
Dimethoxybromoamphetamine (DOB), also known as brolamfetamine (INN) and bromo-DMA, is a psychedelic drug and substituted amphetamine of the phenethylamine class of compounds. DOB was first synthesized by Alexander Shulgin in 1967. Its synthesis and effects are documented in Shulgin's book PiHKAL: A Chemical Love Story.

3,4-Methylenedioxyamphetamine (MDA), known commonly as sass, is an empathogen-entactogen, psychostimulant, and psychedelic drug of the amphetamine family that is encountered mainly as a recreational drug. In terms of pharmacology, MDA acts most importantly as a serotonin-norepinephrine-dopamine releasing agent (SNDRA). In most countries, the drug is a controlled substance and its possession and sale are illegal.
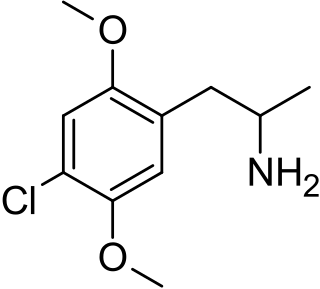
2,5-Dimethoxy-4-chloroamphetamine (DOC) is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was presumably first synthesized by Alexander Shulgin, and was described in his book PiHKAL.

2C-TFM is a psychedelic phenethylamine of the 2C family. It was first synthesized in the laboratory of David E. Nichols. It has also been called 2C-CF3, a name derived from the Para-trifluoromethyl group it contains.
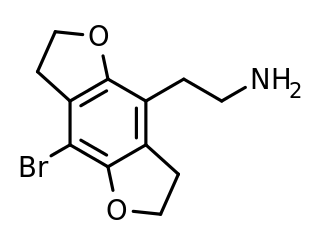
2C-B-FLY is a psychedelic phenethylamine of the 2C family. It was first synthesized in 1996 by Aaron P. Monte.

2,5-Dimethoxy-4-ethylamphetamine is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin, and was described in his book PiHKAL.

Aleph is a psychedelic hallucinogenic drug and a substituted amphetamine of the phenethylamine class of compounds, which can be used as an entheogen. It was first synthesized by Alexander Shulgin. In his book PiHKAL, Shulgin lists the dosage range as 5–10 mg. According to Shulgin, the effects of aleph typically last for 6 to 8 hours.

2C-H (2,5-dimethoxyphenethylamine) is a lesser-known substituted phenethylamine of the 2C family.
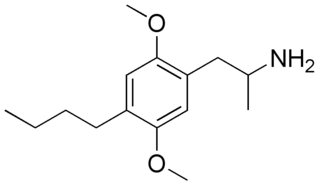
2,5-Dimethoxy-4-butylamphetamine (DOBU) is a lesser-known psychedelic drug and a substituted Amphetamine. DOBU was first synthesized by Alexander Shulgin. In his book PiHKAL (Phenethylamines i Have Known And Loved), only low dosages of 2–3 mg were tested, with the duration simply listed as "very long". DOBU produces paresthesia and difficulty sleeping, but with few other effects. Compared to shorter chain homologues such as DOM, DOET and DOPR which are all potent hallucinogens, DOBU has an even stronger 5-HT2 binding affinity but fails to substitute for hallucinogens in animals or produce hallucinogenic effects in humans, suggesting it has low efficacy and is thus an antagonist or weak partial agonist at the 5-HT2A receptor.

Dimethoxy-4-amylamphetamine (DOAM) is a lesser-known psychedelic drug and a substituted amphetamine. DOAM was first synthesized by Alexander Shulgin. In his book PiHKAL (Phenethylamines i Have Known And Loved), the minimum dosage is listed as 10 mg, and the duration is unknown. DOAM produces a bare threshold and tenseness. As the 4-alkyl chain length is increased from shorter homologues such as DOM, DOET, DOPR, and DOBU which are all potent hallucinogens, the 5-HT2 binding affinity increases, rising to a maximum with the 4-(n-hexyl) derivative before falling again with even longer chains, but compounds with chain length longer than n-propyl, or with other bulky groups such as isopropyl, t-butyl or γ-phenylpropyl at the 4- position, fail to substitute for hallucinogens in animals or produce hallucinogenic effects in humans, suggesting these have low efficacy and are thus antagonists or weak partial agonists at the 5-HT2A receptor.

2,5-Dimethoxy-4-ethoxyamphetamine (MEM) is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin. In his book PiHKAL, he lists the active dose range as 20–50 mg, and the duration as 10–14 hours. According to Shulgin, MEM produces color enhancement, visual phenomena, and pattern movement, among other effects.
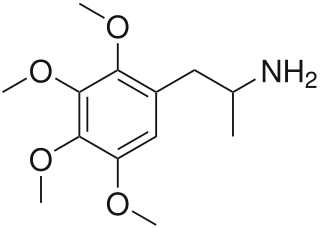
Tetramethoxyamphetamine, or 2,3,4,5-tetramethoxyamphetamine, is a lesser-known psychedelic drug and a substituted amphetamine. Tetramethoxyamphetamine was first synthesized by Alexander Shulgin. In his book PiHKAL , the minimum dosage is listed as 50 mg, and the duration unknown. Tetramethoxyamphetamine produces a threshold, mydriasis, and a headache. Limited data exists about its pharmacological properties, metabolism, and toxicity.
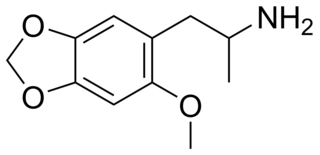
MMDA-2 (2-methoxy-4,5-methylenedioxyamphetamine) is a psychedelic drug of the amphetamine class. It is closely related to MMDA and MDA.

2,5-Dimethoxy-4-fluoroamphetamine (DOF) is a psychedelic drug of the phenethylamine and amphetamine classes. Alexander Shulgin briefly describes DOF in his book PiHKAL:
Animal studies that have compared DOF to the highly potent DOI and DOB imply that the human activity will be some four to six times less than these two heavier halide analogues.

4-Substituted-2,5-dimethoxyamphetamines (DOx) is a chemical class of substituted amphetamine derivatives featuring methoxy groups at the 2- and 5- positions of the phenyl ring, and a substituent such as alkyl or halogen at the 4- position of the phenyl ring. Most compounds of this class are potent and long-lasting psychedelic drugs, and act as highly selective 5-HT2A, 5-HT2B, and 5-HT2C receptor partial agonists. A few bulkier derivatives such as DOAM have similarly high binding affinity for 5-HT2 receptors but instead act as antagonists, and so do not produce psychedelic effects though they retain amphetamine-like stimulant effects.

5-Methoxy-7,N,N-trimethyltryptamine (5-MeO-7,N,N-TMT, 5-MeO-7-TMT), is a tryptamine derivative which acts as an agonist at the 5-HT2 serotonin receptors. In animal tests, both 7,N,N-TMT and 5-MeO-7,N,N-TMT produced behavioural responses similar to those of psychedelic drugs such as DMT and 5-MeO-DMT, but compounds with larger 7-position substituents such as 7-ethyl-DMT and 7-bromo-DMT did not produce psychedelic-appropriate responding despite high 5-HT2 receptor binding affinity, suggesting these may be antagonists or weak partial agonists for the 5-HT2 receptors. The related compound 7-MeO-MiPT (cf. 5-MeO-MiPT) was also found to be inactive, suggesting that the 7-position has poor tolerance for bulky groups at this position, at least if agonist activity is desired.

7,N,N-Trimethyltryptamine (7-methyl-DMT, 7-TMT), is a tryptamine derivative which acts as an agonist of 5-HT2 receptors. In animal tests, both 7-TMT and its 5-methoxy derivative 5-MeO-7-TMT produced behavioural responses similar to those of psychedelic drugs such as DMT, but the larger 7-ethyl and 7-bromo derivatives of DMT did not produce psychedelic responses despite having higher 5-HT2 receptor affinity in vitro (cf. DOBU, DOAM). 7-TMT also weakly inhibits reuptake of serotonin but with little effect on dopamine or noradrenaline reuptake.
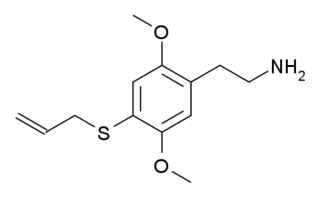
2C-T-16 is a lesser-known psychedelic drug. It was originally named by Alexander Shulgin as described in his book PiHKAL, however while Shulgin began synthesis of this compound he only got as far as the nitrostyrene intermediate, and did not complete the final synthetic step. Synthesis of 2C-T-16 was finally achieved by Daniel Trachsel some years later, and it was subsequently reported as showing similar psychedelic activity to related compounds, with a dose range of 10–25 mg and a duration of 4–6 hours, making it around the same potency as the better-known saturated analogue 2C-T-7, but with a significantly shorter duration of action. Binding studies in vitro showed 2C-T-16 to have a binding affinity of 44nM at 5-HT2A and 15nM at 5-HT2C. 2C-T-16 and related derivatives are potent partial agonists of the 5-HT1A, 5-HT2A, 5-HT2B and 5-HT2C receptors and induce a head-twitch response in mice.

2,5-Dimethoxy-4-isopropylamphetamine is a psychedelic drug of the phenethylamine and amphetamine chemical classes. It was first synthesized by Alexander Shulgin, and was described in his book PiHKAL. Shulgin described DOiPR as being at least an order of magnitude weaker than DOPr, with doses of 20–30 mg required to produce valid changes in mental state. Very little data exists about the pharmacological properties, metabolism, and toxicity of DOiPR.
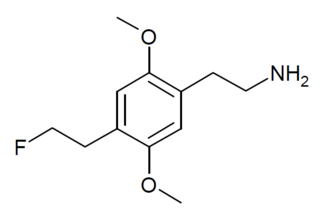
2C-EF is a lesser-known psychedelic drug. It was originally named by Alexander Shulgin as described in his book PiHKAL, but he never synthesised it himself, though it has been synthesised and its activity tested in subsequent years.




















