
Iron(III) oxide or ferric oxide is the inorganic compound with the formula Fe2O3. It is one of the three main oxides of iron, the other two being iron(II) oxide (FeO), which is rare; and iron(II,III) oxide (Fe3O4), which also occurs naturally as the mineral magnetite. As the mineral known as hematite, Fe2O3 is the main source of iron for the steel industry. Fe2O3 is readily attacked by acids. Iron(III) oxide is often called rust, and to some extent this label is useful, because rust shares several properties and has a similar composition; however, in chemistry, rust is considered an ill-defined material, described as Hydrous ferric oxide.

A ferrite is a ceramic material made by mixing and firing iron(III) oxide with one or more additional metallic elements, such as strontium, barium, manganese, nickel, and zinc. They are ferrimagnetic, meaning they are attracted by magnetic fields and can be magnetized to become permanent magnets. Unlike other ferromagnetic materials, most ferrites are not electrically conductive, making them useful in applications like magnetic cores for transformers to suppress eddy currents. Ferrites can be divided into two families based on their resistance to being demagnetized.

Salen refers to a tetradentate C2-symmetric ligand synthesized from salicylaldehyde (sal) and ethylenediamine (en). It may also refer to a class of compounds, which are structurally related to the classical salen ligand, primarily bis-Schiff bases. Salen ligands are notable for coordinating a wide range of different metals, which they can often stabilise in various oxidation states. For this reason salen-type compounds are used as metal deactivators. Metal salen complexes also find use as catalysts.
Nanomaterial-based catalysts are usually heterogeneous catalysts broken up into metal nanoparticles in order to enhance the catalytic process. Metal nanoparticles have high surface area, which can increase catalytic activity. Nanoparticle catalysts can be easily separated and recycled. They are typically used under mild conditions to prevent decomposition of the nanoparticles.

Lithium cobalt oxide, sometimes called lithium cobaltate or lithium cobaltite, is a chemical compound with formula LiCoO
2. The cobalt atoms are formally in the +3 oxidation state, hence the IUPAC name lithium cobalt(III) oxide.
Magnetic nanoparticles are a class of nanoparticle that can be manipulated using magnetic fields. Such particles commonly consist of two components, a magnetic material, often iron, nickel and cobalt, and a chemical component that has functionality. While nanoparticles are smaller than 1 micrometer in diameter, the larger microbeads are 0.5–500 micrometer in diameter. Magnetic nanoparticle clusters that are composed of a number of individual magnetic nanoparticles are known as magnetic nanobeads with a diameter of 50–200 nanometers. Magnetic nanoparticle clusters are a basis for their further magnetic assembly into magnetic nanochains. The magnetic nanoparticles have been the focus of much research recently because they possess attractive properties which could see potential use in catalysis including nanomaterial-based catalysts, biomedicine and tissue specific targeting, magnetically tunable colloidal photonic crystals, microfluidics, magnetic resonance imaging, magnetic particle imaging, data storage, environmental remediation, nanofluids, optical filters, defect sensor, magnetic cooling and cation sensors.

Cobalt(II) hydroxide or cobaltous hydroxide is the inorganic compound with the formula Co(OH)
2, consisting of divalent cobalt cations Co2+
and hydroxide anions OH−
. The pure compound, often called the "beta form" is a pink solid insoluble in water.

Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, produced by reductive smelting, is a hard, lustrous, silvery metal.

In materials and electric battery research, cobalt oxide nanoparticles usually refers to particles of cobalt(II,III) oxide Co
3O
4 of nanometer size, with various shapes and crystal structures.

Iron–platinum nanoparticles are 3D superlattices composed of an approximately equal atomic ratio of Fe and Pt. Under standard conditions, FePt NPs exist in the face-centered cubic phase but can change to a chemically ordered face-centered tetragonal phase as a result of thermal annealing. Currently there are many synthetic methods such as water-in-oil microemulsion, one-step thermal synthesis with metal precursors, and exchanged-coupled assembly for making FePt NPs. An important property of FePt NPs is their superparamagnetic character below 10 nanometers. The superparamagnetism of FePt NPs has made them attractive candidates to be used as MRI/CT scanning agents and a high-density recording material.
The borophosphates are mixed anion compounds containing borate and phosphate anions, which may be joined together by a common oxygen atom. Compounds that contain water or hydroxy groups can also be included in the class of compounds.
Borate phosphates are mixed anion compounds containing separate borate and phosphate anions. They are distinct from the borophosphates where the borate is linked to a phosphate via a common oxygen atom. The borate phosphates have a higher ratio of cations to number of borates and phosphates, as compared to the borophosphates.
Fluoride nitrates are mixed anion compounds that contain both fluoride ions and nitrate ions. Compounds are known for some amino acids and for some heavy elements. Some transition metal fluorido complexes that are nitrates are also known. There are also fluorido nitrato complex ions known in solution.
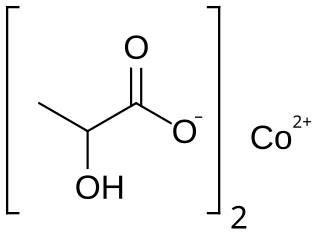
Cobalt lactate is a chemical compound, a salt of cobalt and lactic acid with the formula Co(C3H5O3)2.

Transition metal carbonate and bicarbonate complexes are coordination compounds containing carbonate (CO32-) and bicarbonate (HCO3-) as ligands. The inventory of complexes is large, enhanced by the fact that the carbonate ligand can bind metal ions in a variety of bonding modes.
Copper(II) laurate is an metal-organic compound with the chemical formula Cu(C
11H
23COO)
2. It is classified as a metallic soap, i.e. a metal derivative of a fatty acid.
Nickel(II) laurate is an metal-organic compound with the chemical formula C
24H
46NiO
4. It is classified as a metallic soap, i.e. a metal derivative of a fatty acid.
Zinc laurate is an metal-organic compound with the chemical formula C
24H
46ZnO
4. It is classified as a metallic soap, i.e. a metal derivative of a fatty acid.
Manganese laurate is an metal-organic compound with the chemical formula C
24H
48MnO
4. The compound is classified as a metallic soap, i.e. a metal derivative of a fatty acid.
Potassium laurate is a metal-organic compound with the chemical formula C
12H
23KO
2. The compound is classified as a metallic soap, i.e. a metal derivative of a fatty acid.









