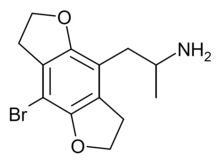
Bromo-DragonFLY is a substance related to the phenethylamine family. It acts as a potent full agonist for the 5-HT2A receptor.

2C-TFM is a psychedelic phenethylamine of the 2C family. It was first synthesized in the laboratory of David E. Nichols. It has also been called 2C-CF3, a name derived from the Para-trifluoromethyl group it contains.
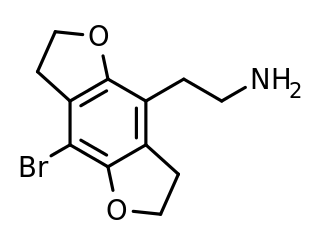
2C-B-FLY is a psychedelic phenethylamine and designer drug of the 2C family. It was first synthesized in 1996 by Aaron Monte, Professor of Chemistry at UW-La Crosse.
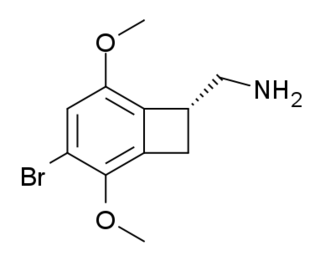
TCB-2 is a hallucinogen discovered in 2006 by Thomas McLean working in the lab of David Nichols at Purdue University. It is a conformationally-restricted derivative of the phenethylamine 2C-B, also a hallucinogen, and acts as a potent agonist for the 5-HT2A and 5-HT2C receptors with a Ki of 0.26 nM at the human 5-HT2A receptor. In drug-substitution experiments in rats, TCB-2 was found to be of similar potency to both LSD and Bromo-DragonFLY, ranking it among the most potent phenethylamine hallucinogens yet discovered. This high potency and selectivity has made TCB-2 useful for distinguishing 5-HT2A mediated responses from those produced by other similar receptors. TCB-2 has similar but not identical effects in animals to related phenethylamine hallucinogens such as DOI, and has been used for studying how the function of the 5-HT2A receptor differs from that of other serotonin receptors in a number of animal models, such as studies of cocaine addiction and neuropathic pain.

2CBCB-NBOMe (NBOMe-TCB-2) is a compound indirectly derived from the phenethylamine series of hallucinogens, which was discovered in 2007 at Purdue University as part of the ongoing research program of the team led by David Nichols focusing on the mapping of the specific amino acid residues responsible for ligand binding to the 5HT2A receptor. 2CBCB-NBOMe acts as a potent and selective agonist for the 5-HT2A and 5-HT2C receptors, with a Ki of 0.27 nM at the human 5-HT2A receptor, a similar potency to other agonists such as TCB-2, NBOMe-2C-I and Bromo-DragonFLY.

Jimscaline (C-(4,5,6-trimethoxyindan-1-yl)methanamine) is a conformationally-restricted derivative of the cactus-derived hallucinogen mescaline, which was discovered in 2006 by a team at Purdue University led by David E. Nichols. It acts as a potent agonist for the 5-HT2A and 5-HT2C receptors with the more active (R)-enantiomer having a Ki of 69 nM at the human 5-HT2A receptor, and around three times the potency of mescaline in drug-substitution experiments in animals. This discovery that the side chain of the phenethylamine hallucinogens could be constrained to give chiral ligands with increased activity then led to the later development of the super-potent benzocyclobutene derivative TCB-2.
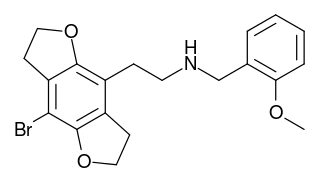
2CBFly-NBOMe is a compound indirectly derived from the phenethylamine hallucinogen 2C-B, and related to benzodifurans like 2C-B-FLY and N-benzylphenethylamines like 25I-NBOMe. It was discovered in 2002, and further researched by Ralf Heim at the Free University of Berlin, and subsequently investigated in more detail by a team at Purdue University led by David E. Nichols. It acts as a potent partial agonist for the 5-HT2A serotonin receptor subtype.

TFMFly is a compound related to psychedelic phenethylamines such as 2C-B-FLY and 2C-TFM. It was first reported in 2005 by a team at Purdue University led by David Nichols. It acts as a potent agonist at the 5HT2A serotonin receptor subtype, and is a chiral compound with the more active (R) enantiomer having a Ki of 0.12 nM at the human 5-HT2A receptor. While the fully aromatic benzodifurans such as Bromo-DragonFLY generally have higher binding affinity than saturated compounds like 2C-B-FLY, the saturated compounds have higher efficacy as agonists.

2,5-Dimethoxy-4-fluoroamphetamine (DOF) is a psychedelic drug of the phenethylamine and amphetamine classes. Alexander Shulgin briefly describes DOF in his book PiHKAL:
Animal studies that have compared DOF to the highly potent DOI and DOB imply that the human activity will be some four to six times less than these two heavier halide analogues.
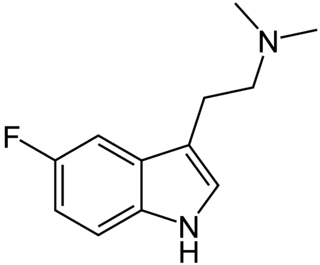
5-Fluoro-N,N-dimethyltryptamine is a tryptamine derivative related to compounds such as 5-bromo-DMT and 5-MeO-DMT. Fluorination of psychedelic tryptamines either reduces or has little effect on 5-HT2A/C receptor affinity or intrinsic activity, although 6-fluoro-DET is inactive as a psychedelic despite acting as a 5-HT2A agonist, while 4-fluoro-5-methoxy-DMT is a much stronger agonist at 5-HT1A than 5-HT2A.

2CB-Ind is a conformationally-restricted derivative of the phenethylamine hallucinogen 2C-B, discovered in 1974 by Alexander Shulgin. It acts as a moderately potent and selective agonist for the 5-HT2A and 5-HT2C receptors, but unlike the corresponding benzocyclobutene derivative TCB-2 which is considerably more potent than the parent compound 2C-B, 2CB-Ind is several times weaker, with racemic 2CB-Ind having a Ki of 47nM at the human 5-HT2A receptor, only slightly more potent than the mescaline analogue (R)-jimscaline.
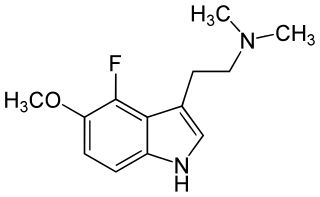
4-Fluoro-5-Methoxy-N,N-dimethyltryptamine (4-F-5-MeO-DMT) was first described by David E. Nichols team in 2000. It is a potent 5-HT1A agonist. Substitution with the 4-fluorine markedly increased 5-HT1A selectivity over 5-HT2A/2C receptors with potency greater than that of the 5-HT1A agonist 8-OH-DPAT.

DMBMPP, or 2-(2,5-dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine, is a 2-benzylpiperidine analog of the hallucinogenic N-benzylphenethylamine 25B-NBOMe and was discovered in 2011 by Jose Juncosa in the group of David E. Nichols at Purdue University. DMBMPP differs from 25B-NBOMe by incorporating the amine within a piperidine ring, making for a more rigid molecular structure than that of the open-chain 25B-NBOMe. The presence of the piperidine ring introduces two stereocenters, thus, four stereoisomers of this compound can be made.
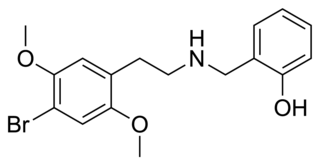
25B-NBOH is a derivative of the phenethylamine derived hallucinogen 2C-B which has been sold as a designer drug. It acts as a potent serotonin receptor agonist with similar affinity to the better-known compound 25B-NBOMe at 5-HT2A and 5-HT2C receptors with pKis values of 8.3 and 9.4, respectively.
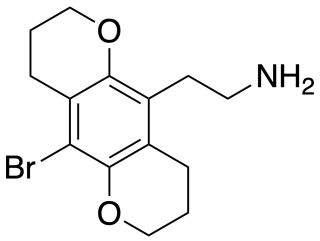
2C-B-BUTTERFLY is a conformationally-restricted derivative of the phenethylamine hallucinogen 2C-B, which was discovered in 1999 by Michael S. Whiteside and Aaron Monte. It is a ring-expanded homologue of the better known compound 2C-B-FLY, and has similar properties as an agonist for serotonin receptors, but with more selectivity for 5-HT2C over 5-HT2A.

The substituted benzofurans are a class of chemical compounds based on the heterocyclyc and polycyclic compound benzofuran. Many medicines use the benzofuran core as a scaffold, but most commonly the term is used to refer to the simpler compounds in this class which include numerous psychoactive drugs, including stimulants, psychedelics and empathogens. In general, these compounds have a benzofuran core to which a 2-aminoethyl group is attached, and combined with a range of other substituents. Some psychoactive derivatives from this family have been sold under the name Benzofury.
25B-NBF is a derivative of the phenethylamine hallucinogen 2C-B, which acts as a highly potent partial agonist for the human 5-HT2A receptor.

2C-B-DRAGONFLY (2C-B-DFLY) is a recreational designer drug with psychedelic effects. It can be regarded as the fully aromatic derivative of 2C-B-FLY. 2C-B-DRAGONFLY is stronger than 2C-B or 2C-B-FLY with around 2-3x the potency of 2C-B in animal studies, demonstrating the importance of the fully aromatic benzodifuran ring system for optimum receptor binding at 5-HT2A, but it is still considerably less potent than its alpha-methyl derivative Bromo-DragonFLY.

DOB-2-DRAGONFLY-5-BUTTERFLY is a drug with an unusual furo[2,3-g]chromene core structure which acts as a 5-HT2A receptor agonist. It was first synthesised by David E. Nichols and colleagues in 2008, and while it is weaker than similar compounds such as Bromo-DragonFLY it is still the most potent among a number of related derivatives.