Related Research Articles

Poliovirus, the causative agent of polio, is a serotype of the species Enterovirus C, in the family of Picornaviridae. There are three poliovirus serotypes: types 1, 2, and 3.

Rhabdoviridae is a family of negative-strand RNA viruses in the order Mononegavirales. Vertebrates, invertebrates, plants, fungi and protozoans serve as natural hosts. Diseases associated with member viruses include rabies encephalitis caused by the rabies virus, and flu-like symptoms in humans caused by vesiculoviruses. The name is derived from Ancient Greek rhabdos, meaning rod, referring to the shape of the viral particles. The family has 40 genera, most assigned to three subfamilies.

Defective interfering particles (DIPs), also known as defective interfering viruses, are spontaneously generated virus mutants in which a critical portion of the particle's genome has been lost due to defective replication or non-homologous recombination. The mechanism of their formation is presumed to be as a result of template-switching during replication of the viral genome, although non-replicative mechanisms involving direct ligation of genomic RNA fragments have also been proposed. DIPs are derived from and associated with their parent virus, and particles are classed as DIPs if they are rendered non-infectious due to at least one essential gene of the virus being lost or severely damaged as a result of the defection. A DIP can usually still penetrate host cells, but requires another fully functional virus particle to co-infect a cell with it, in order to provide the lost factors.
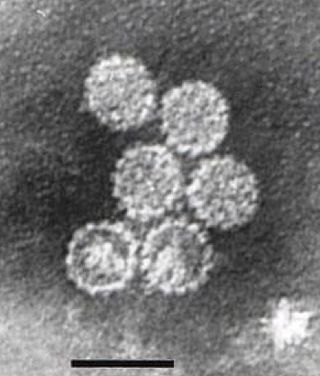
Papillomaviridae is a family of non-enveloped DNA viruses whose members are known as papillomaviruses. Several hundred species of papillomaviruses, traditionally referred to as "types", have been identified infecting all carefully inspected mammals, but also other vertebrates such as birds, snakes, turtles and fish. Infection by most papillomavirus types, depending on the type, is either asymptomatic or causes small benign tumors, known as papillomas or warts. Papillomas caused by some types, however, such as human papillomaviruses 16 and 18, carry a risk of becoming cancerous.
Polyomaviridae is a family of viruses whose natural hosts are primarily mammals and birds. As of 2020, there are six recognized genera and 117 species, five of which are unassigned to a genus. 14 species are known to infect humans, while others, such as Simian Virus 40, have been identified in humans to a lesser extent. Most of these viruses are very common and typically asymptomatic in most human populations studied. BK virus is associated with nephropathy in renal transplant and non-renal solid organ transplant patients, JC virus with progressive multifocal leukoencephalopathy, and Merkel cell virus with Merkel cell cancer.

Dengue virus (DENV) is the cause of dengue fever. It is a mosquito-borne, single positive-stranded RNA virus of the family Flaviviridae; genus Flavivirus. Four serotypes of the virus have been found, and a reported fifth has yet to be confirmed, all of which can cause the full spectrum of disease. Nevertheless, scientists' understanding of dengue virus may be simplistic as, rather than distinct antigenic groups, a continuum appears to exist. This same study identified 47 strains of dengue virus. Additionally, coinfection with and lack of rapid tests for Zika virus and chikungunya complicate matters in real-world infections.
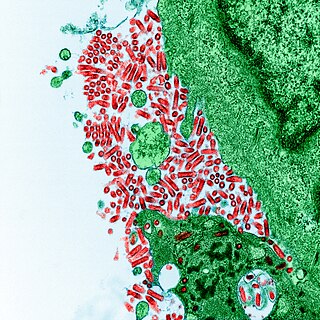
Rabies virus, scientific name Rabies lyssavirus, is a neurotropic virus that causes rabies in animals, including humans. Rabies transmission can occur through the saliva of animals and less commonly through contact with human saliva. Rabies lyssavirus, like many rhabdoviruses, has an extremely wide host range. In the wild it has been found infecting many mammalian species, while in the laboratory it has been found that birds can be infected, as well as cell cultures from mammals, birds, reptiles and insects. Rabies is reported in more than 150 countries and on all continents except Antarctica. The main burden of disease is reported in Asia and Africa, but some cases have been reported also in Europe in the past 10 years, especially in returning travellers.

Birnaviridae is a family of double-stranded RNA viruses. Salmonid fish, birds and insects serve as natural hosts. There are currently 11 species in this family, divided among seven genera. Diseases associated with this family include infectious pancreatic necrosis in salmonid fish, which causes significant losses to the aquaculture industry, with chronic infection in adult salmonid fish and acute viral disease in young salmonid fish.

An arenavirus is a bi- or trisegmented ambisense RNA virus that is a member of the family Arenaviridae. These viruses infect rodents and occasionally humans. A class of novel, highly divergent arenaviruses, properly known as reptarenaviruses, have also been discovered which infect snakes to produce inclusion body disease. At least eight arenaviruses are known to cause human disease. The diseases derived from arenaviruses range in severity. Aseptic meningitis, a severe human disease that causes inflammation covering the brain and spinal cord, can arise from the lymphocytic choriomeningitis virus. Hemorrhagic fever syndromes, including Lassa fever, are derived from infections such as Guanarito virus, Junin virus, Lassa virus, Lujo virus, Machupo virus, Sabia virus, or Whitewater Arroyo virus. Because of the epidemiological association with rodents, some arenaviruses and bunyaviruses are designated as roboviruses.
GB virus C (GBV-C), formerly known as hepatitis G virus (HGV) and also known as human pegivirus – HPgV is a virus in the family Flaviviridae and a member of the Pegivirus, is known to infect humans, but is not known to cause human disease. Reportedly, HIV patients coinfected with GBV-C can survive longer than those without GBV-C, but the patients may be different in other ways. Research is active into the virus' effects on the immune system in patients coinfected with GBV-C and HIV.
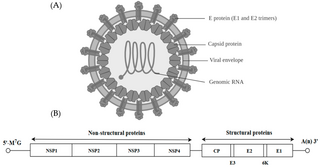
Alphavirus is a genus of RNA viruses, the sole genus in the Togaviridae family. Alphaviruses belong to group IV of the Baltimore classification of viruses, with a positive-sense, single-stranded RNA genome. There are 32 alphaviruses, which infect various vertebrates such as humans, rodents, fish, birds, and larger mammals such as horses, as well as invertebrates. Alphaviruses that could infect both vertebrates and arthropods are referred dual-host alphaviruses, while insect-specific alphaviruses such as Eilat virus and Yada yada virus are restricted to their competent arthropod vector. Transmission between species and individuals occurs mainly via mosquitoes, making the alphaviruses a member of the collection of arboviruses – or arthropod-borne viruses. Alphavirus particles are enveloped, have a 70 nm diameter, tend to be spherical, and have a 40 nm isometric nucleocapsid.

Herpes simplex virus1 and 2, also known by their taxonomic names Human alphaherpesvirus 1 and Human alphaherpesvirus 2, are two members of the human Herpesviridae family, a set of viruses that produce viral infections in the majority of humans. Both HSV-1 and HSV-2 are very common and contagious. They can be spread when an infected person begins shedding the virus.
HIV superinfection is a condition in which a person with an established human immunodeficiency virus infection acquires a second strain of HIV, often of a different subtype. These can form a recombinant strain that co-exists with the strain from the initial infection, as well from reinfection with a new virus strain, and may cause more rapid disease progression or carry multiple resistances to certain HIV medications.

Antibody-dependent enhancement (ADE), sometimes less precisely called immune enhancement or disease enhancement, is a phenomenon in which binding of a virus to suboptimal antibodies enhances its entry into host cells, followed by its replication. The suboptimal antibodies can result from natural infection or from vaccination. ADE may cause enhanced respiratory disease, but is not limited to respiratory disease. It has been observed in HIV, RSV virus and Dengue virus and is monitored for in vaccine development.
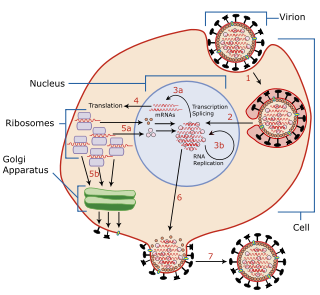
Viral shedding is the expulsion and release of virus progeny following successful reproduction during a host cell infection. Once replication has been completed and the host cell is exhausted of all resources in making viral progeny, the viruses may begin to leave the cell by several methods.
In the diagnostic laboratory, virus infections can be confirmed by a myriad of methods. Diagnostic virology has changed rapidly due to the advent of molecular techniques and increased clinical sensitivity of serological assays.
Lactate dehydrogenase elevating virus (LDV) constitutes the species Gamamaarterivirus lacdeh which is part of the family Arteriviridae and order Nidovirales. The order Nidovirales also includes the family of coronaviruses. Arteriviruses infect macrophages in animals and cause a variety of diseases. LDV specifically causes lifelong persistent viremia in mice, but does not harm the host and only slightly harms the immune system. The main clinical sign is an increased level of the plasma enzyme lactate dehydrogenase (LDH). LDV has a remarkably narrow cell type specificity, meaning nothing homologous with LDV in mice has been found in another species.
Pseudotyping is the process of producing viruses or viral vectors in combination with foreign viral envelope proteins. The result is a pseudotyped virus particle, also called a pseudovirus. With this method, the foreign viral envelope proteins can be used to alter host tropism or increase or decrease the stability of the virus particles. Pseudotyped particles do not carry the genetic material to produce additional viral envelope proteins, so the phenotypic changes cannot be passed on to progeny viral particles. In some cases, the inability to produce viral envelope proteins renders the pseudovirus replication incompetent. In this way, the properties of dangerous viruses can be studied in a lower risk setting.
Infections of the hepatitis C virus (HCV) in children and pregnant women are less understood than those in other adults. Worldwide, the prevalence of HCV infection in pregnant women and children has been estimated to 1-8% and 0.05-5% respectively. The vertical transmission rate has been estimated to be 3-5% and there is a high rate of spontaneous clearance (25-50%) in the children. Higher rates have been reported for both vertical transmission. and prevalence in children (15%).
Cache Valley orthobunyavirus (CVV) is a member of the order Bunyavirales, genus Orthobunyavirus, and serogroup Bunyamwera, which was first isolated in 1956 from Culiseta inornata mosquitos collected in Utah's Cache Valley. CVV is an enveloped arbovirus, nominally 80–120 nm in diameter, whose genome is composed of three single-stranded, negative-sense RNA segments. The large segment of related bunyaviruses is approximately 6800 bases in length and encodes a probable viral polymerase. The middle CVV segment has a 4463-nucleotide sequence and the smallest segment encodes for the nucleocapsid, and a second non-structural protein. CVV has been known to cause outbreaks of spontaneous abortion and congenital malformations in ruminants such as sheep and cattle. CVV rarely infects humans, but when they are infected it has caused encephalitis and multiorgan failure.
References
- ↑ Ryan KJ, Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. p. 881. ISBN 978-0-8385-8529-0.
- ↑ Burrell CJ, Howard CR, Murphy FA (2017-01-01), Burrell CJ, Howard CR, Murphy FA (eds.), "Chapter 7 - Pathogenesis of Virus Infections", Fenner and White's Medical Virology (Fifth Edition), London: Academic Press, pp. 77–104, doi:10.1016/b978-0-12-375156-0.00007-2, ISBN 978-0-12-375156-0, PMC 7150039
- ↑ Cerino A, Bissolati M, Cividini A, Nicosia A, Esumi M, Hayashi N, Mizuno K, Slobbe R, Oudshoorn P, Silini E, Asti M, Mondelli MU (Jan 1997). "Antibody responses to the hepatitis C virus E2 protein: relationship to viraemia and prevalence in anti-HCV seronegative subjects". J Med Virol. 51 (1): 1–5. doi:10.1002/(sici)1096-9071(199701)51:1<1::aid-jmv1>3.3.co;2-3. PMID 8986942.
- ↑ Lodmell DL, Dimcheff DE, Ewalt LC (Mar 2006). "Viral RNA in the bloodstream suggests viremia occurs in clinically ill rabies-infected mice". Virus Res. (Submitted manuscript). 116 (1–2): 114–8. doi:10.1016/j.virusres.2005.09.004. PMID 16242805.
- ↑ Gribencha SV, Barinsky IF (Jul 1982). "Viraemia in rabies". Acta Virol. 26 (4): 301. PMID 6127940.
- ↑ Mulupuri P, Zimmerman JJ, Hermann J, Johnson CR, Cano JP, Yu W, Dee SA, Murtaugh MP (2007-12-10). "Antigen-Specific B-Cell Responses to Porcine Reproductive and Respiratory Syndrome Virus Infection". Journal of Virology . 82 (1): 358–370. doi: 10.1128/JVI.01023-07 . ISSN 0022-538X. PMC 2224379 . PMID 17942527.
- ↑ Lai CJ, Goncalvez AP, Men R, Wernly C, Donau O, Engle RE, Purcell RH (Dec 2007). "Epitope determinants of a chimpanzee dengue virus type 4 (DENV-4)-neutralizing antibody and protection against DENV-4 challenge in mice and rhesus monkeys by passively transferred humanized antibody". J Virol. 81 (23): 12766–74. doi:10.1128/jvi.01420-07. PMC 2169078 . PMID 17881450.