Isomerases are a general class of enzymes that convert a molecule from one isomer to another. Isomerases facilitate intramolecular rearrangements in which bonds are broken and formed. The general form of such a reaction is as follows:

Apigenin (4′,5,7-trihydroxyflavone), found in many plants, is a natural product belonging to the flavone class that is the aglycone of several naturally occurring glycosides. It is a yellow crystalline solid that has been used to dye wool.
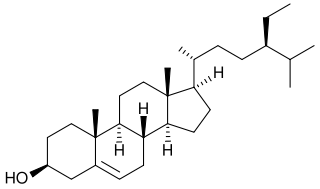
β-sitosterol (beta-sitosterol) is one of several phytosterols with chemical structures similar to that of cholesterol. It is a white, waxy powder with a characteristic odor, and is one of the components of the food additive E499. Phytosterols are hydrophobic and soluble in alcohols.
Squalene synthase (SQS) or farnesyl-diphosphate:farnesyl-diphosphate farnesyl transferase is an enzyme localized to the membrane of the endoplasmic reticulum. SQS participates in the isoprenoid biosynthetic pathway, catalyzing a two-step reaction in which two identical molecules of farnesyl pyrophosphate (FPP) are converted into squalene, with the consumption of NADPH. Catalysis by SQS is the first committed step in sterol synthesis, since the squalene produced is converted exclusively into various sterols, such as cholesterol, via a complex, multi-step pathway. SQS belongs to squalene/phytoene synthase family of proteins.

Lanosterol 14α-demethylase (CYP51A1) is the animal version of a cytochrome P450 enzyme that is involved in the conversion of lanosterol to 4,4-dimethylcholesta-8(9),14,24-trien-3β-ol. The cytochrome P450 isoenzymes are a conserved group of proteins that serve as key players in the metabolism of organic substances and the biosynthesis of important steroids, lipids, and vitamins in eukaryotes. As a member of this family, lanosterol 14α-demethylase is responsible for an essential step in the biosynthesis of sterols. In particular, this protein catalyzes the removal of the C-14α-methyl group from lanosterol. This demethylation step is regarded as the initial checkpoint in the transformation of lanosterol to other sterols that are widely used within the cell.

Isopentenyl pyrophosphate isomerase, also known as Isopentenyl-diphosphate delta isomerase, is an isomerase that catalyzes the conversion of the relatively un-reactive isopentenyl pyrophosphate (IPP) to the more-reactive electrophile dimethylallyl pyrophosphate (DMAPP). This isomerization is a key step in the biosynthesis of isoprenoids through the mevalonate pathway and the MEP pathway.
In enzymology, a 24-methylenesterol C-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a sterol 24-C-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a Δ7-sterol 5(6)-desaturase is an enzyme that catalyzes the chemical reaction
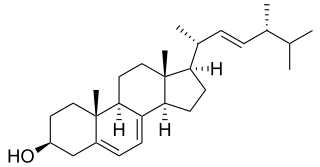
In enzymology, a sterol 14-demethylase (EC 1.14.13.70) is an enzyme of the Cytochrome P450 (CYP) superfamily. It is any member of the CYP51 family. It catalyzes a chemical reaction such as:

In enzymology, a chalcone isomerase is an enzyme that catalyzes the chemical reaction
In enzymology, a copalyl diphosphate synthase is an enzyme that catalyzes the chemical reaction
In enzymology, an inositol-3-phosphate synthase is an enzyme that catalyzes the chemical reaction

Diphosphomevalonate decarboxylase (EC 4.1.1.33), most commonly referred to in scientific literature as mevalonate diphosphate decarboxylase, is an enzyme that catalyzes the chemical reaction
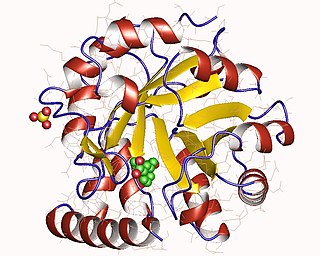
The enzyme indole-3-glycerol-phosphate synthase (IGPS) (EC 4.1.1.48) catalyzes the chemical reaction
The enzyme hydroperoxide dehydratase (EC 4.2.1.92) catalyzes the chemical reaction
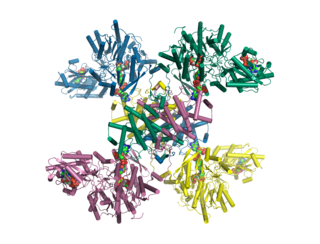
ATP citrate synthase (also ATP citrate lyase (ACLY)) is an enzyme that in animals represents an important step in fatty acid biosynthesis. By converting citrate to acetyl-CoA, the enzyme links carbohydrate metabolism, which yields citrate as an intermediate, with fatty acid biosynthesis, which consumes acetyl-CoA. In plants, ATP citrate lyase generates cytosolic acetyl-CoA precursors of thousands of specialized metabolites, including waxes, sterols, and polyketides.
Fenpropimorph is a morpholine-derived fungicide used in agriculture, primarily on cereal crops such as wheat. It has been reported to disrupt eukaryotic sterol biosynthesis pathways, notably by inhibiting fungal Δ14 reductases. It has also been reported to inhibit mammalian sterol biosynthesis by affecting lanosterol demethylation. Although used in agriculture for pest management purposes, it has been reported to have a strong adverse effect on sterol biosynthesis in higher-plants by inhibiting the cycloeucalenol-obtusifoliol isomerase. This inhibition was shown to not only alter the lipid composition of the plasma-membrane, but also impact cell division and growth, in plants.

C-5 sterol desaturase is an enzyme that is highly conserved among eukaryotes and catalyzes the dehydrogenation of a C-5(6) bond in a sterol intermediate compound as a step in the biosynthesis of major sterols. The precise structure of the enzyme's substrate varies by species. For example, the human C-5 sterol desaturase oxidizes lathosterol, while its ortholog ERG3 in the yeast Saccharomyces cerevisiae oxidizes episterol.
Pierre Benveniste, born on 22 December 1937 in Neuilly-sur-Seine, is a French researcher in plant biochemistry and professor at the University of Strasbourg.












