This article may require cleanup to meet Wikipedia's quality standards. The specific problem is: Need to explain the deletion of EC due to being "covered by EC 7.1.1.2" (the ubiquinone one). Such a merge makes sense for some proteins tagged with that EC, but definitely does not make sense for this article.(April 2022) |
| NADH dehydrogenase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 7.1.1.2 | ||||||||
| CAS no. | 9079-67-8 | ||||||||
| Alt. names | cytochrome c reductase, type 1 dehydrogenase, beta-NADH dehydrogenase dinucleotide, diaphorase, dihydrocodehydrogenase I dehydrogenase, dihydronicotinamide adenine dinucleotide dehydrogenase, diphosphopyridine diaphorase, DPNH diaphorase, NADH diaphorase, NADH hydrogenase, NADH oxidoreductase, NADH-menadione oxidoreductase, reduced diphosphopyridine nucleotide diaphorase [1] | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
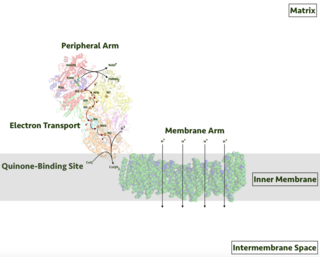
NADH dehydrogenase is an enzyme that converts nicotinamide adenine dinucleotide (NAD) from its reduced form (NADH) to its oxidized form (NAD+). Members of the NADH dehydrogenase family and analogues are commonly systematically named using the format NADH:acceptor oxidoreductase. [2] [3] [4] [5] The chemical reaction these enzymes catalyze is generally represented with the following equation:
- NADH + H+ + acceptor ⇌ NAD+ + reduced acceptor
NADH dehydrogenase is a flavoprotein that contains iron-sulfur centers.
NADH dehydrogenase is used in the electron transport chain for generation of ATP.
The EC term NADH dehydrogenase (quinone) (EC 1.6.5.11) is defined for NADH dehydrogenases that use a quinone (excluding ubiquinone) as the acceptor. The EC term NADH dehydrogenase (ubiquinone) (EC 7.1.1.2) is defined for those with ubiquinone as the acceptor.