
Acute radiation syndrome (ARS), also known as radiation sickness or radiation poisoning, is a collection of health effects that are caused by being exposed to high amounts of ionizing radiation in a short period of time. Symptoms can start within an hour of exposure, and can last for several months. Early symptoms are usually nausea, vomiting and loss of appetite. In the following hours or weeks, initial symptoms may appear to improve, before the development of additional symptoms, after which either recovery or death follow.

Hormesis is a two-phased dose-response relationship to an environmental agent whereby low-dose amounts have a beneficial effect and high-dose amounts are either inhibitory to function or toxic. Within the hormetic zone, the biological response to low-dose amounts of some stressors is generally favorable. An example is the breathing of oxygen, which is required in low amounts via respiration in living animals, but can be toxic in high amounts, even in a managed clinical setting.

Hibakusha is a word of Japanese origin generally designating the people affected by the atomic bombings of Hiroshima and Nagasaki at the end of World War II.

The linear no-threshold model (LNT) is a dose-response model used in radiation protection to estimate stochastic health effects such as radiation-induced cancer, genetic mutations and teratogenic effects on the human body due to exposure to ionizing radiation. The model statistically extrapolates effects of radiation from very high doses into very low doses, where no biological effects may be observed. The LNT model lies at a foundation of a postulate that all exposure to ionizing radiation is harmful, regardless of how low the dose is, and that the effect is cumulative over lifetime.

Radiation hormesis is the hypothesis that low doses of ionizing radiation are beneficial, stimulating the activation of repair mechanisms that protect against disease, that are not activated in absence of ionizing radiation. The reserve repair mechanisms are hypothesized to be sufficiently effective when stimulated as to not only cancel the detrimental effects of ionizing radiation but also inhibit disease not related to radiation exposure. It has been a mainstream concept since at least 2009.

John William Gofman was an American scientist and advocate. He was Professor Emeritus of Molecular and Cell Biology at the University of California at Berkeley.
Downwinders were individuals and communities in the intermountain between the Cascade and Rocky Mountain ranges primarily in Arizona, Nevada, New Mexico and Utah but also in Oregon, Washington, and Idaho who were exposed to radioactive contamination or nuclear fallout from atmospheric or underground nuclear weapons testing, and nuclear accidents.

On 6 and 9 August 1945, the United States detonated two atomic bombs over the Japanese cities of Hiroshima and Nagasaki respectively. The bombings killed between 129,000 and 226,000 people, most of whom were civilians, and remain the only use of nuclear weapons in an armed conflict. Japan surrendered to the Allies on 15 August, six days after the bombing of Nagasaki and the Soviet Union's declaration of war against Japan and invasion of Japanese-occupied Manchuria. The Japanese government signed the instrument of surrender on 2 September, effectively ending the war.
The Radiation Effects Research Foundation (RERF) is a joint U.S.-Japan research organization responsible for studying the medical effects of radiation and associated diseases in humans for the welfare of the survivors and all humankind. The organization's scientific laboratories are located in Hiroshima and Nagasaki, Japan.
Radiobiology is a field of clinical and basic medical sciences that involves the study of the effects of ionizing radiation on living things, in particular health effects of radiation. Ionizing radiation is generally harmful and potentially lethal to living things but can have health benefits in radiation therapy for the treatment of cancer and thyrotoxicosis. Its most common impact is the induction of cancer with a latent period of years or decades after exposure. High doses can cause visually dramatic radiation burns, and/or rapid fatality through acute radiation syndrome. Controlled doses are used for medical imaging and radiotherapy.

The medical effects of the atomic bomb upon humans can be put into the four categories below, with the effects of larger thermonuclear weapons producing blast and thermal effects so large that there would be a negligible number of survivors close enough to the center of the blast who would experience prompt/acute radiation effects, which were observed after the 16 kiloton yield Hiroshima bomb, due to its relatively low yield:
The effects of the 1979 Three Mile Island nuclear accident are widely agreed to be very low by scientists in the relevant fields. The American Nuclear Society concluded that average local radiation exposure was equivalent to a chest X-ray and maximum local exposure equivalent to less than a year's background radiation. The U.S. BEIR report on the Biological Effects of Ionizing Radiation states that "the collective dose equivalent resulting from the radioactivity released in the Three Mile Island accident was so low that the estimated number of excess cancer cases to be expected, if any were to occur, would be negligible and undetectable." A variety of epidemiology studies have concluded that the accident has had no observable long term health effects. One dissenting study is "a re-evaluation of cancer incidence near the Three Mile Island nuclear plant" by Dr Steven Wing of the University of North Carolina. In this study, Dr Wing and his colleagues argue that earlier findings had "logical and methodological problems" and conclude that "cancer incidence, specifically lung cancer and leukemia, increased following the TMI accident in areas estimated to have been in the pathway of radioactive plumes than in other areas." Other dissenting opinions can be found in the Radiation and Public Health Project, whose leader, Joseph Mangano, has questioned the safety of nuclear power since 1985.
The Chernobyl disaster, considered the worst nuclear disaster in history, occurred on 26 April 1986 at the Chernobyl Nuclear Power Plant in the Ukrainian Soviet Socialist Republic, then part of the Soviet Union, now in Ukraine. From 1986 onward, the total death toll of the disaster has lacked consensus; as peer-reviewed medical journal The Lancet and other sources have noted, it remains contested. There is consensus that a total of approximately 30 people died from immediate blast trauma and acute radiation syndrome (ARS) in the seconds to months after the disaster, respectively, with 60 in total in the decades since, inclusive of later radiation induced cancer. However, there is considerable debate concerning the accurate number of projected deaths that have yet to occur due to the disaster's long-term health effects; long-term death estimates range from up to 4,000 for the most exposed people of Ukraine, Belarus, and Russia, to 16,000 cases in total for all those exposed on the entire continent of Europe, with figures as high as 60,000 when including the relatively minor effects around the globe. Such numbers are based on the heavily contested linear no-threshold model.

Astronauts are exposed to approximately 50-2,000 millisieverts (mSv) while on six-month-duration missions to the International Space Station (ISS), the Moon and beyond. The risk of cancer caused by ionizing radiation is well documented at radiation doses beginning at 100mSv and above.
Exposure to ionizing radiation is known to increase the future incidence of cancer, particularly leukemia. The mechanism by which this occurs is well understood, but quantitative models predicting the level of risk remain controversial. The most widely accepted model posits that the incidence of cancers due to ionizing radiation increases linearly with effective radiation dose at a rate of 5.5% per sievert; if correct, natural background radiation is the most hazardous source of radiation to general public health, followed by medical imaging as a close second. Additionally, the vast majority of non-invasive cancers are non-melanoma skin cancers caused by ultraviolet radiation. Non-ionizing radio frequency radiation from mobile phones, electric power transmission, and other similar sources have been investigated as a possible carcinogen by the WHO's International Agency for Research on Cancer, but to date, no evidence of this has been observed.
Studies with protons and HZE nuclei of relative biological effectiveness for molecular, cellular, and tissue endpoints, including tumor induction, demonstrate risk from space radiation exposure. This evidence may be extrapolated to applicable chronic conditions that are found in space and from the heavy ion beams that are used at accelerators.

William Jackson (Jack) Schull was an American geneticist and Professor Emeritus of Human Genetics at The University of Texas Health Science Center at Houston. He worked for the Atomic Bomb Casualty Commission in Japan, was one of the founding members of the Department of Human Genetics at the University of Michigan, and was the founding director of the Center for Demographic and Population Genetics at the University of Texas at Houston. His scientific contributions include studies on the effects of ionizing radiation on human health, the role of heredity and the interaction of heredity and environment in the etiology of chronic disease, the effects of inbreeding in human populations, the mechanisms of adaptations to hypoxic conditions, and the genetic epidemiology of populations burdened by chronic diseases associated with low socio-economic status.
Travel outside the Earth's protective atmosphere, magnetosphere, and in free fall can harm human health, and understanding such harm is essential for successful crewed spaceflight. Potential effects on the central nervous system (CNS) are particularly important. A vigorous ground-based cellular and animal model research program will help quantify the risk to the CNS from space radiation exposure on future long distance space missions and promote the development of optimized countermeasures.
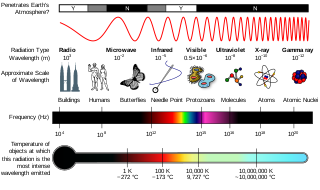
Radiation exposure is a measure of the ionization of air due to ionizing radiation from photons. It is defined as the electric charge freed by such radiation in a specified volume of air divided by the mass of that air. As of 2007, "medical radiation exposure" was defined by the International Commission on Radiological Protection as exposure incurred by people as part of their own medical or dental diagnosis or treatment; by persons, other than those occupationally exposed, knowingly, while voluntarily helping in the support and comfort of patients; and by volunteers in a programme of biomedical research involving their exposure. Common medical tests and treatments involving radiation include X-rays, CT scans, mammography, lung ventilation and perfusion scans, bone scans, cardiac perfusion scan, angiography, radiation therapy, and more. Each type of test carries its own amount of radiation exposure. There are two general categories of adverse health effects caused by radiation exposure: deterministic effects and stochastic effects. Deterministic effects are due to the killing/malfunction of cells following high doses; and stochastic effects involve either cancer development in exposed individuals caused by mutation of somatic cells, or heritable disease in their offspring from mutation of reproductive (germ) cells.

Gilbert Wheeler Beebe was an American epidemiologist and statistician who performed studies of radiation-related mortality and morbidity among those exposed to the atomic bombings of Hiroshima and Nagasaki and the Chernobyl reactor accident.











