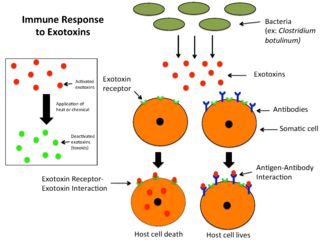
An exotoxin is a toxin secreted by bacteria. An exotoxin can cause damage to the host by destroying cells or disrupting normal cellular metabolism. They are highly potent and can cause major damage to the host. Exotoxins may be secreted, or, similar to endotoxins, may be released during lysis of the cell. Gram negative pathogens may secrete outer membrane vesicles containing lipopolysaccharide endotoxin and some virulence proteins in the bounding membrane along with some other toxins as intra-vesicular contents, thus adding a previously unforeseen dimension to the well-known eukaryote process of membrane vesicle trafficking, which is quite active at the host–pathogen interface.

The DPT vaccine or DTP vaccine is a class of combination vaccines against three infectious diseases in humans: diphtheria, pertussis, and tetanus. The vaccine components include diphtheria and tetanus toxoids and either killed whole cells of the bacterium that causes pertussis or pertussis antigens. The term toxoid refers to vaccines which use an inactivated toxin produced by the pathogen which they are targeted against to generate an immune response. In this way, the toxoid vaccine generates an immune response which is targeted against the toxin which is produced by the pathogen and causes disease, rather than a vaccine which is targeted against the pathogen itself. The whole cells or antigens will be depicted as either "DTwP" or "DTaP", where the lower-case "w" indicates whole-cell inactivated pertussis and the lower-case "a" stands for "acellular". In comparison to alternative vaccine types, such as live attenuated vaccines, the DTP vaccine does not contain any live pathogen, but rather uses inactivated toxoid to generate an immune response; therefore, there is not a risk of use in populations that are immune compromised since there is not any known risk of causing the disease itself. As a result, the DTP vaccine is considered a safe vaccine to use in anyone and it generates a much more targeted immune response specific for the pathogen of interest.

A vaccination schedule is a series of vaccinations, including the timing of all doses, which may be either recommended or compulsory, depending on the country of residence. A vaccine is an antigenic preparation used to produce active immunity to a disease, in order to prevent or reduce the effects of infection by any natural or "wild" pathogen. Vaccines go through multiple phases of trials to ensure safety and effectiveness.
In immunology, the Arthus reaction is a type of local type III hypersensitivity reaction. Type III hypersensitivity reactions are immune complex-mediated, and involve the deposition of antigen/antibody complexes mainly in the vascular walls, serosa, and glomeruli. This reaction is usually encountered in experimental settings following the injection of antigens.
A booster dose is an extra administration of a vaccine after an earlier (primer) dose. After initial immunization, a booster provides a re-exposure to the immunizing antigen. It is intended to increase immunity against that antigen back to protective levels after memory against that antigen has declined through time. For example, tetanus shot boosters are often recommended every 10 years, by which point memory cells specific against tetanus lose their function or undergo apoptosis.
Immunization during pregnancy is the administration of a vaccine to a pregnant individual. This may be done either to protect the individual from disease or to induce an antibody response, such that the antibodies cross the placenta and provide passive immunity to the infant after birth. In many countries, including the US, Canada, UK, Australia and New Zealand, vaccination against influenza, COVID-19 and whooping cough is routinely offered during pregnancy.

Hepatitis B vaccine is a vaccine that prevents hepatitis B. The first dose is recommended within 24 hours of birth with either two or three more doses given after that. This includes those with poor immune function such as from HIV/AIDS and those born premature. It is also recommended that health-care workers be vaccinated. In healthy people, routine immunization results in more than 95% of people being protected.

The Haemophilus influenzae type B vaccine, also known as Hib vaccine, is a vaccine used to prevent Haemophilus influenzae type b (Hib) infection. In countries that include it as a routine vaccine, rates of severe Hib infections have decreased more than 90%. It has therefore resulted in a decrease in the rate of meningitis, pneumonia, and epiglottitis.

Diphtheria vaccine is a toxoid vaccine against diphtheria, an illness caused by Corynebacterium diphtheriae. Its use has resulted in a more than 90% decrease in number of cases globally between 1980 and 2000. The first dose is recommended at six weeks of age with two additional doses four weeks apart, after which it is about 95% effective during childhood. Three further doses are recommended during childhood. It is unclear if further doses later in life are needed.
Pertussis vaccine is a vaccine that protects against whooping cough (pertussis). There are two main types: whole-cell vaccines and acellular vaccines. The whole-cell vaccine is about 78% effective while the acellular vaccine is 71–85% effective. The effectiveness of the vaccines appears to decrease by between 2 and 10% per year after vaccination with a more rapid decrease with the acellular vaccines. The vaccine is only available in combination with tetanus and diphtheria vaccines. Pertussis vaccine is estimated to have saved over 500,000 lives in 2002.
Meningococcal vaccine refers to any vaccine used to prevent infection by Neisseria meningitidis. Different versions are effective against some or all of the following types of meningococcus: A, B, C, W-135, and Y. The vaccines are between 85 and 100% effective for at least two years. They result in a decrease in meningitis and sepsis among populations where they are widely used. They are given either by injection into a muscle or just under the skin.

Cocooning, also known as the Cocoon Strategy, is a vaccination strategy to protect infants and other vulnerable individuals from infectious diseases by vaccinating those in close contact with them. If the people most likely to transmit an infection are immune, their immunity creates a "cocoon" of protection around the newborn.
A Vaccine Information Statement (VIS) is a document designed by the Centers for Disease Control and Prevention (CDC) to provide information to a patient receiving a vaccine in the United States. The National Childhood Vaccine Injury Act requires that medical professionals provide a VIS to patients before receiving certain vaccinations. The VIS includes information about the vaccine's benefits and risks, a description of the vaccine, indications and contraindications, instructions for patients experiencing an adverse reaction, and additional resources.

DTaP-IPV vaccine is a combination vaccine whose full generic name is diphtheria and tetanus toxoids and acellular pertussis adsorbed and inactivated poliovirus vaccine (IPV).
DTaP-IPV/Hib vaccine is a 5-in-1 combination vaccine that protects against diphtheria, tetanus, whooping cough, polio, and Haemophilus influenzae type B.
DTaP-IPV-HepB vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and acellular pertussis adsorbed, hepatitis B (recombinant) and inactivated polio vaccine or DTaP-IPV-Hep B. It protects against the infectious diseases diphtheria, tetanus, pertussis, poliomyelitis, and hepatitis B.
DPT-Hib vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and whole-cell pertussis vaccine adsorbed with Hib conjugate vaccine, sometimes abbreviated to DPT-Hib. It protects against the infectious diseases diphtheria, tetanus, pertussis, and Haemophilus influenzae type B.
DTaP-Hib vaccine is a combination vaccine whose generic name is diphtheria and tetanus toxoids and acellular pertussis adsorbed with Haemophilus B conjugate vaccine, sometimes abbreviated to DTaP-Hib. It protects against the infectious diseases diphtheria, tetanus, pertussis, and Haemophilus influenzae type B.

A hexavalent vaccine, or 6-in-1 vaccine, is a combination vaccine with six individual vaccines conjugated into one, intended to protect people from multiple diseases. The term usually refers to the children's vaccine that protects against diphtheria, tetanus, pertussis, poliomyelitis, haemophilus B, and hepatitis B, which is used in more than 90 countries around the world including in Europe, Canada, Australia, Jordan, and New Zealand.
Trudy Virginia Noller Murphy is an American pediatric infectious diseases physician, public health epidemiologist and vaccinologist. During the 1980s and 1990s, she conducted research at Southwestern Medical School in Dallas, Texas on three bacterial pathogens: Haemophilus influenzae type b (Hib), Streptococcus pneumoniae (pneumococcus), and methicillin-resistant Staphylococcus aureus (MRSA). Murphy's studies advanced understanding of how these organisms spread within communities, particularly among children attending day care centers. Her seminal work on Hib vaccines elucidated the effects of introduction of new Hib vaccines on both bacterial carriage and control of invasive Hib disease. Murphy subsequently joined the National Immunization Program at the Centers for Disease Control and Prevention (CDC) where she led multi-disciplinary teams in the Divisions of Epidemiology and Surveillance and The Viral Hepatitis Division. Among her most influential work at CDC was on Rotashield™, which was a newly licensed vaccine designed to prevent severe diarrheal disease caused by rotavirus. Murphy and her colleagues uncovered that the vaccine increased the risk of acute bowel obstruction (intussusception). This finding prompted suspension of the national recommendation to vaccinate children with Rotashield, and led the manufacturer to withdraw the vaccine from the market. For this work Murphy received the United States Department of Health and Human Services Secretary's Award for Distinguished Service in 2000, and the publication describing this work was recognized in 2002 by the Charles C. Shepard Science Award from the Centers for Disease Control and Prevention.












