A prosthetic group is the non-amino acid component that is part of the structure of the heteroproteins or conjugated proteins, being tightly linked to the apoprotein.
Cysteine metabolism refers to the biological pathways that consume or create cysteine. The pathways of different amino acids and other metabolites interweave and overlap to creating complex systems.

Thiosulfate dehydrogenase is an enzyme that catalyzes the chemical reaction:

Cystathionine beta-lyase, also commonly referred to as CBL or β-cystathionase, is an enzyme that primarily catalyzes the following α,β-elimination reaction

In enzymology, a 3-mercaptopyruvate sulfurtransferase is an enzyme that catalyzes the chemical reactions of 3-mercaptopyruvate. This enzyme belongs to the family of transferases, specifically the sulfurtransferases. This enzyme participates in cysteine metabolism. It is encoded by the MPST gene.

Lipoyl synthase is an enzyme that belongs to the radical SAM (S-adenosyl methionine) family. Within the radical SAM superfamily, lipoyl synthase is in a sub-family of enzymes that catalyze sulfur insertion reactions. The enzymes in this subfamily differ from general radical SAM enzymes, as they contain two 4Fe-4S clusters. From these clusters, the enzymes obtain the sulfur groups that will be transferred onto the corresponding substrates. This particular enzyme participates in the final step of lipoic acid metabolism, transferring two sulfur atoms from its 4Fe-4S cluster onto the protein N6-(octanoyl)lysine through radical generation. This enzyme is usually localized to the mitochondria. Two organisms that have been extensively studied with regards to this enzyme are Escherichia coli and Mycobacterium tuberculosis. It is currently being studied in other organisms including yeast, plants, and humans.

Molybdenum cofactor biosynthesis protein 1 is a protein that in humans and other animals, fungi, and cellular slime molds, is encoded by the MOCS1 gene.

MOSC domain-containing protein 2, mitochondrial is a protein that in humans is encoded by the MOSC2 gene.
Molybdopterin synthase (EC 2.8.1.12, MPT synthase) is an enzyme required to synthesize molybdopterin (MPT) from precursor Z (now known as cyclic pyranopterin monophosphate). Molydopterin is subsequently complexed with molybdenum to form molybdenum cofactor (MoCo). MPT synthase catalyses the following chemical reaction:
Radical SAM is a designation for a superfamily of enzymes that use a [4Fe-4S]+ cluster to reductively cleave S-adenosyl-L-methionine (SAM) to generate a radical, usually a 5′-deoxyadenosyl radical (5'-dAdo), as a critical intermediate. These enzymes utilize this radical intermediate to perform diverse transformations, often to functionalize unactivated C-H bonds. Radical SAM enzymes are involved in cofactor biosynthesis, enzyme activation, peptide modification, post-transcriptional and post-translational modifications, metalloprotein cluster formation, tRNA modification, lipid metabolism, biosynthesis of antibiotics and natural products etc. The vast majority of known radical SAM enzymes belong to the radical SAM superfamily, and have a cysteine-rich motif that matches or resembles CxxxCxxC. rSAMs comprise the largest superfamily of metal-containing enzymes.
Molybdopterin adenylyltransferase is an enzyme with systematic name ATP:molybdopterin adenylyltransferase. This enzyme catalyses the following chemical reaction
Molybdenum cofactor cytidylyltransferase is an enzyme with systematic name CTP:molybdenum cofactor cytidylyltransferase. This enzyme catalyses the following chemical reaction:
Molybdenum cofactor guanylyltransferase is an enzyme with systematic name GTP:molybdenum cofactor guanylyltransferase. This enzyme catalyses the following chemical reaction:
Molybdopterin-synthase adenylyltransferase is an enzyme with systematic name ATP:molybdopterin-synthase adenylyltransferase. This enzyme catalyses the following chemical reaction
Molybdopterin synthase sulfurtransferase is an enzyme with systematic name persulfurated L-cysteine desulfurase:(molybdopterin-synthase sulfur-carrier protein)-Gly-Gly sulfurtransferase. This enzyme catalyses the following chemical reaction
Molybdopterin molybdotransferase is an enzyme with systematic name adenylyl-molybdopterin:molybdate molybdate transferase (AMP-forming). This enzyme catalyses the following chemical reaction
Cyclic pyranopterin monophosphate synthase is an enzyme with systematic name GTP 8,9-lyase . This enzyme catalyses the following chemical reaction

Mitochondrial amidoxime-reducing component 1 is a mammalian molybdenum-containing enzyme. It is located in the outer mitochondrial membrane and consists of a N-terminal mitochondrial signal domain facing the inter-membrane space, a transmembrane domain, and a C-terminal catalytic domain facing the cytosol. In humans it is encoded by the MOSC1 gene.

Fosdenopterin, sold under the brand name Nulibry, is a medication used to reduce the risk of death due to a rare genetic disease known as molybdenum cofactor deficiency type A.
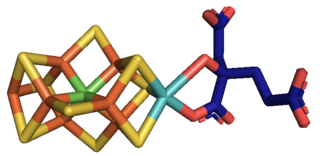
Molybdenum is an essential element in most organisms. It is most notably present in nitrogenase which is an essential part of nitrogen fixation.








