Endocytosis is a cellular process in which substances are brought into the cell. The material to be internalized is surrounded by an area of cell membrane, which then buds off inside the cell to form a vesicle containing the ingested materials. Endocytosis includes pinocytosis and phagocytosis. It is a form of active transport.

A lysosome is a membrane-bound organelle found in many animal cells. They are spherical vesicles that contain hydrolytic enzymes that digest many kinds of biomolecules. A lysosome has a specific composition, of both its membrane proteins and its lumenal proteins. The lumen's pH (~4.5–5.0) is optimal for the enzymes involved in hydrolysis, analogous to the activity of the stomach. Besides degradation of polymers, the lysosome is involved in cell processes of secretion, plasma membrane repair, apoptosis, cell signaling, and energy metabolism.

Endosomes are a collection of intracellular sorting organelles in eukaryotic cells. They are parts of endocytic membrane transport pathway originating from the trans Golgi network. Molecules or ligands internalized from the plasma membrane can follow this pathway all the way to lysosomes for degradation or can be recycled back to the cell membrane in the endocytic cycle. Molecules are also transported to endosomes from the trans Golgi network and either continue to lysosomes or recycle back to the Golgi apparatus.

Autophagy is the natural, conserved degradation of the cell that removes unnecessary or dysfunctional components through a lysosome-dependent regulated mechanism. It allows the orderly degradation and recycling of cellular components. Although initially characterized as a primordial degradation pathway induced to protect against starvation, it has become increasingly clear that autophagy also plays a major role in the homeostasis of non-starved cells. Defects in autophagy have been linked to various human diseases, including neurodegeneration and cancer, and interest in modulating autophagy as a potential treatment for these diseases has grown rapidly.

CD36, also known as platelet glycoprotein 4, fatty acid translocase (FAT), scavenger receptor class B member 3 (SCARB3), and glycoproteins 88 (GP88), IIIb (GPIIIB), or IV (GPIV) is a protein that in humans is encoded by the CD36 gene. The CD36 antigen is an integral membrane protein found on the surface of many cell types in vertebrate animals. It imports fatty acids inside cells and is a member of the class B scavenger receptor family of cell surface proteins. CD36 binds many ligands including collagen, thrombospondin, erythrocytes parasitized with Plasmodium falciparum, oxidized low density lipoprotein, native lipoproteins, oxidized phospholipids, and long-chain fatty acids.

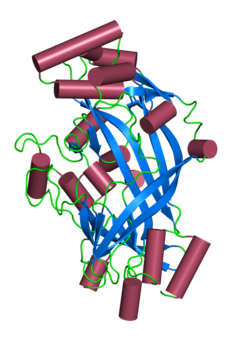
β-Glucocerebrosidase is an enzyme with glucosylceramidase activity that cleaves by hydrolysis the β-glycosidic linkage of the chemical glucocerebroside, an intermediate in glycolipid metabolism that is abundant in cell membranes. It is localized in the lysosome, where it remains associated with the lysosomal membrane. β-Glucocerebrosidase is 497 amino acids in length and has a molecular mass of 59,700 Da.

The bafilomycins are a family of macrolide antibiotics produced from a variety of Streptomycetes. Their chemical structure is defined by a 16-membered lactone ring scaffold. Bafilomycins exhibit a wide range of biological activity, including anti-tumor, anti-parasitic, immunosuppressant and anti-fungal activity. The most used bafilomycin is bafilomycin A1, a potent inhibitor of cellular autophagy. Bafilomycins have also been found to act as ionophores, transporting potassium K+ across biological membranes and leading to mitochondrial damage and cell death.

The luteinizing hormone/choriogonadotropin receptor (LHCGR), also lutropin/choriogonadotropin receptor (LCGR) or luteinizing hormone receptor (LHR), is a transmembrane receptor found predominantly in the ovary and testis, but also many extragonadal organs such as the uterus and breasts. The receptor interacts with both luteinizing hormone (LH) and chorionic gonadotropins and represents a G protein-coupled receptor (GPCR). Its activation is necessary for the hormonal functioning during reproduction.

Galectins are a class of proteins that bind specifically to β-galactoside sugars, such as N-acetyllactosamine, which can be bound to proteins by either N-linked or O-linked glycosylation. They are also termed S-type lectins due to their dependency on disulphide bonds for stability and carbohydrate binding. There have been about 15 galectins discovered in mammals, encoded by the LGALS genes, which are numbered in a consecutive manner. Only galectin-1, -2, -3, -4, -7, -7B, -8, -9, -9B, 9C, -10, -12, -13, -14, and -16 have been identified in humans. Galectin-5 and -6 are found in rodents, whereas galectin-11 and -15 are uniquely found in sheep and goats. Members of the galectin family have also been discovered in other mammals, birds, amphibians, fish, nematodes, sponges, and some fungi. Unlike the majority of lectins they are not membrane bound, but soluble proteins with both intra- and extracellular functions. They have distinct but overlapping distributions but found primarily in the cytosol, nucleus, extracellular matrix or in circulation. Although many galectins must be secreted, they do not have a typical signal peptide required for classical secretion. The mechanism and reason for this non-classical secretion pathway is unknown.
The mannose receptor is a C-type lectin primarily present on the surface of macrophages, immature dendritic cells and liver sinusoidal endothelial cells, but is also expressed on the surface of skin cells such as human dermal fibroblasts and keratinocytes. It is the first member of a family of endocytic receptors that includes Endo180 (CD280), M-type PLA2R, and DEC-205 (CD205).

CD68 is a protein highly expressed by cells in the monocyte lineage, by circulating macrophages, and by tissue macrophages.
The mannose 6-phosphate receptors (MPRs) are transmembrane glycoproteins that target enzymes to lysosomes in vertebrates.
The epididymal secretory protein E1, also known as NPC2, is one of two main lysosomal transport proteins that assist in the regulation of cellular cholesterol by exportation of LDL-derived cholesterol from lysosomes. Lysosomes have digestive enzymes that allow it to break down LDL particles to LDL-derived cholesterol once the LDL particle is engulfed into the cell via receptor mediated endocytosis.

Cadherin-2 also known as Neural cadherin (N-cadherin), is a protein that in humans is encoded by the CDH2 gene. CDH2 has also been designated as CD325 . Cadherin-2 is a transmembrane protein expressed in multiple tissues and functions to mediate cell–cell adhesion. In cardiac muscle, Cadherin-2 is an integral component in adherens junctions residing at intercalated discs, which function to mechanically and electrically couple adjacent cardiomyocytes. Alterations in expression and integrity of Cadherin-2 has been observed in various forms of disease, including human dilated cardiomyopathy. Variants in CDH2 have also been identified to cause a syndromic neurodevelopmental disorder.
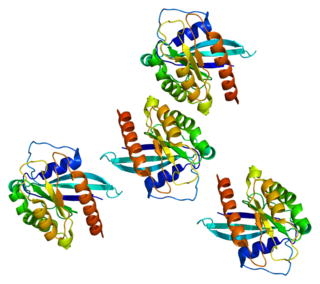
Ras-related protein Rab-7a is a protein that in humans is encoded by the RAB7A gene.

Lysosome-associated membrane protein 2 (LAMP2), also known as CD107b and Mac-3, is a human gene. Its protein, LAMP2, is one of the lysosome-associated membrane glycoproteins.

Lysosomal-associated membrane protein 1 (LAMP-1) also known as lysosome-associated membrane glycoprotein 1 and CD107a, is a protein that in humans is encoded by the LAMP1 gene. The human LAMP1 gene is located on the long arm (q) of chromosome 13 at region 3, band 4 (13q34).

CTNS may also refer to the Center for Theology and the Natural Sciences.

Lysosome-associated membrane glycoprotein 3 is a protein that in humans is encoded by the LAMP3 gene. It is one of the lysosome-associated membrane glycoproteins.

Chaperone-mediated autophagy (CMA) refers to the chaperone-dependent selection of soluble cytosolic proteins that are then targeted to lysosomes and directly translocated across the lysosome membrane for degradation. The unique features of this type of autophagy are the selectivity on the proteins that are degraded by this pathway and the direct shuttling of these proteins across the lysosomal membrane without the requirement for the formation of additional vesicles.