| The complete data for Arginine () | ||||||||||||||||
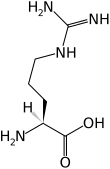  | General information Chemical formula : C 6 H 14 N 4 O 2 Molar mass : 174.2 g·mol−1 Systematic name : 2-amino-5-(diaminomethylidene amino)pentanoic acid Abbreviations : R, Arg Synonyms : 2-amino-5-guanidinopentanoic acid 2-amino-5-guanidinovaleric acid AIDS{-}121865 AIDS{-}159840 Argamine Arginin Argivene CHEBI:29016 CHEMBANK2983 Detoxargin Harg Levargin Minophagen A R-Gene | |||||||||||||||
| Database data | ||||||||||||||||
| SMILES : NC(=N)NCCCC(N)C(=O)O InChI =1/C6H14N4O2/c7-4(5(11)12)2-1-3-10-6(8)9/h4H,1-3,7H2,(H,11,12)(H4,8,9,10)/f/h11H,8-9H2
| ||||||||||||||||
| Physical properties | ||||||||||||||||
| ||||||||||||||||
| Hazard properties | ||||||||||||||||
| ||||||||||||||||
| Chemical properties | ||||||||||||||||
| ||||||||||||||||
| Pharmacological properties | ||||||||||||||||
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.