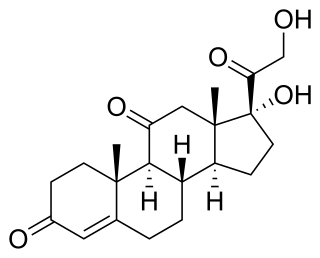
Cortisone is a pregnene (21-carbon) steroid hormone. It is a naturally-occurring corticosteroid metabolite that is also used as a pharmaceutical prodrug; it is not synthesized in the adrenal glands. Cortisol is converted by the action of the enzyme corticosteroid 11-beta-dehydrogenase isozyme 2 into the inactive metabolite cortisone, particularly in the kidneys. Cortisone is converted back to the active steroid cortisol by the action of the enzyme 11β-Hydroxysteroid dehydrogenase type 1, particularly in the liver.

Cortisol is a steroid hormone, in the glucocorticoid class of hormones. When used as a medication, it is known as hydrocortisone.

Congenital adrenal hyperplasia due to 3β-hydroxysteroid dehydrogenase deficiency is an uncommon form of congenital adrenal hyperplasia (CAH) resulting from a mutation in the gene for one of the key enzymes in cortisol synthesis by the adrenal gland, 3β-hydroxysteroid dehydrogenase (3β-HSD) type II (HSD3B2). As a result, higher levels of 17α-hydroxypregnenolone appear in the blood with adrenocorticotropic hormone (ACTH) challenge, which stimulates adrenal corticosteroid synthesis.

Enoxolone is a pentacyclic triterpenoid derivative of the beta-amyrin type obtained from the hydrolysis of glycyrrhizic acid, which was obtained from the herb liquorice. It is used in flavoring and it masks the bitter taste of drugs like aloe and quinine. It is effective in the treatment of peptic ulcer and also has expectorant (antitussive) properties. It has some additional pharmacological properties with possible antiviral, antifungal, antiprotozoal, and antibacterial activities.
11β-Hydroxysteroid dehydrogenase enzymes catalyze the conversion of inert 11 keto-products (cortisone) to active cortisol, or vice versa, thus regulating the access of glucocorticoids to the steroid receptors.

Apparent mineralocorticoid excess is an autosomal recessive disorder causing hypertension, hypernatremia and hypokalemia. It results from mutations in the HSD11B2 gene, which encodes the kidney isozyme of 11β-hydroxysteroid dehydrogenase type 2. In an unaffected individual, this isozyme inactivates circulating cortisol to the less active metabolite cortisone. The inactivating mutation leads to elevated local concentrations of cortisol in the aldosterone sensitive tissues like the kidney. Cortisol at high concentrations can cross-react and activate the mineralocorticoid receptor due to the non-selectivity of the receptor, leading to aldosterone-like effects in the kidney. This is what causes the hypokalemia, hypertension, and hypernatremia associated with the syndrome. Patients often present with severe hypertension and end-organ changes associated with it like left ventricular hypertrophy, retinal, renal and neurological vascular changes along with growth retardation and failure to thrive. In serum both aldosterone and renin levels are low.

Corticosteroid 11-β-dehydrogenase isozyme 2 also known as 11-β-hydroxysteroid dehydrogenase 2 is an enzyme that in humans is encoded by the HSD11B2 gene.

11β-Hydroxysteroid dehydrogenase type 1, also known as cortisone reductase, is an NADPH-dependent enzyme highly expressed in key metabolic tissues including liver, adipose tissue, and the central nervous system. In these tissues, HSD11B1 reduces cortisone to the active hormone cortisol that activates glucocorticoid receptors. It belongs to the family of short-chain dehydrogenases. It is encoded by the HSD11B1 gene.
Pseudohyperaldosteronism is a medical condition which mimics the effects of elevated aldosterone (hyperaldosteronism) by presenting with high blood pressure (hypertension), low blood potassium levels (hypokalemia), metabolic alkalosis, and low levels of plasma renin activity (PRA). However, unlike hyperaldosteronism, this conditions exhibits low or normal levels of aldosterone in the blood. Causes include genetic disorders, acquired conditions, metabolic disorders, and dietary imbalances including excessive consumption of licorice. Confirmatory diagnosis depends on the specific root cause and may involve blood tests, urine tests, or genetic testing; however, all forms of this condition exhibit abnormally low concentrations of both plasma renin activity (PRA) and plasma aldosterone concentration (PAC) which differentiates this group of conditions from other forms of secondary hypertension. Treatment is tailored to the specific cause and focuses on symptom control, blood pressure management, and avoidance of triggers.

Trilostane, sold under the brand names Modrenal and Vetoryl among others, is a medication which has been used in the treatment of Cushing's syndrome, Conn's syndrome, and postmenopausal breast cancer in humans. It was withdrawn for use in humans in the United States in the 1990s but was subsequently approved for use in veterinary medicine in the 2000s to treat Cushing's syndrome in dogs. It is taken by mouth.

Fluoxymesterone, sold under the brand names Halotestin and Ultandren among others, is an androgen and anabolic steroid (AAS) medication which is used in the treatment of low testosterone levels in men, delayed puberty in boys, breast cancer in women, and anemia. It is taken by mouth.
3β-Hydroxysteroid dehydrogenase/Δ5-4 isomerase (3β-HSD) is an enzyme that catalyzes the biosynthesis of the steroid progesterone from pregnenolone, 17α-hydroxyprogesterone from 17α-hydroxypregnenolone, and androstenedione from dehydroepiandrosterone (DHEA) in the adrenal gland. It is the only enzyme in the adrenal pathway of corticosteroid synthesis that is not a member of the cytochrome P450 family. It is also present in other steroid-producing tissues, including the ovary, testis and placenta. In humans, there are two 3β-HSD isozymes encoded by the HSD3B1 and HSD3B2 genes.
17β-Hydroxysteroid dehydrogenases, also 17-ketosteroid reductases (17-KSR), are a group of alcohol oxidoreductases which catalyze the reduction of 17-ketosteroids and the dehydrogenation of 17β-hydroxysteroids in steroidogenesis and steroid metabolism. This includes interconversion of DHEA and androstenediol, androstenedione and testosterone, and estrone and estradiol.

Formebolone, also known as formyldienolone, as well as 2-formyl-11α-hydroxy-17α-methyl-δ1-testosterone, is an orally active anabolic-androgenic steroid (AAS) described as an anticatabolic and anabolic drug that is or has been marketed in Spain and Italy. As an AAS, it shows some anabolic activity, though it is inferior to testosterone in terms of potency, but is said to have virtually no androgenic activity. Formebolone counteracts the catabolic effects of potent glucocorticoids like dexamethasone phosphate. A close analogue, roxibolone, shows similar antiglucocorticoid activity to formebolone but, in contrast, is devoid of activity as an AAS.

Roxibolone (INN), also known as 11β,17β-dihydroxy-17α-methyl-3-oxoandrosta-1,4-diene-2-carboxylic acid, is a steroidal antiglucocorticoid described as an anticholesterolemic (cholesterol-lowering) and anabolic drug which was never marketed. Roxibolone is closely related to formebolone, which shows antiglucocorticoid activity similarly and, with the exception of having a carboxaldehyde group at the C2 position instead of a carboxylic acid group, roxibolone is structurally almost identical to. The 2-decyl ester of roxibolone, decylroxibolone, is a long-acting prodrug of roxibolone with similar activity.
Cortisone reductase deficiency is caused by dysregulation of the 11β-hydroxysteroid dehydrogenase type 1 enzyme (11β-HSD1), otherwise known as cortisone reductase, a bi-directional enzyme, which catalyzes the interconversion of cortisone to cortisol in the presence of NADH as a co-factor. If levels of NADH are low, the enzyme catalyses the reverse reaction, from cortisol to cortisone, using NAD+ as a co-factor.
Cortisol is a glucocorticoid that plays a variety of roles in many different biochemical pathways, including, but not limited to: gluconeogenesis, suppressing immune system responses and carbohydrate metabolism.
One of the symptoms of cortisone reductase deficiency is hyperandrogenism, resulting from activation of the Hypothalamic–pituitary–adrenal axis. The deficiency has been known to exhibit symptoms of other disorders such as Polycystic Ovary Syndrome in women. Cortisone Reductase Deficiency alone has been reported in fewer than ten cases in total, all but one case were women. Elevated activity of 11β-HSD1 can lead to obesity or Type II Diabetes, because of the role of cortisol in carbohydrate metabolism and gluconeogenesis.

11α-Hydroxyprogesterone (11α-OHP), or 11α-hydroxypregn-4-ene-3,20-dione is an endogenous steroid and metabolite of progesterone. It is a weak antiandrogen, and is devoid of androgenic, estrogenic, and progestogenic activity.

21-Deoxycortisone, also known as 21-desoxycortisone, 11-keto-17α-hydroxyprogesterone, or 17α-hydroxypregn-4-ene-3,11,20-trione, is a naturally occurring, endogenous steroid and minor intermediate and metabolite in corticosteroid metabolism. It is related to 21-deoxycortisol (11β,17α-dihydroxyprogesterone) and is reversibly formed from it by 11β-hydroxysteroid dehydrogenase, analogously to the reversible formation of cortisone from cortisol. 21-Deoxycortisone can be transformed into cortisone by 21-hydroxylase.
Adrenal steroids are steroids that are derived from the adrenal glands. They include corticosteroids, which consist of glucocorticoids like cortisol and mineralocorticoids like aldosterone, adrenal androgens like dehydroepiandrosterone (DHEA), DHEA sulfate (DHEA-S), and androstenedione (A4), and neurosteroids like DHEA and DHEA-S, as well as pregnenolone and pregnenolone sulfate (P5-S). Adrenal steroids are specifically produced in the adrenal cortex.
A progesterone synthesis inhibitor, or progestogen synthesis inhibitor, is a type of drug which inhibits the enzymatic synthesis of progesterone. They include:















