
Homocysteine or Hcy: is a non-proteinogenic α-amino acid. It is a homologue of the amino acid cysteine, differing by an additional methylene bridge (-CH2-). It is biosynthesized from methionine by the removal of its terminal Cε methyl group. In the body, homocysteine can be recycled into methionine or converted into cysteine with the aid of vitamin B6, B9, and B12.
In biology, quorum sensing or quorum signaling (QS) is the ability to detect and respond to cell population density by gene regulation. Quorum sensing is a type of cellular signaling, and more specifically can be considered a type of paracrine signaling. However, it also contains traits of both autocrine signaling: a cell produces both the autoinducer molecule and the receptor for the autoinducer. As one example, QS enables bacteria to restrict the expression of specific genes to the high cell densities at which the resulting phenotypes will be most beneficial, especially for phenotypes that would be ineffective at low cell densities and therefore too energetically costly to express. Many species of bacteria use quorum sensing to coordinate gene expression according to the density of their local population. In a similar fashion, some social insects use quorum sensing to determine where to nest. Quorum sensing in pathogenic bacteria activates host immune signaling and prolongs host survival, by limiting the bacterial intake of nutrients, such as tryptophan, which further is converted to serotonin. As such, quorum sensing allows a commensal interaction between host and pathogenic bacteria. Quorum sensing may also be useful for cancer cell communications.
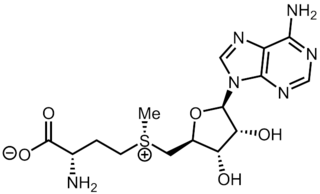
S-Adenosyl methionine (SAM), also known under the commercial names of SAMe, SAM-e, or AdoMet, is a common cosubstrate involved in methyl group transfers, transsulfuration, and aminopropylation. Although these anabolic reactions occur throughout the body, most SAM is produced and consumed in the liver. More than 40 methyl transfers from SAM are known, to various substrates such as nucleic acids, proteins, lipids and secondary metabolites. It is made from adenosine triphosphate (ATP) and methionine by methionine adenosyltransferase. SAM was first discovered by Giulio Cantoni in 1952.

Aliivibrio fischeri is a Gram-negative, rod-shaped bacterium found globally in marine environments. This species has bioluminescent properties, and is found predominantly in symbiosis with various marine animals, such as the Hawaiian bobtail squid. It is heterotrophic, oxidase-positive, and motile by means of a single polar flagella. Free-living A. fischeri cells survive on decaying organic matter. The bacterium is a key research organism for examination of microbial bioluminescence, quorum sensing, and bacterial-animal symbiosis. It is named after Bernhard Fischer, a German microbiologist.

Vibrio harveyi is a Gram-negative, bioluminescent, marine bacterium in the genus Vibrio. V. harveyi is rod-shaped, motile, facultatively anaerobic, halophilic, and competent for both fermentative and respiratory metabolism. It does not grow below 4 °C. V. harveyi can be found free-swimming in tropical marine waters, commensally in the gut microflora of marine animals, and as both a primary and opportunistic pathogen of marine animals, including Gorgonian corals, oysters, prawns, lobsters, the common snook, barramundi, turbot, milkfish, and seahorses. It is responsible for luminous vibriosis, a disease that affects commercially farmed penaeid prawns. Additionally, based on samples taken by ocean-going ships, V. harveyi is thought to be the cause of the milky seas effect, in which, during the night, a uniform blue glow is emitted from the seawater. Some glows can cover nearly 6,000 sq mi (16,000 km2).

N-Acyl homoserine lactones are a class of signaling molecules involved in bacterial quorum sensing, a means of communication between bacteria enabling behaviors based on population density.

Eikenella corrodens is a Gram-negative facultative anaerobic bacillus that can cause severe invasive disease in humans. It was first identified by M. Eiken in 1958, who called it Bacteroides corrodens. E. corrodens is a rare pericarditis associated pathogen. It is a fastidious, slow growing, human commensal bacillus, capable of acting as an opportunistic pathogen and causing abscesses in several anatomical sites, including the liver, lung, spleen, and submandibular region. E. corrodens could independently cause serious infection in both immunocompetent and immunocompromised hosts.
In enzymology, a hexaprenyldihydroxybenzoate methyltransferase is an enzyme that catalyzes the chemical reaction

Cystathionine beta-lyase, also commonly referred to as CBL or β-cystathionase, is an enzyme that primarily catalyzes the following α,β-elimination reaction
Autoinducers are signaling molecules that are produced in response to changes in cell-population density. As the density of quorum sensing bacterial cells increases so does the concentration of the autoinducer. Detection of signal molecules by bacteria acts as stimulation which leads to altered gene expression once the minimal threshold is reached. Quorum sensing is a phenomenon that allows both Gram-negative and Gram-positive bacteria to sense one another and to regulate a wide variety of physiological activities. Such activities include symbiosis, virulence, motility, antibiotic production, and biofilm formation. Autoinducers come in a number of different forms depending on the species, but the effect that they have is similar in many cases. Autoinducers allow bacteria to communicate both within and between different species. This communication alters gene expression and allows bacteria to mount coordinated responses to their environments, in a manner that is comparable to behavior and signaling in higher organisms. Not surprisingly, it has been suggested that quorum sensing may have been an important evolutionary milestone that ultimately gave rise to multicellular life forms.
In enzymology, an adenosylhomocysteine nucleosidase (EC 3.2.2.9) is an enzyme that catalyzes the chemical reaction

Autoinducer-2 (AI-2), a furanosyl borate diester or tetrahydroxy furan, is a member of a family of signaling molecules used in quorum sensing. AI-2 is one of only a few known biomolecules incorporating boron. First identified in the marine bacterium Vibrio harveyi, AI-2 is produced and recognized by many Gram-negative and Gram-positive bacteria. AI-2 arises by the reaction of 4,5-dihydroxy-2,3-pentanedione, which is produced enzymatically, with boric acid and is recognized by the two-component sensor kinase LuxPQ in Vibrionaceae.
Interspecies quorum sensing is a type of quorum sensing in which bacteria send and receive signals to other species besides their own. This is accomplished by the secretion of signaling molecules which trigger a response in nearby bacteria at high enough concentrations. Once the molecule hits a certain concentration it triggers the transcription of certain genes such as virulence factors. It has been discovered that bacteria can not only interact via quorum sensing with members of their own species but that there is a kind of universal molecule that allows them to gather information about other species as well. This universal molecule is called autoinducer 2 or AI-2.

Uroporphyrinogen-III C-methyltransferase, uroporphyrinogen methyltransferase, uroporphyrinogen-III methyltransferase, adenosylmethionine-uroporphyrinogen III methyltransferase, S-adenosyl-L-methionine-dependent uroporphyrinogen III methylase, uroporphyrinogen-III methylase, SirA, CysG, CobA, uroporphyrin-III C-methyltransferase, S-adenosyl-L-methionine:uroporphyrin-III C-methyltransferase) is an enzyme with systematic name S-adenosyl-L-methionine:uroporphyrinogen-III C-methyltransferase. This enzyme catalyses the following chemical reaction
Acyl-homoserine-lactone synthase is an enzyme with systematic name acyl-(acyl-carrier protein):S-adenosyl-L-methionine acyltranserase . This enzyme catalyses the following chemical reaction
Gellan lyase is an enzyme with systematic name gellan β-D-glucopyranosyl-(1→4)-D-glucopyranosyluronate lyase. This enzyme catalyses the following process:

Bioluminescent bacteria are light-producing bacteria that are predominantly present in sea water, marine sediments, the surface of decomposing fish and in the gut of marine animals. While not as common, bacterial bioluminescence is also found in terrestrial and freshwater bacteria. These bacteria may be free living or in symbiosis with animals such as the Hawaiian Bobtail squid or terrestrial nematodes. The host organisms provide these bacteria a safe home and sufficient nutrition. In exchange, the hosts use the light produced by the bacteria for camouflage, prey and/or mate attraction. Bioluminescent bacteria have evolved symbiotic relationships with other organisms in which both participants benefit close to equally. Another possible reason bacteria use luminescence reaction is for quorum sensing, an ability to regulate gene expression in response to bacterial cell density.
Everett Peter Greenberg is an American microbiologist. He is the inaugural Eugene and Martha Nester Professor of Microbiology at the Department of Microbiology of the University of Washington School of Medicine. He is best known for his research on quorum sensing, and has received multiple awards for his work.

4,5-Dihydroxy-2,3-pentanedione (DPD) is an organic compound that occurs naturally but exists as several related structures. The idealized formula for this species is CH3C(O)C(O)CH(OH)CH2OH, but it is known to exist as several other forms resulting from cyclization. It is not stable at room temperature as a pure material, which has further complicated its analysis. The (S)-stereoisomer occurs naturally. It is typically hydrated, i.e., one keto group has added water to give the geminal diol.











