Succinyl-coenzyme A, abbreviated as succinyl-CoA or SucCoA, is a thioester of succinic acid and coenzyme A.
In chemistry, homolysis or homolytic fission is the dissociation of a molecular bond by a process where each of the fragments retains one of the originally bonded electrons. During homolytic fission of a neutral molecule with an even number of electrons, two free radicals will be generated. That is, the two electrons involved in the original bond are distributed between the two fragment species. Bond cleavage is also possible by a process called heterolysis.

Methylmalonyl-CoA mutase is a mitochondrial homodimer apoenzyme that focuses on the catalysis of methylmalonyl CoA to succinyl CoA. The enzyme is bound to adenosylcobalamin, a hormonal derivative of vitamin B12 in order to function. Methylmalonyl-CoA mutase deficiency is caused by genetic defect in the MUT gene responsible for encoding the enzyme. Deficiency in this enzyme accounts for 60% of the cases of methylmalonic acidemia.
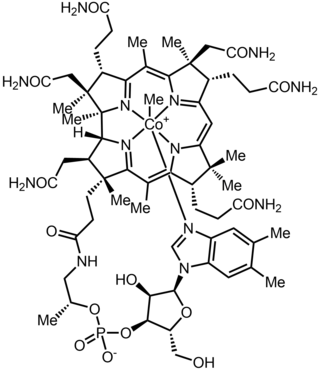
Methylcobalamin (mecobalamin, MeCbl, or MeB12) is a cobalamin, a form of vitamin B12. It differs from cyanocobalamin in that the cyano group at the cobalt is replaced with a methyl group. Methylcobalamin features an octahedral cobalt(III) centre and can be obtained as bright red crystals. From the perspective of coordination chemistry, methylcobalamin is notable as a rare example of a compound that contains metal–alkyl bonds. Nickel–methyl intermediates have been proposed for the final step of methanogenesis.

Phosphoglycerate mutase (PGM) is any enzyme that catalyzes step 8 of glycolysis - the internal transfer of a phosphate group from C-3 to C-2 which results in the conversion of 3-phosphoglycerate (3PG) to 2-phosphoglycerate (2PG) through a 2,3-bisphosphoglycerate intermediate. These enzymes are categorized into the two distinct classes of either cofactor-dependent (dPGM) or cofactor-independent (iPGM). The dPGM enzyme is composed of approximately 250 amino acids and is found in all vertebrates as well as in some invertebrates, fungi, and bacteria. The iPGM class is found in all plants and algae as well as in some invertebrate, fungi, and Gram-positive bacteria. This class of PGM enzyme shares the same superfamily as alkaline phosphatase.

Methylmalonyl-CoA mutase (EC 5.4.99.2, MCM), mitochondrial, also known as methylmalonyl-CoA isomerase, is a protein that in humans is encoded by the MUT gene. This vitamin B12-dependent enzyme catalyzes the isomerization of methylmalonyl-CoA to succinyl-CoA in humans. Mutations in MUT gene may lead to various types of methylmalonic aciduria.
Propionyl-CoA is a coenzyme A derivative of propionic acid. It is composed of a 24 total carbon chain and its production and metabolic fate depend on which organism it is present in. Several different pathways can lead to its production, such as through the catabolism of specific amino acids or the oxidation of odd-chain fatty acids. It later can be broken down by propionyl-CoA carboxylase or through the methylcitrate cycle. In different organisms, however, propionyl-CoA can be sequestered into controlled regions, to alleviate its potential toxicity through accumulation. Genetic deficiencies regarding the production and breakdown of propionyl-CoA also have great clinical and human significance.

Methylmalonyl-CoA is the thioester consisting of coenzyme A linked to methylmalonic acid. It is an important intermediate in the biosynthesis of succinyl-CoA, which plays an essential role in the tricarboxylic acid cycle. The compound is sometimes referred to as "methylmalyl-CoA".

Methylmalonyl CoA epimerase is an enzyme involved in fatty acid catabolism that is encoded in human by the "MCEE" gene located on chromosome 2. It is routinely and incorrectly labeled as "methylmalonyl-CoA racemase". It is not a racemase because the CoA moiety has 5 other stereocenters.

Adenosylcobalamin (AdoCbl), also known as coenzyme B12, cobamamide, and dibencozide, is, along with methylcobalamin (MeCbl), one of the biologically active forms of vitamin B12.
In enzymology, a 2-methyleneglutarate mutase is an enzyme that catalyzes the chemical reaction

In enzymology, D-lysine 5,6-aminomutase is an enzyme that catalyzes the chemical reaction
In enzymology, a methylaspartate mutase is an enzyme that catalyzes the chemical reaction

The enzyme methylisocitrate lyase catalyzes the chemical reaction
The enzyme propanediol dehydratase (EC 4.2.1.28) catalyzes the chemical reaction

Cyanocobalamin is a form of vitamin B
12 used to treat vitamin B
12 deficiency except in the presence of cyanide toxicity. The deficiency may occur in pernicious anemia, following surgical removal of the stomach, with fish tapeworm, or due to bowel cancer. It is less preferred than hydroxocobalamin for treating vitamin B
12 deficiency. Some study have shown that it has an antihypotensive effect. It is used by mouth, by injection into a muscle, or as a nasal spray.

In enzymology, a nicotinate-nucleotide-dimethylbenzimidazole phosphoribosyltransferase is an enzyme that catalyzes the chemical reaction
In molecular biology, the vitamin B12-binding domain is a protein domain which binds to cobalamin. It can bind two different forms of the cobalamin cofactor, with cobalt bonded either to a methyl group (methylcobalamin) or to 5'-deoxyadenosine (adenosylcobalamin). Cobalamin-binding domains are mainly found in two families of enzymes present in animals and prokaryotes, which perform distinct kinds of reactions at the cobalt-carbon bond. Enzymes that require methylcobalamin carry out methyl transfer reactions. Enzymes that require adenosylcobalamin catalyse reactions in which the first step is the cleavage of adenosylcobalamin to form cob(II)alamin and the 5'-deoxyadenosyl radical, and thus act as radical generators. In both types of enzymes the B12-binding domain uses a histidine to bind the cobalt atom of cobalamin cofactors. This histidine is embedded in a DXHXXG sequence, the most conserved primary sequence motif of the domain. Proteins containing the cobalamin-binding domain include:

Cobalamin biosynthesis is the process by which bacteria and archea make cobalamin, vitamin B12. Many steps are involved in converting aminolevulinic acid via uroporphyrinogen III and adenosylcobyric acid to the final forms in which it is used by enzymes in both the producing organisms and other species, including humans who acquire it through their diet.
Odd-chain fatty acids are those fatty acids that contain an odd number of carbon atoms. In addition to being classified according to their saturation or unsaturation, fatty acids are also classified according to their odd or even numbers of constituent carbon atoms. With respect to natural abundance, most fatty acids are even chain, e.g. palmitic (C16) and stearic (C18). In terms of physical properties, odd and even fatty acids are similar, generally being colorless, soluble in alcohols, and often somewhat oily. The odd-chain fatty acids are biosynthesized and metabolized slightly differently from the even-chained relatives. In addition to the usual C12-C22 long chain fatty acids, some very long chain fatty acids (VLCFAs) are also known. Some of these VLCFAs are also of the odd-chain variety.











