An organic acid is an organic compound with acidic properties. The most common organic acids are the carboxylic acids, whose acidity is associated with their carboxyl group –COOH. Sulfonic acids, containing the group –SO2OH, are relatively stronger acids. Alcohols, with –OH, can act as acids but they are usually very weak. The relative stability of the conjugate base of the acid determines its acidity. Other groups can [also] confer acidity, usually weakly: the thiol group –SH, the enol group, and the phenol group. In biological systems, organic compounds containing these groups are generally referred to as organic acids.
Pyruvic acid (IUPAC name: 2-oxopropanoic acid, also called acetoic acid) (CH3COCOOH) is the simplest of the alpha-keto acids, with a carboxylic acid and a ketone functional group. Pyruvate, the conjugate base, CH3COCOO−, is an intermediate in several metabolic pathways throughout the cell.

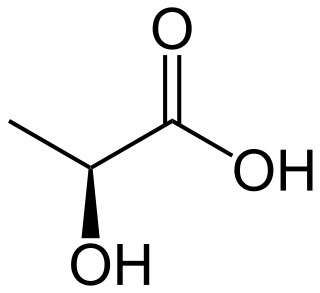
Lactic acid is an organic acid. It has a molecular formula CH3CH(OH)COOH. It is white in the solid state and it is miscible with water. When in the dissolved state, it forms a colorless solution. Production includes both artificial synthesis as well as natural sources. Lactic acid is an alpha-hydroxy acid (AHA) due to the presence of a hydroxyl group adjacent to the carboxyl group. It is used as a synthetic intermediate in many organic synthesis industries and in various biochemical industries. The conjugate base of lactic acid is called lactate. The name of the derived acyl group is lactoyl.
Lactic acid fermentation is a metabolic process by which glucose or other six-carbon sugars are converted into cellular energy and the metabolite lactate, which is lactic acid in solution. It is an anaerobic fermentation reaction that occurs in some bacteria and animal cells, such as muscle cells.

Lactic acidosis is a medical condition characterized by a build-up of lactate in the body, with formation of an excessively low pH in the bloodstream. It is a form of metabolic acidosis, in which excessive acid accumulates due to a problem with the body's oxidative metabolism.

Dairy cattle are cattle bred for the ability to produce large quantities of milk, from which dairy products are made. Dairy cattle generally are of the species Bos taurus.

Terephthalic acid is an organic compound with formula C6H4(CO2H)2. This white solid is a commodity chemical, used principally as a precursor to the polyester PET, used to make clothing and plastic bottles. Several million tons are produced annually. The common name is derived from the turpentine-producing tree Pistacia terebinthus and phthalic acid.

The Cori cycle, named after its discoverers, Carl Ferdinand Cori and Gerty Cori, is a metabolic pathway in which lactate, produced by anaerobic glycolysis in muscles, is transported to the liver and converted to glucose, which then returns to the muscles and is cyclically metabolized back to lactate.

Calcium lactate is a white crystalline salt with formula C
6H
10CaO
6, consisting of two lactate anions H
3C(CHOH)CO−
2 for each calcium cation Ca2+
. It forms several hydrates, the most common being the pentahydrate C
6H
10CaO
6·5H
2O.

Cobalt(II) oxide is an inorganic compound that has been described as an olive-green or gray solid. It is used extensively in the ceramics industry as an additive to create blue-colored glazes and enamels, as well as in the chemical industry for producing cobalt(II) salts. A related material is cobalt(II,III) oxide, a black solid with the formula Co3O4.

Fermentation is a metabolic process that produces chemical changes in organic substances through the action of enzymes. In biochemistry, it is narrowly defined as the extraction of energy from carbohydrates in the absence of oxygen. In food production, it may more broadly refer to any process in which the activity of microorganisms brings about a desirable change to a foodstuff or beverage. The science of fermentation is known as zymology.

Total mixed ration (TMR) is a method of feeding dairy cattle. The purpose of feeding a TMR diet is that each cow can consume the required level of nutrients in each bite. A cow's ration should include good quality forages, a balance of grains and proteins, vitamins and minerals.

Sodium lactate is the sodium salt of lactic acid, and has a mild saline taste. It is produced by fermentation of a sugar source, such as corn or beets, and then, by neutralizing the resulting lactic acid to create a compound having the formula NaC3H5O3.

Zirconium lactate is the zirconium salt of lactic acid. It is used in some deodorants. Zirconium carboxylates adopt highly complex structures and are heterogeneous compositions with the approximate formula Zr(OH)4-n(O2CCHOHCH3)n(H2O)x where 1 < n < 3.

A dry cow refers to a dairy cow that is in a stage of their lactation cycle where milk production ceases prior to calving. This part of their lactation cycle is referred to as the cows dry period and typically last between 40 and 65 days. Dry cows are typically divided into two groups: far-off and close-up. Once the cow has entered this stage, producers will seal the cows teat while following a veterinarian recommended, dry cow therapy for their herd. This dry period is a critical part of their lactation cycle and is important for the cows health, the newborn calf and future milk production, as it allows the cow time to rest, eat and prepare for birth. During this time, the cow will produce colostrum for the newly born calf.

Lithium lactate is a chemical compound, a salt of lithium and lactic acid with the formula CH3CH(OH)COOLi, an amorphous solid, very soluble in water.
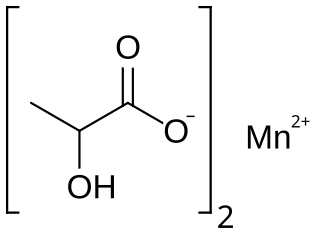
Manganese lactate is an organic chemical compound, a salt of manganese and lactic acid with the formula Mn(C3H5O3)2. The compound forms light pink crystals, soluble in water, forming crystalline hydrates.
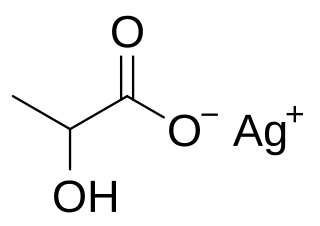
Silver lactate is an organic chemical compound, a salt of silver and lactic acid with the formula CH3CH(OH)COOAg.

Cadmium lactate is an organic chemical compound, a salt of cadmium and lactic acid with the formula Cd(C3H5O3)2.

Zinc lactate is a chemical compound, a salt of zinc and lactic acid with the formula Zn(C3H5O3)2.

















