrigeration and air conditioning systems that controls the amount of refrigerant released into the evaporator and is intended to regulate the superheat of the refrigerant that flows out of the evaporator to a steady value. Although often described as a "thermostatic" valve, an expansion valve is not able to regulate the evaporator's temperature to a precise value. The evaporator's temperature will vary only with the evaporating pressure, which will have to be regulated through other means.

A flaming drink is a cocktail or other mixed drink that contains flammable, high-proof alcohol, which is ignited before consumption. The alcohol may be an integral part of the drink, or it may be floated as a thin layer across the top of the drink. The flames are mostly for dramatic flair. However, in combination with certain ingredients, the flavor of the drink is altered. Some flavors are enhanced, and the process may impart a toasted flavor to some drinks.

In chemistry, a condenser is laboratory apparatus used to condense vapors – that is, turn them into liquids – by cooling them down.

In systems involving heat transfer, a condenser is a heat exchanger used to condense a gaseous substance into a liquid state through cooling. In so doing, the latent heat is released by the substance and transferred to the surrounding environment. Condensers are used for efficient heat rejection in many industrial systems. Condensers can be made according to numerous designs, and come in many sizes ranging from rather small (hand-held) to very large. For example, a refrigerator uses a condenser to get rid of heat extracted from the interior of the unit to the outside air.

Reflux is a technique involving the condensation of vapors and the return of this condensate to the system from which it originated. It is used in industrial and laboratory distillations. It is also used in chemistry to supply energy to reactions over a long period of time.

A cryogenic gas plant is an industrial facility that creates molecular oxygen, molecular nitrogen, argon, krypton Helium and Xenon at relatively high purity. As air is made up of nitrogen, the most common gas in the atmosphere, at 78%, with Oxygen at 19%, and Argon at 1%, with trace gasses making up the rest, Cryogenic gas plants separate air inside a distillation column at cryogenic temperatures to produce high purity gasses such as Argon, Nitrogen, Oxygen, and many more with 1 ppm or less impurities. The process is based on the general theory of the Hampson-Linde cycle of air separation, which was invented by Dr. Carl von Linde in 1895.

A hand boiler or love meter is a glass sculpture used as an experimental tool to demonstrate vapour-liquid equilibrium, or as a collector's item to whimsically "measure love." It consists of a lower bulb containing a volatile liquid and a mixture of gases that is connected usually by a twisting glass tube that connects to an upper or "receiving" glass bulb.

A Codd-neck bottle is a type of bottle used for carbonated drinks. It has a closing design based on a glass marble which is held against a rubber seal, which sits within a recess in the lip.


















