
The pituitary gland or hypophysis is an endocrine gland in vertebrates. In humans, the pituitary gland is located at the base of the brain, protruding off the bottom of the hypothalamus. The human pituitary gland is oval shaped, about 1 cm in diameter, 0.5–1 gram (0.018–0.035 oz) in weight on average, and about the size of a kidney bean.

The hypothalamus is a small part of the vertebrate brain that contains a number of nuclei with a variety of functions. One of the most important functions is to link the nervous system to the endocrine system via the pituitary gland. The hypothalamus is located below the thalamus and is part of the limbic system. It forms the basal part of the diencephalon. All vertebrate brains contain a hypothalamus. In humans, it is about the size of an almond.

Corticotropin-releasing hormone (CRH) is a peptide hormone involved in stress responses. It is a releasing hormone that belongs to corticotropin-releasing factor family. In humans, it is encoded by the CRH gene. Its main function is the stimulation of the pituitary synthesis of adrenocorticotropic hormone (ACTH), as part of the hypothalamic–pituitary–adrenal axis.
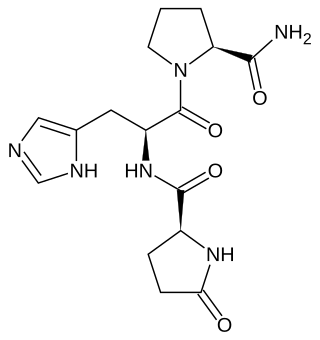
Thyrotropin-releasing hormone (TRH) is a hypophysiotropic hormone produced by neurons in the hypothalamus that stimulates the release of thyroid-stimulating hormone (TSH) and prolactin from the anterior pituitary.

Human vasopressin, also called antidiuretic hormone (ADH), arginine vasopressin (AVP) or argipressin, is a hormone synthesized from the AVP gene as a peptide prohormone in neurons in the hypothalamus, and is converted to AVP. It then travels down the axon terminating in the posterior pituitary, and is released from vesicles into the circulation in response to extracellular fluid hypertonicity (hyperosmolality). AVP has two primary functions. First, it increases the amount of solute-free water reabsorbed back into the circulation from the filtrate in the kidney tubules of the nephrons. Second, AVP constricts arterioles, which increases peripheral vascular resistance and raises arterial blood pressure.

The posterior pituitary is the posterior lobe of the pituitary gland which is part of the endocrine system. The posterior pituitary is not glandular as is the anterior pituitary. Instead, it is largely a collection of axonal projections from the hypothalamus that terminate behind the anterior pituitary, and serve as a site for the secretion of neurohypophysial hormones directly into the blood. The hypothalamic–neurohypophyseal system is composed of the hypothalamus, posterior pituitary, and these axonal projections.

The supraoptic nucleus (SON) is a nucleus of magnocellular neurosecretory cells in the hypothalamus of the mammalian brain. The nucleus is situated at the base of the brain, adjacent to the optic chiasm. In humans, the SON contains about 3,000 neurons.
Corticotropic cells, are basophilic cells in the anterior pituitary that produce pro-opiomelanocortin (POMC) which undergoes cleavage to adrenocorticotropin (ACTH), β-lipotropin (β-LPH), and melanocyte-stimulating hormone (MSH). These cells are stimulated by corticotropin releasing hormone (CRH) and make up 15–20% of the cells in the anterior pituitary. The release of ACTH from the corticotropic cells is controlled by CRH, which is formed in the cell bodies of parvocellular neurosecretory cells within the paraventricular nucleus of the hypothalamus and passes to the corticotropes in the anterior pituitary via the hypophyseal portal system. Adrenocorticotropin hormone stimulates the adrenal cortex to release glucocorticoids and plays an important role in the stress response.
Magnocellular neurosecretory cells are large neuroendocrine cells within the supraoptic nucleus and paraventricular nucleus of the hypothalamus. They are also found in smaller numbers in accessory cell groups between these two nuclei, the largest one being the circular nucleus. There are two types of magnocellular neurosecretory cells, oxytocin-producing cells and vasopressin-producing cells, but a small number can produce both hormones. These cells are neuroendocrine neurons, are electrically excitable, and generate action potentials in response to afferent stimulation. Vasopressin is produced from the vasopressin-producing cells via the AVP gene, a molecular output of circadian pathways.
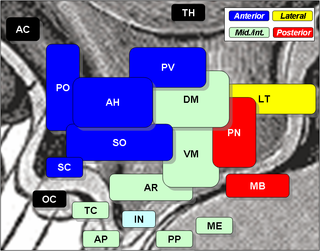
The arcuate nucleus of the hypothalamus (ARH), or ARC, is also known as the infundibular nucleus to distinguish it from the arcuate nucleus of the medulla oblongata in the brainstem. The arcuate nucleus is an aggregation of neurons in the mediobasal hypothalamus, adjacent to the third ventricle and the median eminence. The arcuate nucleus includes several important and diverse populations of neurons that help mediate different neuroendocrine and physiological functions, including neuroendocrine neurons, centrally projecting neurons, and astrocytes. The populations of neurons found in the arcuate nucleus are based on the hormones they secrete or interact with and are responsible for hypothalamic function, such as regulating hormones released from the pituitary gland or secreting their own hormones. Neurons in this region are also responsible for integrating information and providing inputs to other nuclei in the hypothalamus or inputs to areas outside this region of the brain. These neurons, generated from the ventral part of the periventricular epithelium during embryonic development, locate dorsally in the hypothalamus, becoming part of the ventromedial hypothalamic region. The function of the arcuate nucleus relies on its diversity of neurons, but its central role is involved in homeostasis. The arcuate nucleus provides many physiological roles involved in feeding, metabolism, fertility, and cardiovascular regulation.

The median eminence is generally defined as the portion of the ventral hypothalamus from which the portal vessels arise. The median eminence is a small swelling on the tuber cinereum, posterior to and on top of the pituitary stalk; it lies in the area roughly bounded on its posterolateral region by the cerebral peduncles, and on its anterolateral region by the optic chiasm.
A neurohormone is any hormone produced and released by neuroendocrine cells into the blood. By definition of being hormones, they are secreted into the circulation for systemic effect, but they can also have a role of neurotransmitter or other roles such as autocrine (self) or paracrine (local) messenger.
Neuroendocrine cells are cells that receive neuronal input and, as a consequence of this input, release messenger molecules (hormones) into the blood. In this way they bring about an integration between the nervous system and the endocrine system, a process known as neuroendocrine integration. An example of a neuroendocrine cell is a cell of the adrenal medulla, which releases adrenaline to the blood. The adrenal medullary cells are controlled by the sympathetic division of the autonomic nervous system. These cells are modified postganglionic neurons. Autonomic nerve fibers lead directly to them from the central nervous system. The adrenal medullary hormones are kept in vesicles much in the same way neurotransmitters are kept in neuronal vesicles. Hormonal effects can last up to ten times longer than those of neurotransmitters. Sympathetic nerve fiber impulses stimulate the release of adrenal medullary hormones. In this way the sympathetic division of the autonomic nervous system and the medullary secretions function together.
Releasing hormones and inhibiting hormones are hormones whose main purpose is to control the release of other hormones, either by stimulating or inhibiting their release. They are also called liberins and statins (respectively), or releasing factors and inhibiting factors. The principal examples are hypothalamic-pituitary hormones that can be classified from several viewpoints: they are hypothalamic hormones, they are hypophysiotropic hormones, and they are tropic hormones.
Neuroendocrinology is the branch of biology which studies the interaction between the nervous system and the endocrine system; i.e. how the brain regulates the hormonal activity in the body. The nervous and endocrine systems often act together in a process called neuroendocrine integration, to regulate the physiological processes of the human body. Neuroendocrinology arose from the recognition that the brain, especially the hypothalamus, controls secretion of pituitary gland hormones, and has subsequently expanded to investigate numerous interconnections of the endocrine and nervous systems.

Neurophysin II is a carrier protein with a size of 19,687.3 Da and is made up of a dimer of two virtually identical chains of amino acids. Neurophysin II is a cleavage product of the AVP gene. It is a neurohypophysial hormone that is transported in vesicles with vasopressin, the other cleavage product, along axons, from magnocellular neurons of the hypothalamus to the posterior lobe of the pituitary. Although it is stored in neurosecretory granules with vasopressin and released with vasopressin into the bloodstream, its biological action is unclear. Neurophysin II is also known as a stimulator of prolactin secretion.
Hypothalamic–pituitary hormones are hormones that are produced by the hypothalamus and pituitary gland. Although the organs in which they are produced are relatively small, the effects of these hormones cascade throughout the body. They can be classified as a hypothalamic–pituitary axis of which the adrenal, gonadal, thyroid, somatotropic, and prolactin axes are branches.
Hypothalamic disease is a disorder presenting primarily in the hypothalamus, which may be caused by damage resulting from malnutrition, including anorexia and bulimia eating disorders, genetic disorders, radiation, surgery, head trauma, lesion, tumour or other physical injury to the hypothalamus. The hypothalamus is the control center for several endocrine functions. Endocrine systems controlled by the hypothalamus are regulated by antidiuretic hormone (ADH), corticotropin-releasing hormone, gonadotropin-releasing hormone, growth hormone-releasing hormone, oxytocin, all of which are secreted by the hypothalamus. Damage to the hypothalamus may impact any of these hormones and the related endocrine systems. Many of these hypothalamic hormones act on the pituitary gland. Hypothalamic disease therefore affects the functioning of the pituitary and the target organs controlled by the pituitary, including the adrenal glands, ovaries and testes, and the thyroid gland.
Parvocellular neurosecretory cells are small neurons that produce hypothalamic releasing and inhibiting hormones. The cell bodies of these neurons are located in various nuclei of the hypothalamus or in closely related areas of the basal brain, mainly in the medial zone of the hypothalamus. All or most of the axons of the parvocellular neurosecretory cells project to the median eminence, at the base of the brain, where their nerve terminals release the hypothalamic hormones. These hormones are then immediately absorbed into the blood vessels of the hypothalamo-pituitary portal system, which carry them to the anterior pituitary gland, where they regulate the secretion of hormones into the systemic circulation.
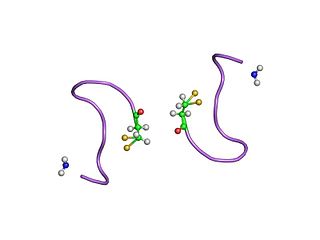
The neurohypophysial hormones form a family of structurally and functionally related peptide hormones. Their representatives in humans are oxytocin and vasopressin. They are named after the location of their release into the blood, the neurohypophysis.











