Related Research Articles

A vaccine is a biological preparation that provides active acquired immunity to a particular infectious or malignant disease. The safety and effectiveness of vaccines has been widely studied and verified. A vaccine typically contains an agent that resembles a disease-causing microorganism and is often made from weakened or killed forms of the microbe, its toxins, or one of its surface proteins. The agent stimulates the body's immune system to recognize the agent as a threat, destroy it, and recognize further and destroy any of the microorganisms associated with that agent that it may encounter in the future.

Polio vaccines are vaccines used to prevent poliomyelitis (polio). Two types are used: an inactivated poliovirus given by injection (IPV) and a weakened poliovirus given by mouth (OPV). The World Health Organization (WHO) recommends all children be fully vaccinated against polio. The two vaccines have eliminated polio from most of the world, and reduced the number of cases reported each year from an estimated 350,000 in 1988 to 33 in 2018.
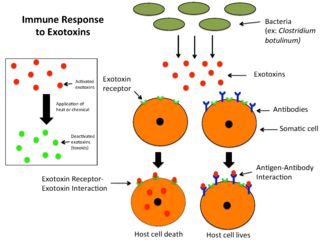
An exotoxin is a toxin secreted by bacteria. An exotoxin can cause damage to the host by destroying cells or disrupting normal cellular metabolism. They are highly potent and can cause major damage to the host. Exotoxins may be secreted, or, similar to endotoxins, may be released during lysis of the cell. Gram negative pathogens may secrete outer membrane vesicles containing lipopolysaccharide endotoxin and some virulence proteins in the bounding membrane along with some other toxins as intra-vesicular contents, thus adding a previously unforeseen dimension to the well-known eukaryote process of membrane vesicle trafficking, which is quite active at the host–pathogen interface.
ATC code J07Vaccines is a therapeutic subgroup of the Anatomical Therapeutic Chemical Classification System, a system of alphanumeric codes developed by the World Health Organization (WHO) for the classification of drugs and other medical products. Subgroup J07 is part of the anatomical group J Antiinfectives for systemic use.

The DPT vaccine or DTP vaccine is a class of combination vaccines against three infectious diseases in humans: diphtheria, pertussis, and tetanus. The vaccine components include diphtheria and tetanus toxoids and either killed whole cells of the bacterium that causes pertussis or pertussis antigens. The term toxoid refers to vaccines which use an inactivated toxin produced by the pathogen which they are targeted against to generate an immune response. In this way, the toxoid vaccine generates an immune response which is targeted against the toxin which is produced by the pathogen and causes disease, rather than a vaccine which is targeted against the pathogen itself. The whole cells or antigens will be depicted as either "DTwP" or "DTaP", where the lower-case "w" indicates whole-cell inactivated pertussis and the lower-case "a" stands for "acellular". In comparison to alternative vaccine types, such as live attenuated vaccines, the DTP vaccine does not contain any live pathogen, but rather uses inactivated toxoid to generate an immune response; therefore, there is not a risk of use in populations that are immune compromised since there is not any known risk of causing the disease itself. As a result, the DTP vaccine is considered a safe vaccine to use in anyone and it generates a much more targeted immune response specific for the pathogen of interest.

The schedule for childhood immunizations in the United States is published by the Centers for Disease Control and Prevention (CDC). The vaccination schedule is broken down by age: birth to six years of age, seven to eighteen, and adults nineteen and older. Childhood immunizations are key in preventing diseases with epidemic potential.
A booster dose is an extra administration of a vaccine after an earlier (primer) dose. After initial immunization, a booster provides a re-exposure to the immunizing antigen. It is intended to increase immunity against that antigen back to protective levels after memory against that antigen has declined through time. For example, tetanus shot boosters are often recommended every 10 years, by which point memory cells specific against tetanus lose their function or undergo apoptosis.

Hepatitis B vaccine is a vaccine that prevents hepatitis B. The first dose is recommended within 24 hours of birth with either two or three more doses given after that. This includes those with poor immune function such as from HIV/AIDS and those born premature. It is also recommended that health-care workers be vaccinated. In healthy people, routine immunization results in more than 95% of people being protected.

The Haemophilus influenzae type B vaccine, also known as Hib vaccine, is a vaccine used to prevent Haemophilus influenzae type b (Hib) infection. In countries that include it as a routine vaccine, rates of severe Hib infections have decreased more than 90%. It has therefore resulted in a decrease in the rate of meningitis, pneumonia, and epiglottitis.

Diphtheria vaccine is a toxoid vaccine against diphtheria, an illness caused by Corynebacterium diphtheriae. Its use has resulted in a more than 90% decrease in number of cases globally between 1980 and 2000. The first dose is recommended at six weeks of age with two additional doses four weeks apart, after which it is about 95% effective during childhood. Three further doses are recommended during childhood. It is unclear if further doses later in life are needed.
Pertussis vaccine is a vaccine that protects against whooping cough (pertussis). There are two main types: whole-cell vaccines and acellular vaccines. The whole-cell vaccine is about 78% effective while the acellular vaccine is 71–85% effective. The effectiveness of the vaccines appears to decrease by between 2 and 10% per year after vaccination with a more rapid decrease with the acellular vaccines. The vaccine is only available in combination with tetanus and diphtheria vaccines. Pertussis vaccine is estimated to have saved over 500,000 lives in 2002.
A Vaccine Information Statement (VIS) is a document designed by the Centers for Disease Control and Prevention (CDC) to provide information to a patient receiving a vaccine in the United States. The National Childhood Vaccine Injury Act requires that medical professionals provide a VIS to patients before receiving certain vaccinations. The VIS includes information about the vaccine's benefits and risks, a description of the vaccine, indications and contraindications, instructions for patients experiencing an adverse reaction, and additional resources.

Tetanus vaccine, also known as tetanus toxoid (TT), is a toxoid vaccine used to prevent tetanus. During childhood, five doses are recommended, with a sixth given during adolescence.

DTaP-IPV vaccine is a combination vaccine whose full generic name is diphtheria and tetanus toxoids and acellular pertussis adsorbed and inactivated poliovirus vaccine (IPV).
DTaP-IPV/Hib vaccine is a 5-in-1 combination vaccine that protects against diphtheria, tetanus, whooping cough, polio, and Haemophilus influenzae type B.
DPT-Hib vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and whole-cell pertussis vaccine adsorbed with Hib conjugate vaccine, sometimes abbreviated to DPT-Hib. It protects against the infectious diseases diphtheria, tetanus, pertussis, and Haemophilus influenzae type B.
DTaP-Hib vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and acellular pertussis adsorbed with Haemophilus B conjugate vaccine, sometimes abbreviated to DTaP-Hib. It protects against the infectious diseases diphtheria, tetanus, pertussis, and Haemophilus influenzae type B.

A hexavalent vaccine, or 6-in-1 vaccine, is a combination vaccine with six individual vaccines conjugated into one, intended to protect people from multiple diseases. The term usually refers to the children's vaccine that protects against diphtheria, tetanus, pertussis, poliomyelitis, haemophilus B, and hepatitis B, which is used in more than 90 countries around the world including in Europe, Canada, Australia, Jordan, and New Zealand.
DTP-HepB vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and whole-cell pertussis and hepatitis B (recombinant) vaccine (adsorbed) or DTP-Hep B. It protects against the infectious diseases diphtheria, tetanus, pertussis, and hepatitis B.
References
- ↑ "Diphtheria, Tetanus Toxoids, Acellular Pertussis, Hepatitis B (Recombinant), and Poliovirus (Inactivated) Vaccine". Drugs.com. Retrieved 15 July 2018.
- ↑ Centers for Disease Control and Prevention (CDC) (March 2003). "FDA licensure of diphtheria and tetanus toxoids and acellular pertussis adsorbed, hepatitis B (recombinant), and poliovirus vaccine combined, (PEDIARIX) for use in infants". MMWR. Morbidity and Mortality Weekly Report. 52 (10): 203–204. PMID 12653460.
- ↑ Centers for Disease Control and Prevention (CDC) (October 2008). "Licensure of a diphtheria and tetanus toxoids and acellular pertussis adsorbed and inactivated poliovirus vaccine and guidance for use as a booster dose". MMWR. Morbidity and Mortality Weekly Report. 57 (39): 1078–1079. PMID 18830212.
- ↑ Schillie S, Vellozzi C, Reingold A, Harris A, Haber P, Ward JW, Nelson NP (January 2018). "Prevention of Hepatitis B Virus Infection in the United States: Recommendations of the Advisory Committee on Immunization Practices". MMWR. Recommendations and Reports. 67 (1): 1–31. doi:10.15585/mmwr.rr6701a1. PMC 5837403 . PMID 29939980.
- ↑ "Pediarix". Food and Drug Administration (FDA). 21 February 2018. Archived from the original on 22 September 2019. Retrieved 22 September 2019.
- 1 2 3 4 5 6 7 "Diphtheria-Tetanus-Pertussis Vaccine Information Statement | CDC". www.cdc.gov. 27 June 2022. Archived from the original on 28 December 2019. Retrieved 4 December 2022.
- 1 2 Skibinski DA, Baudner BC, Singh M, O'Hagan DT (January 2011). "Combination vaccines". Journal of Global Infectious Diseases. 3 (1): 63–72. doi: 10.4103/0974-777X.77298 . PMC 3068581 . PMID 21572611.
- 1 2 3 4 5 "Your Child's Immunizations: Polio Vaccine (IPV) (for Parents) - Nemours KidsHealth". kidshealth.org. Archived from the original on 30 April 2023. Retrieved 4 December 2022.
- 1 2 3 "DTaP/IPV/Hib/HepB vaccine (6-in-1 vaccine)". www.nhsinform.scot. Archived from the original on 1 February 2023. Retrieved 4 December 2022.
- 1 2 3 4 "Hepatitis B". www.nhsinform.scot. Archived from the original on 4 February 2023. Retrieved 4 December 2022.
- 1 2 3 4 5 6 "About Combination Vaccines for Children | CDC". www.cdc.gov. 25 May 2022. Archived from the original on 7 March 2023. Retrieved 4 December 2022.
- 1 2 "History of polio vaccination". www.who.int. Archived from the original on 4 April 2023. Retrieved 4 December 2022.
- 1 2 "PEDIARIX Safety Data | PEDIARIX (Diphtheria and Tetanus Toxoids and Acellular Pertussis Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus Vaccine) for Healthcare Professionals". www.pediarix.com. Archived from the original on 30 March 2023. Retrieved 4 December 2022.
- 1 2 3 Center for Biologics Evaluation and Research (6 November 2019). "PEDIARIX". FDA. Archived from the original on 22 September 2019. Retrieved 16 December 2019.
- 1 2 3 4 "PEDIARIX Administration | PEDIARIX (Diphtheria and Tetanus Toxoids and Acellular Pertussis Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus Vaccine) for Healthcare Professionals". www.pediarix.com. Archived from the original on 30 March 2023. Retrieved 4 December 2022.
- 1 2 3 4 5 "Ask the Experts: Combination Vaccines". www.immunize.org. Archived from the original on 6 May 2023. Retrieved 4 December 2022.
- 1 2 "State of Pertussis | PEDIARIX (Diphtheria and Tetanus Toxoids and Acellular Pertussis Adsorbed, Hepatitis B (Recombinant) and Inactivated Poliovirus Vaccine) for Healthcare Professionals". www.pediarix.com. Archived from the original on 30 March 2023. Retrieved 4 December 2022.