
Lysergic acid diethylamide, commonly known as LSD, and known colloquially as acid or lucy is a potent psychedelic drug. Effects typically include intensified thoughts, emotions, and sensory perception. At sufficiently high dosages LSD manifests primarily mental, visual, and auditory hallucinations. Dilated pupils, increased blood pressure, and increased body temperature are typical. Effects typically begin within half an hour and can last for up to 20 hours. LSD is also capable of causing mystical experiences and ego dissolution. It is used mainly as a recreational drug or for spiritual reasons. LSD is both the prototypical psychedelic and one of the "classical" psychedelics, being the psychedelic with the greatest scientific and cultural significance. LSD is synthesized as a solid compound, typically in the form of a powder or a crystalline material. This solid LSD is then dissolved in a liquid solvent, such as ethanol or distilled water, to create a solution. The liquid serves as a carrier for the LSD, allowing for accurate dosage and administration onto small pieces of blotter paper called tabs. LSD is typically either swallowed or held under the tongue. In pure form, LSD is clear or white in color, has no smell, and is crystalline. It breaks down with exposure to ultraviolet light.
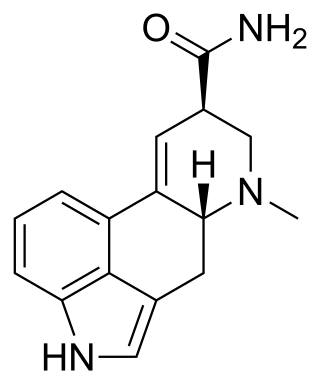
Ergine, also known as d-lysergic acid amide (LSA) and d-lysergamide, is an ergoline alkaloid that occurs in various species of vines of the Convolvulaceae and some species of fungi. The psychedelic properties in the seeds of ololiuhqui, Hawaiian baby woodrose and morning glories have been linked to ergine and/or isoergine, its epimer, as it is an alkaloid present in the seeds.
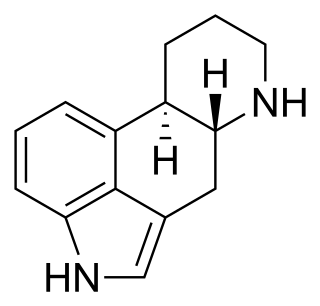
Ergoline is a chemical compound whose structural skeleton is contained in a variety of alkaloids, referred to as ergoline derivatives or ergoline alkaloids. Ergoline alkaloids, one being ergine, were initially characterized in ergot. Some of these are implicated in the condition ergotism, which can take a convulsive form or a gangrenous form. Even so, many ergoline alkaloids have been found to be clinically useful. Annual world production of ergot alkaloids has been estimated at 5,000–8,000 kg of all ergopeptines and 10,000–15,000 kg of lysergic acid, used primarily in the manufacture of semi-synthetic derivatives.

D-Lysergic acid α-hydroxyethylamide, also known as D-lysergic acid methyl carbinolamide, is a is a Lysergamide and alkaloid of the Ergoline family, it is present in higher concentrations in the parasitic fungi species "Claviceps", mainly the Claviceps Paspali, also in Claviceps Purpurea. This fungi grows in various species in the Convolvulaceae family like the Ipomoea violacea, the Rivea corymbosa (Ololiuhqui), and the Argyreia nervosa. Heavenly Blue Morning Glory and Hawaiian Baby Woodrose especially contain high amounts of LSH, with content varying between species and by how fresh the seeds are. LSH is a psychoactive Ergoline and has effects similar to LSD due to similarity in the structure and is the main psychoactive compound found in Claviceps Paspali and in (fresh) Heavenly Blue Morning Glory Seeds. LSH is unstable and breaks down into LSA quickly, so old seeds often only contains LSA and iso-LSA. When the seeds are fresh, they contain significantly higher amounts of LSH.

2C-TFM is a psychedelic phenethylamine of the 2C family. It was first synthesized in the laboratory of David E. Nichols. It has also been called 2C-CF3, a name derived from the Para-trifluoromethyl group it contains.

Methylergometrine, also known as methylergonovine and sold under the brand name Methergine, is a medication of the ergoline and lysergamide groups which is used as an oxytocic in obstetrics and in the treatment of migraine. It reportedly produces psychedelic effects similar to those of lysergic acid diethylamide (LSD) at high doses.
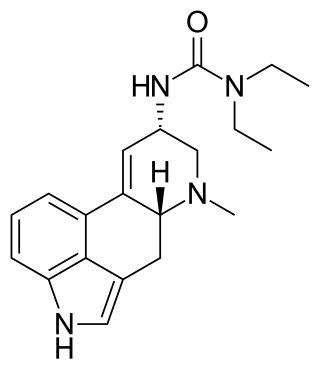
Lisuride, sold under the brand name Dopergin among others, is a monoaminergic medication of the ergoline class which is used in the treatment of Parkinson's disease, migraine, and high prolactin levels. It is taken by mouth.
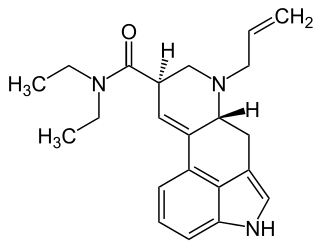
AL-LAD, also known as 6-allyl-6-nor-LSD, is a psychedelic drug and an analog of lysergic acid diethylamide (LSD). It is described by Alexander Shulgin in the book TiHKAL. It is synthesized starting from nor-LSD as a precursor, using allyl bromide as a reactant.

ETH-LAD, 6-ethyl-6-nor-lysergic acid diethylamide is an analogue of LSD. Its human psychopharmacology was first described by Alexander Shulgin in the book TiHKAL. ETH-LAD is a psychedelic drug similar to LSD, and is slightly more potent than LSD itself, with an active dose reported at between 20 and 150 micrograms. ETH-LAD has subtly different effects to LSD, described as less demanding.

PRO-LAD is an analogue of LSD. It is described by Alexander Shulgin in the book TiHKAL. PRO-LAD is a psychedelic drug similar to LSD, and is around as potent as LSD itself with an active dose reported at between 100 and 200 micrograms.

A serotonin receptor agonist is an agonist of one or more serotonin receptors. They activate serotonin receptors in a manner similar to that of serotonin, a neurotransmitter and hormone and the endogenous ligand of the serotonin receptors.

4-HO-MPMI (also known as 4-Hydroxy-N-methyl-(α,N-trimethylene)-tryptamine or lucigenol) is a tryptamine derivative that is a psychedelic drug. It was developed by the team led by David Nichols from Purdue University in the late 1990s. This compound produces hallucinogen-appropriate responding in animal tests with a similar potency to the amphetamine-derived psychedelic DOI, and has two enantiomers, with only the (R)-enantiomer being active.

Lysergic acid 2,4-dimethylazetidide (LA-SS-Az, LSZ) is an analog of LSD developed by the team led by David E. Nichols at Purdue University. It was developed as a rigid analog of LSD with the diethylamide group constrained into an azetidine ring in order to map the binding site at the 5-HT2A receptor. There are three possible stereoisomers around the azetidine ring, with the (S,S)-(+) isomer being the most active, slightly more potent than LSD itself in drug discrimination tests using trained rats.

Methylisopropyllysergamide is an analogue of LSD that was originally discovered by Albert Hofmann at Sandoz during the original structure-activity research into LSD. It has subsequently been investigated in more detail by the team led by David E. Nichols at Purdue University. Methylisopropyllysergamide is a structural isomer of LSD, with the alkyl groups on the amide nitrogen having been subjected to a methylene shuffle. MIPLA and its ethylisopropyl homologue are the only simple N,N-dialkyl lysergamides that approach the potency of LSD itself, being around 1/3-1/2 the potency of LSD, while all other dialkyl analogues tested are only around 1/10 as potent as LSD, although some N-monoalkyl lysergamides such as the sec-butyl and t-butyl derivatives were also found to show an activity profile and potency comparable to LSD, and the mono-isopropyl derivative is only slightly weaker than MIPLA. Apart from its lower potency, the hallucinogenic effects of methylisopropyllysergamide are similar to those of LSD itself, and the main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects.

Lysergic acid 2-butyl amide (2-Butyllysergamide, LSB) is an analogue of LSD originally developed by Richard Pioch at Eli Lilly in the 1950s, but mostly publicised through research conducted by the team led by David E. Nichols at Purdue University. It is a structural isomer of LSD, with the two ethyl groups on the amide nitrogen having been replaced by a single sec-butyl group, joined at the 2-position. It is one of the few lysergamide derivatives to exceed the potency of LSD in animal drug discrimination assays, with the (R) isomer having an ED50 of 33nmol/kg for producing drug-appropriate responding, vs 48nmol/kg for LSD itself. The corresponding (R)-2-pentyl analogue has higher binding affinity for the 5-HT1A and 5-HT2A receptors, but is less potent in producing drug-appropriate responding, suggesting that the butyl compound has a higher efficacy at the receptor target. The drug discrimination assay for LSD in rats involves both 5-HT1A and 5-HT2A mediated components, and while lysergic acid 2-butyl amide is more potent than LSD as a 5-HT1A agonist, it is slightly less potent as a 5-HT2A agonist, and so would probably be slightly less potent than LSD as a hallucinogen in humans. The main use for this drug has been in studies of the binding site at the 5-HT2A receptor through which LSD exerts most of its pharmacological effects, with the stereoselective activity of these unsymmetric monoalkyl lysergamides foreshadowing the subsequent development of compounds such as lysergic acid 2,4-dimethylazetidide (LSZ).
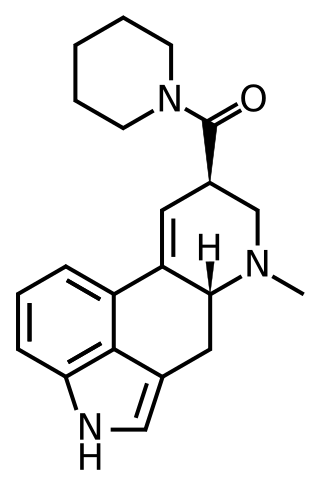
LSD-Pip is a compound from the ergoline family, related to LSD but with the N,N-diethyl substitution replaced by a piperidine group. It is more potent than the corresponding pyrrolidine and morpholine analogues, but is still several times less potent than LSD as a 5-HT2A agonist. Early studies suggested this compound to be inactive as a psychedelic in humans, though this does not seem to have been confirmed by any more recent work.

25D-NBOMe is a derivative of the phenethylamine derived hallucinogen 2C-D. It acts in a similar manner to related compounds such as 25I-NBOMe, which is a potent agonist at the 5-HT2A receptor. 25D-NBOMe has been sold as a street drug since 2010 and produces similar effects in humans to related compounds such as 25I-NBOMe and 25C-NBOMe. It was banned as a Temporary Class Drug in the UK on 10 June 2013 after concerns about its recreational use.

25E-NBOMe is a derivative of the phenethylamine 2C-E. It acts in a similar manner to related compounds such as 25I-NBOMe, which are potent agonists at the 5-HT2A receptor. 25E-NBOMe has been sold as a drug and produces similar effects in humans to related compounds such as 25I-NBOMe and 25C-NBOMe.

The 25-NB (25x-NBx) series, sometimes alternatively referred to as the NBOMe compounds, is a family of serotonergic psychedelics. They are substituted phenethylamines and were derived from the 2C family. They act as selective agonists of the serotonin 5-HT2A receptor. The 25-NB family is unique relative to other classes of psychedelics in that they are, generally speaking, extremely potent and relatively selective for the 5-HT2A receptor. Use of NBOMe series drugs has caused many deaths and hospitalisations since the drugs popularisation in the 2010s. This is primarily due to their high potency, unpredictable pharmacokinetics, and sellers passing off the compounds in the series as LSD.
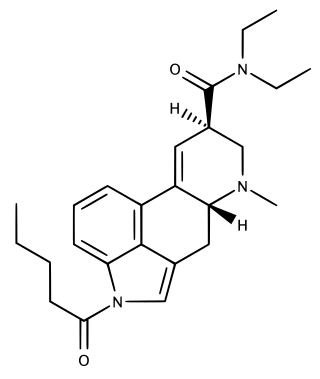
1V-LSD, sometimes nicknamed Valerie, is a psychotropic substance and a research chemical with psychedelic effects. 1V-LSD is an artificial derivative of natural lysergic acid, which occurs in ergot alkaloids, as well as being an analogue of LSD. 1V-LSD has been sold online until an amendment to the German NpSG was enforced in 2022 which controls 1P-LSD and now 1cP-LSD, 1V-LSD and several other lysergamides.




















