Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri Mendeleev in 1869, though it was not identified until 1923, by Dirk Coster and George de Hevesy, making it the penultimate stable element to be discovered. Hafnium is named after Hafnia, the Latin name for Copenhagen, where it was discovered.

Hafnium(IV) chloride is the inorganic compound with the formula HfCl4. This colourless solid is the precursor to most hafnium organometallic compounds. It has a variety of highly specialized applications, mainly in materials science and as a catalyst.
Zirconium(IV) bromide is the inorganic compound with the formula ZrBr4. This colourless solid is the principal precursor to other Zr–Br compounds.

Hafnium carbide (HfC) is a chemical compound of hafnium and carbon. Previously the material was estimated to have a melting point of about 3,900 °C. More recent tests have been able to conclusively prove that the substance has an even higher melting point of 3,958 °C exceeding those of tantalum carbide and tantalum hafnium carbide which were both previously estimated to be higher. However, it has a low oxidation resistance, with the oxidation starting at temperatures as low as 430 °C. Experimental testing in 2018 confirmed the higher melting point yielding a result of 3,982 (±30°C) with a small possibility that the melting point may even exceed 4,000°C.

Hafnium(IV) oxide is the inorganic compound with the formula HfO
2. Also known as hafnium dioxide or hafnia, this colourless solid is one of the most common and stable compounds of hafnium. It is an electrical insulator with a band gap of 5.3~5.7 eV. Hafnium dioxide is an intermediate in some processes that give hafnium metal.
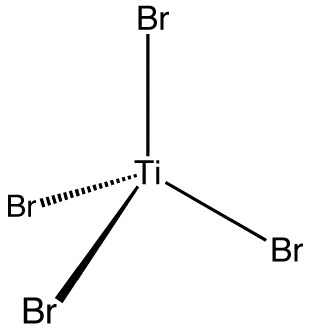
Titanium tetrabromide is the chemical compound with the formula TiBr4. It is the most volatile transition metal bromide. The properties of TiBr4 are an average of TiCl4 and TiI4. Some key properties of these four-coordinated Ti(IV) species are their high Lewis acidity and their high solubility in nonpolar organic solvents. TiBr4 is diamagnetic, reflecting the d0 configuration of the metal centre.

Silicon tetrabromide is the inorganic compound with the formula SiBr4. This colorless liquid has a suffocating odor due to its tendency to hydrolyze with release of hydrogen bromide. The general properties of silicon tetrabromide closely resemble those of the more commonly used silicon tetrachloride.

Zirconium(III) chloride is an inorganic compound with formula ZrCl3. It is a blue-black solid that is highly sensitive to air.
tetrabromide may refer to:

Hafnium tetrafluoride is the inorganic compound with the formula HfF4. It is a white solid. It adopts the same structure as zirconium tetrafluoride, with 8-coordinate Hf(IV) centers.

Hafnium acetylacetonate, also known as Hf(acac)4, is a coordination compound with formula Hf(C5H7O2)4. This white solid is the main hafnium complex of acetylacetonate. The complex has a square antiprismatic geometry with eight nearly equivalent Hf-O bonds. The molecular symmetry is D2, i.e., the complex is chiral. It is prepared from hafnium tetrachloride and acetylacetone, and base. Zr(acac)4 is very similar in structure and properties.
Titanium(III) bromide is the inorganic compound with the formula TiBr3. It is a blue black paramagnetic solid with a reddish reflection. It has few applications, although it is a catalyst for the polymerization of alkenes.

Hafnium(IV) iodide is the inorganic compound with the formula HfI4. It is a red-orange, moisture sensitive, sublimable solid that is produced by heating a mixture of hafnium with excess iodine. It is an intermediate in the crystal bar process for producing hafnium metal.

Hafnium disulfide is an inorganic compound of hafnium and sulfur. It is a layered dichalcogenide with the chemical formula is HfS2. A few atomic layers of this material can be exfoliated using the standard Scotch Tape technique (see graphene) and used for the fabrication of a field-effect transistor. High-yield synthesis of HfS2 has also been demonstrated using liquid phase exfoliation, resulting in the production of stable few-layer HfS2 flakes. Hafnium disulfide powder can be produced by reacting hydrogen sulfide and hafnium oxides at 500–1300 °C.

Selenium tetrabromide is an inorganic compound with a chemical formula SeBr4.

Germanium tetrabromide is an inorganic compound with the formula GeBr4. It can be formed by reacting solid germanium and gaseous bromine.

Hafnium(IV) nitrate is an inorganic compound, a salt of hafnium and nitric acid with the chemical formula Hf(NO3)4.
Osmium tetrabromide is the inorganic compound with the formula OsBr4. A black solid, this compound can be produced by heating osmium tetrachloride and bromine under pressure.
Hafnium compounds are compounds containing the element hafnium (Hf). Due to the lanthanide contraction, the ionic radius of hafnium(IV) (0.78 ångström) is almost the same as that of zirconium(IV) (0.79 angstroms). Consequently, compounds of hafnium(IV) and zirconium(IV) have very similar chemical and physical properties. Hafnium and zirconium tend to occur together in nature and the similarity of their ionic radii makes their chemical separation rather difficult. Hafnium tends to form inorganic compounds in the oxidation state of +4. Halogens react with it to form hafnium tetrahalides. At higher temperatures, hafnium reacts with oxygen, nitrogen, carbon, boron, sulfur, and silicon. Some compounds of hafnium in lower oxidation states are known.

Hafnium(III) iodide is an inorganic compound of hafnium and iodine with the formula HfI3. It is a black solid.















