Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri Mendeleev in 1869, though it was not identified until 1923, by Coster and Hevesy, making it the last stable element to be discovered. Hafnium is named after Hafnia, the Latin name for Copenhagen, where it was discovered.

Zirconium is a chemical element with the symbol Zr and atomic number 40. The name zirconium is taken from the name of the mineral zircon, the most important source of zirconium. It is a lustrous, grey-white, strong transition metal that closely resembles hafnium and, to a lesser extent, titanium. Zirconium is mainly used as a refractory and opacifier, although small amounts are used as an alloying agent for its strong resistance to corrosion. Zirconium forms a variety of inorganic and organometallic compounds such as zirconium dioxide and zirconocene dichloride, respectively. Five isotopes occur naturally, three of which are stable. Zirconium compounds have no known biological role.

Group 4 is a group of elements in the periodic table. It contains the elements titanium (Ti), zirconium (Zr), hafnium (Hf) and rutherfordium (Rf). This group lies in the d-block of the periodic table. The group itself has not acquired a trivial name; it belongs to the broader grouping of the transition metals.
In inorganic chemistry, Fajans' rules, formulated by Kazimierz Fajans in 1923, are used to predict whether a chemical bond will be covalent or ionic, and depend on the charge on the cation and the relative sizes of the cation and anion. They can be summarized in the following table:

Hafnium(IV) chloride is the inorganic compound with the formula HfCl4. This colourless solid is the precursor to most hafnium organometallic compounds. It has a variety of highly specialized applications, mainly in materials science and as a catalyst.

Copper(I) iodide is the inorganic compound with the formula CuI. It is also known as cuprous iodide. It is useful in a variety of applications ranging from organic synthesis to cloud seeding.
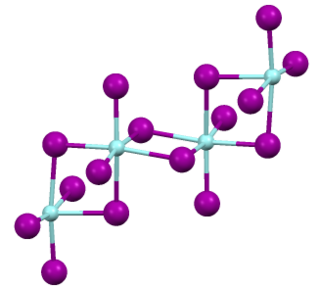
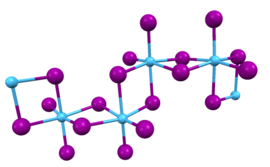
Zirconium(IV) iodide is the chemical compound with the formula ZrI4. It is the most readily available iodide of zirconium. It is an orange-coloured solid that degrades in the presence of water. The compound was once prominent as an intermediate in the purification of zirconium metal.

Hafnium carbide is a chemical compound of hafnium and carbon. With a melting point of about 3900 °C it is one of the most refractory binary compounds known. However, it has a low oxidation resistance, with the oxidation starting at temperatures as low as 430 °C. This compound may be seen on future spacecraft as part of the heat shield.
Tantalum hafnium carbide is a refractory chemical compound with a general formula TaxHfy-xCy, which can be considered as a solid solution of tantalum carbide and hafnium carbide.

The group 2 elements are known to form organometallic compounds. Of these, organomagnesium compounds, usually in the form of Grignard reagents are widely used in organic chemistry, while the other organometallic compounds of this group are largely academic.

Organozirconium compounds are organometallic compounds containing a carbon to zirconium chemical bond. Organozirconium chemistry is the corresponding science exploring properties, structure and reactivity of these compounds. Organozirconium compounds have been widely studied, in part because they are useful catalysts in Ziegler-Natta polymerization.
Organochromium chemistry is a branch of organometallic chemistry that deals with organic compounds containing a chromium to carbon bond and their reactions. The field is of some relevance to organic synthesis. The relevant oxidation states for organochromium complexes encompass the entire range of possible oxidation states from –4 (d10) in Na4[Cr–IV(CO)4] to +6 (d0) in oxo-alkyl complexes like Cp*CrVI(=O)2Me.

Diarsenic tetraiodide is an inorganic compound of arsenic and iodine. It is a dark red metastable solid. The compound is a closely related to the better characterized diphosphorus tetraiodide. Identified in the late 19th century with the correct empirical formula, the compound was assigned the formula (As2I4) several years later.

Hafnium tetrabromide is the inorganic compound with the formula HfBr4. It is the most common bromide of hafnium. It is a colorless, diamagnetic moisture sensitive solid that sublimes in vacuum. It adopts a structure very similar to that of zirconium tetrabromide, featuring tetrahedral Hf centers, in contrast to the polymeric nature of hafnium tetrachloride.
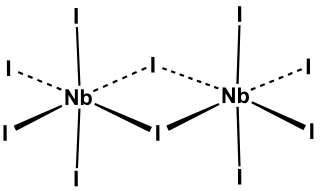
Niobium pentaiodide is the inorganic compound with the formula Nb2I10. It is a yellow, moisture-sensitive, diamagnetic solid. It forms upon heating metallic niobium with iodine. Its structure has been verified by single crystal X-ray diffraction, which reveals an edge-shared bioctahedral structure, as observed for many pentahalides of Nb and Ta. The bonds to the bridging iodides are 0.3 Å longer than the terminal Nb-I bonds.
Werner Urland is a German chemist whose name is imprinted in the pioneering implementation of the Angular Overlap Model for the interpretation of optical and magnetic properties of rare-earth coordination compounds. This approach receives a renewed value in the context of the vogue around the lanthanide-based new materials, such as achieving magnets at molecular scale, or designing new phosphor materials.

Hafnium disulfide is an inorganic compound of hafnium and sulfur. It is a layered dichalcogenide with the chemical formula is HfS2. A few atomic layers of this material can be exfoliated using the standard Scotch Tape technique (see graphene) and used for the fabrication of a field-effect transistor. High-yield synthesis of HfS2 has also been demonstrated using liquid phase exfoliation, resulting in the production of stable few-layer HfS2 flakes. Hafnium disulfide powder can be produced by reacting hydrogen sulfide and hafnium oxides at 500–1300 °C.
A hydridonitride is a chemical compound that contains hydride and nitride ions in a single phase. These compounds are distinct from inorganic amides and imides as the hydrogen does not share a bond with nitrogen, and contain a larger proportion of metals.
Caesium azide or cesium azide is an inorganic compound of caesium and azide with the formula CsN
3.
The telluride iodides are chemical compounds that contain both telluride ions (Te2−) and iodide ions (I−). They are in the class of mixed anion compounds or chalcogenide halides.