Phenotype
Naive T cells are commonly characterized by the surface expression of L-selectin (CD62L) and C-C Chemokine receptor type 7 (CCR7); the absence of the activation markers CD25, CD44 or CD69; and the absence of memory CD45RO isoform. [1] [2] They also express functional IL-7 receptors, consisting of subunits IL-7 receptor-α, CD127, and common-γ chain, CD132. In the naive state, T cells are thought to require the common-gamma chain cytokines IL-7 and IL-15 for homeostatic survival mechanisms. [3] While naive T cells are regularly regarded as a developmentally synchronized and fairly homogeneous and quiescent cell population, only differing in T cell receptor specificity, there is increasing evidence that naive T cells are actually heterogeneous in phenotype, function, dynamics and differentiation status, resulting in a whole spectrum of naive cells with different properties. [2] For instance, some non-naive T cells express surface markers similar to naive T cells (Tscm, stem cell memory T cells; [4] Tmp, memory T cells with a naive phenotype [5] ), some antigen-naive T cells have lost their naive phenotype, [6] and some T cells are incorporated within the naive T cell phenotype but are a different T cell subset (Treg, regulatory T cells; RTE, Recent Thymic emigrant). [2] The majority of human naive T cells are produced very early in life when the thymus is large and functional. The subsequent decrease in naive T cell production due to involution of the thymus with age is compensated by so called "peripheral proliferation" or "homeostatic proliferation" of naive T cells which have emigrated from the thymus earlier in life. Homeostatic proliferation causes change to naive T cell gene expression and is manifested by surface expression of CD25.
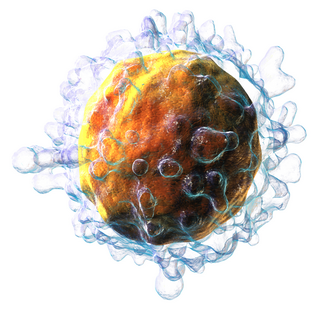
T cells are one of the important types of white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell receptor (TCR) on their cell surface.

A cytotoxic T cell (also known as TC, cytotoxic T lymphocyte, CTL, T-killer cell, cytolytic T cell, CD8+ T-cell or killer T cell) is a T lymphocyte (a type of white blood cell) that kills cancer cells, cells that are infected by intracellular pathogens such as viruses or bacteria, or cells that are damaged in other ways.

The T helper cells (Th cells), also known as CD4+ cells or CD4-positive cells, are a type of T cell that play an important role in the adaptive immune system. They aid the activity of other immune cells by releasing cytokines. They are considered essential in B cell antibody class switching, breaking cross-tolerance in dendritic cells, in the activation and growth of cytotoxic T cells, and in maximizing bactericidal activity of phagocytes such as macrophages and neutrophils. CD4+ cells are mature Th cells that express the surface protein CD4. Genetic variation in regulatory elements expressed by CD4+ cells determines susceptibility to a broad class of autoimmune diseases.
The regulatory T cells (Tregs or Treg cells), formerly known as suppressor T cells, are a subpopulation of T cells that modulate the immune system, maintain tolerance to self-antigens, and prevent autoimmune disease. Treg cells are immunosuppressive and generally suppress or downregulate induction and proliferation of effector T cells. Treg cells express the biomarkers CD4, FOXP3, and CD25 and are thought to be derived from the same lineage as naïve CD4+ cells. Because effector T cells also express CD4 and CD25, Treg cells are very difficult to effectively discern from effector CD4+, making them difficult to study. Research has found that the cytokine transforming growth factor beta (TGF-β) is essential for Treg cells to differentiate from naïve CD4+ cells and is important in maintaining Treg cell homeostasis.

The T-cell receptor (TCR) is a protein complex found on the surface of T cells, or T lymphocytes, that is responsible for recognizing fragments of antigen as peptides bound to major histocompatibility complex (MHC) molecules. The binding between TCR and antigen peptides is of relatively low affinity and is degenerate: that is, many TCRs recognize the same antigen peptide and many antigen peptides are recognized by the same TCR.
In immunology, central tolerance is the process of eliminating any developing T or B lymphocytes that are autoreactive, i.e. reactive to the body itself. Through elimination of autoreactive lymphocytes, tolerance ensures that the immune system does not attack self peptides. Lymphocyte maturation occurs in primary lymphoid organs such as the bone marrow and the thymus. In mammals, B cells mature in the bone marrow and T cells mature in the thymus.
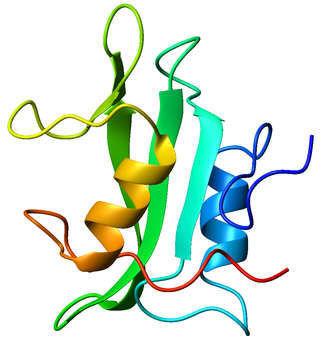
Tyrosin-protein kinase Lck is a 56 kDa protein that is found inside lymphocytes and encoded in the human by the LCK gene. The Lck is a member of Src kinase family (SFK) and is important for the activation of T-cell receptor (TCR) signaling in both naive T cells and effector T cells. The role of Lck is less prominent in the activation or in the maintenance of memory CD8 T cells in comparison to CD4 T cells. In addition, the constitutive activity of the mouse Lck homolog varies among memory T cell subsets. It seems that in mice, in the effector memory T cell (TEM) population, more than 50% of Lck is present in a constitutively active conformation, whereas less than 20% of Lck is present as active form in central memory T cells. These differences are due to differential regulation by SH2 domain–containing phosphatase-1 (Shp-1) and C-terminal Src kinase.
Memory T cells are a subset of T lymphocytes that might have some of the same functions as memory B cells. Their lineage is unclear.
Immune tolerance, also known as immunological tolerance or immunotolerance, refers to the immune system's state of unresponsiveness to substances or tissues that would otherwise trigger an immune response. It arises from prior exposure to a specific antigen and contrasts the immune system's conventional role in eliminating foreign antigens. Depending on the site of induction, tolerance is categorized as either central tolerance, occurring in the thymus and bone marrow, or peripheral tolerance, taking place in other tissues and lymph nodes. Although the mechanisms establishing central and peripheral tolerance differ, their outcomes are analogous, ensuring immune system modulation.

CD3 is a protein complex and T cell co-receptor that is involved in activating both the cytotoxic T cell and T helper cells. It is composed of four distinct chains. In mammals, the complex contains a CD3γ chain, a CD3δ chain, and two CD3ε chains. These chains associate with the T-cell receptor (TCR) and the CD3-zeta (ζ-chain) to generate an activation signal in T lymphocytes. The TCR, CD3-zeta, and the other CD3 molecules together constitute the TCR complex.
MHC-restricted antigen recognition, or MHC restriction, refers to the fact that a T cell can interact with a self-major histocompatibility complex molecule and a foreign peptide bound to it, but will only respond to the antigen when it is bound to a particular MHC molecule.
Intraepithelial lymphocytes (IEL) are lymphocytes found in the epithelial layer of mammalian mucosal linings, such as the gastrointestinal (GI) tract and reproductive tract. However, unlike other T cells, IELs do not need priming. Upon encountering antigens, they immediately release cytokines and cause killing of infected target cells. In the GI tract, they are components of gut-associated lymphoid tissue (GALT).
Immunosenescence is the gradual deterioration of the immune system, brought on by natural age advancement. A 2020 review concluded that the adaptive immune system is affected more than the innate immune system. Immunosenescence involves both the host's capacity to respond to infections and the development of long-term immune memory. Age-associated immune deficiency is found in both long- and short-lived species as a function of their age relative to life expectancy rather than elapsed time.
In immunology, peripheral tolerance is the second branch of immunological tolerance, after central tolerance. It takes place in the immune periphery. Its main purpose is to ensure that self-reactive T and B cells which escaped central tolerance do not cause autoimmune disease. Peripheral tolerance can also serve a purpose in preventing an immune response to harmless food antigens and allergens.

Lymphocyte-activation gene 3, also known as LAG-3, is a protein which in humans is encoded by the LAG3 gene. LAG3, which was discovered in 1990 and was designated CD223 after the Seventh Human Leucocyte Differentiation Antigen Workshop in 2000, is a cell surface molecule with diverse biological effects on T cell function but overall has an immune inhibitory effect. It is an immune checkpoint receptor and as such is the target of various drug development programs by pharmaceutical companies seeking to develop new treatments for cancer and autoimmune disorders. In soluble form it is also being developed as a cancer drug in its own right.

B- and T-lymphocyte attenuator or BTLA is a protein that belongs to the CD28 immunoglobulin superfamily (IgSF) which is encoded by the BTLA gene located on the 3rd human chromosome. BTLA was first discovered in 2003 as an inhibitor of Th1 expansion and it became the 3rd member of the CD28 IgSF. However, its discovered ligand herpes virus entry mediator or HVEM belongs to the tumor necrosis factor receptor superfamily (TNFRSF). This finding was surprising because until the discovery of HVEM it was believed that receptors and ligands always belong to the same family.

The interleukin-2 receptor alpha chain is a protein involved in the assembly of the high-affinity interleukin-2 receptor, consisting of alpha (IL2RA), beta (IL2RB) and the common gamma chain (IL2RG). As the name indicates, this receptor interacts with interleukin-2, a pleiotropic cytokine which plays an important role in immune homeostasis.
Mucosal-associated invariant T cells make up a subset of T cells in the immune system that display innate, effector-like qualities. In humans, MAIT cells are found in the blood, liver, lungs, and mucosa, defending against microbial activity and infection. The MHC class I-like protein, MR1, is responsible for presenting bacterially-produced vitamin B2 and B9 metabolites to MAIT cells. After the presentation of foreign antigen by MR1, MAIT cells secrete pro-inflammatory cytokines and are capable of lysing bacterially-infected cells. MAIT cells can also be activated through MR1-independent signaling. In addition to possessing innate-like functions, this T cell subset supports the adaptive immune response and has a memory-like phenotype. Furthermore, MAIT cells are thought to play a role in autoimmune diseases, such as multiple sclerosis, arthritis and inflammatory bowel disease, although definitive evidence is yet to be published.
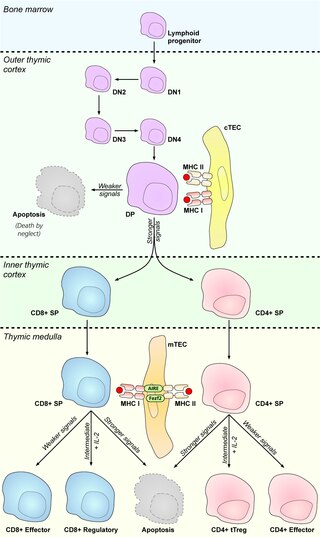
Cortical thymic epithelial cells (cTECs) form unique parenchyma cell population of the thymus which critically contribute to the development of T cells.
Virtual memory T cells (TVM) are a subtype of T lymphocytes. These are cells that have a memory phenotype but have not been exposed to a foreign antigen. They are classified as memory cells but do not have an obvious memory function. They were first observed and described in 2009. The name comes from a computerized "virtual memory" that describes a working memory based on an alternative use of an existing space.









