| |||
| Names | |||
|---|---|---|---|
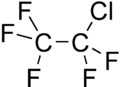
| Preferred IUPAC name 1-Chloro-1,1,2,2,2-pentafluoroethane | |||
| Other names Freon 115, CFC-115, R-115, Fluorocarbon-115, Genetron 115, Halocarbon 115, Monochloropentafluoroethane | |||
| Identifiers | |||
3D model (JSmol) | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.854 | ||
| EC Number |
| ||
| E number | E945 (glazing agents, ...) | ||
PubChem CID | |||
| RTECS number |
| ||
| UNII | |||
| UN number | 1020 | ||
CompTox Dashboard (EPA) | |||
| |||
| |||
| Properties | |||
| C2ClF5 | |||
| Molar mass | 154.466 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Ethereal | ||
| Melting point | −99 °C (−146 °F; 174 K) | ||
| Boiling point | −39.1 °C (−38.4 °F; 234.1 K) | ||
| 59 mg/L | |||
| Vapor pressure | 7.9 atm (21°C) [1] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards | In high concentrations may cause asphyxiation. [2] | ||
| GHS labelling: | |||
  | |||
| Warning | |||
| H420 | |||
| P410+P403, P502 | |||
| Flash point | 70.4 °C (158.7 °F; 343.5 K) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) | none [1] | ||
REL (Recommended) | TWA 1000 ppm (6320 mg/m3) [1] | ||
IDLH (Immediate danger) | N.D. [1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Chloropentafluoroethane is a chlorofluorocarbon (CFC) once used as a refrigerant and also known as R-115 and CFC-115. Its production and consumption has been banned since 1 January 1996 under the Montreal Protocol because of its high ozone depletion potential and very long lifetime when released into the environment. [3] CFC-115 is also a potent greenhouse gas.