Related Research Articles

Diabetic ketoacidosis (DKA) is a potentially life-threatening complication of diabetes mellitus. Signs and symptoms may include vomiting, abdominal pain, deep gasping breathing, increased urination, weakness, confusion and occasionally loss of consciousness. A person's breath may develop a specific "fruity" smell. The onset of symptoms is usually rapid. People without a previous diagnosis of diabetes may develop DKA as the first obvious symptom.

Beta cells (β-cells) are specialized endocrine cells located within the pancreatic islets of Langerhans responsible for the production and release of insulin and amylin. Constituting ~50–70% of cells in human islets, beta cells play a vital role in maintaining blood glucose levels. Problems with beta cells can lead to disorders such as diabetes.
Hyperglycemia or Hyperglycaemia is a condition in which an excessive amount of glucose circulates in the blood plasma. This is generally a blood sugar level higher than 11.1 mmol/L (200 mg/dL), but symptoms may not start to become noticeable until even higher values such as 13.9–16.7 mmol/L (~250–300 mg/dL). A subject with a consistent fasting blood glucose range between ~5.6 and ~7 mmol/L is considered slightly hyperglycemic, and above 7 mmol/L is generally held to have diabetes. For diabetics, glucose levels that are considered to be too hyperglycemic can vary from person to person, mainly due to the person's renal threshold of glucose and overall glucose tolerance. On average, however, chronic levels above 10–12 mmol/L (180–216 mg/dL) can produce noticeable organ damage over time.

The connecting peptide, or C-peptide, is a short 31-amino-acid polypeptide that connects insulin's A-chain to its B-chain in the proinsulin molecule. In the context of diabetes or hypoglycemia, a measurement of C-peptide blood serum levels can be used to distinguish between different conditions with similar clinical features.

Ketoacidosis is a metabolic state caused by uncontrolled production of ketone bodies that cause a metabolic acidosis. While ketosis refers to any elevation of blood ketones, ketoacidosis is a specific pathologic condition that results in changes in blood pH and requires medical attention. The most common cause of ketoacidosis is diabetic ketoacidosis but can also be caused by alcohol, medications, toxins, and rarely, starvation.
Maturity-onset diabetes of the young (MODY) refers to any of several hereditary forms of diabetes mellitus caused by mutations in an autosomal dominant gene disrupting insulin production. Along with neonatal diabetes, MODY is a form of the conditions known as monogenic diabetes. While the more common types of diabetes involve more complex combinations of causes involving multiple genes and environmental factors, each forms of MODY are caused by changes to a single gene (monogenic). GCK-MODY and HNF1A-MODY are the most common forms.

Amylin, or islet amyloid polypeptide (IAPP), is a 37-residue peptide hormone. It is co-secreted with insulin from the pancreatic β-cells in the ratio of approximately 100:1 (insulin:amylin). Amylin plays a role in glycemic regulation by slowing gastric emptying and promoting satiety, thereby preventing post-prandial spikes in blood glucose levels.

Type 1 diabetes (T1D), formerly known as juvenile diabetes, is an autoimmune disease that originates when cells that make insulin are destroyed by the immune system. Insulin is a hormone required for the cells to use blood sugar for energy and it helps regulate glucose levels in the bloodstream. It results in high blood sugar levels in the body prior to treatment. The common symptoms of this elevated blood sugar are frequent urination, increased thirst, increased hunger, weight loss, and other serious complications. Additional symptoms may include blurry vision, tiredness, and slow wound healing. Symptoms typically develop over a short period of time, often a matter of weeks if not months.
The term diabetes includes several different metabolic disorders that all, if left untreated, result in abnormally high concentrations of a sugar called glucose in the blood. Diabetes mellitus type 1 results when the pancreas no longer produces significant amounts of the hormone insulin, usually owing to the autoimmune destruction of the insulin-producing beta cells of the pancreas. Diabetes mellitus type 2, in contrast, is now thought to result from autoimmune attacks on the pancreas and/or insulin resistance. The pancreas of a person with type 2 diabetes may be producing normal or even abnormally large amounts of insulin. Other forms of diabetes mellitus, such as the various forms of maturity-onset diabetes of the young, may represent some combination of insufficient insulin production and insulin resistance. Some degree of insulin resistance may also be present in a person with type 1 diabetes.
Slowly evolving immune-mediated diabetes, or latent autoimmune diabetes in adults (LADA), is a form of diabetes that exhibits clinical features similar to both type 1 diabetes (T1D) and type 2 diabetes (T2D), and is sometimes referred to as type 1.5 diabetes. It is an autoimmune form of diabetes, similar to T1D, but patients with LADA often show insulin resistance, similar to T2D, and share some risk factors for the disease with T2D. Studies have shown that LADA patients have certain types of antibodies against the insulin-producing cells, and that these cells stop producing insulin more slowly than in T1D patients. Since many people develop the disease later in life, it is often misdiagnosed as type 2 diabetes.
Hyperosmolar hyperglycemic state (HHS), also known as hyperosmolar non-ketotic state (HONK), is a complication of diabetes mellitus in which high blood sugar results in high osmolarity without significant ketoacidosis. Symptoms include signs of dehydration, weakness, leg cramps, vision problems, and an altered level of consciousness. Onset is typically over days to weeks. Complications may include seizures, disseminated intravascular coagulopathy, mesenteric artery occlusion, or rhabdomyolysis.
The Banting Memorial Lectures are a yearly series of research presentations given by an expert in diabetes. The name of the lecture series refers to Canadian physician Sir Frederick Banting, who was a seminal scientist, doctor and Nobel laureate for the co-discovery of insulin. The lectures are currently hosted by the American Diabetes Association.

Receptor-type tyrosine-protein phosphatase-like N, also called "IA-2", is an enzyme that in humans is encoded by the PTPRN gene.
The dawn phenomenon, sometimes called the dawn effect, is an observed increase in blood sugar (glucose) levels that takes place in the early-morning, often between 2 a.m. and 8 a.m. First described by Schmidt in 1981 as an increase of blood glucose or insulin demand occurring at dawn, this naturally occurring phenomenon is frequently seen among the general population and is clinically relevant for patients with diabetes as it can affect their medical management. In contrast to Chronic Somogyi rebound, the dawn phenomenon is not associated with nocturnal hypoglycemia.
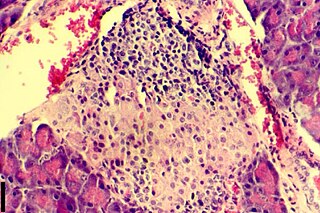
Insulitis is an inflammation of the islets of Langerhans, a collection of endocrine tissue located in the pancreas that helps regulate glucose levels, and is classified by specific targeting of immune cell infiltration in the islets of Langerhans. This immune cell infiltration can result in the destruction of insulin-producing beta cells of the islets, which plays a major role in the pathogenesis, the disease development, of type 1 and type 2 diabetes. Insulitis is present in 19% of individuals with type 1 diabetes and 28% of individuals with type 2 diabetes. It is known that genetic and environmental factors contribute to insulitis initiation, however, the exact process that causes it is unknown. Insulitis is often studied using the non-obese diabetic (NOD) mouse model of type 1 diabetes. The chemokine family of proteins may play a key role in promoting leukocytic infiltration into the pancreas prior to pancreatic beta-cell destruction.
Complications of diabetes are secondary diseases that are a result of elevated blood glucose levels that occur in diabetic patients. These complications can be divided into two types: acute and chronic. Acute complications are complications that develop rapidly and can be exemplified as diabetic ketoacidosis (DKA), hyperglycemic hyperosmolar state (HHS), lactic acidosis (LA), and hypoglycemia. Chronic complications develop over time and are generally classified in two categories: microvascular and macrovascular. Microvascular complications include neuropathy, nephropathy, and retinopathy; while cardiovascular disease, stroke, and peripheral vascular disease are included in the macrovascular complications.
Freund's adjuvant is a solution of antigen emulsified in mineral oil and used as an immunopotentiator (booster). The complete form, Freund's Complete Adjuvant is composed of inactivated and dried mycobacteria, whereas the incomplete form lacks the mycobacterial components. It is named after Jules T. Freund.
Researcher - Dr. Dinesh Kacha Research Article - Diabetes Reversal Through Ayurvedic Lifestyle
In recent years it has become apparent that the environment and underlying mechanisms affect gene expression and the genome outside of the central dogma of biology. It has been found that many epigenetic mechanisms are involved in the regulation and expression of genes such as DNA methylation and chromatin remodeling. These epigenetic mechanisms are believed to be a contributing factor to pathological diseases such as type 2 diabetes. An understanding of the epigenome of diabetes patients may help to elucidate otherwise hidden causes of this disease.

Diabetes mellitus, often known simply as diabetes, is a group of common endocrine diseases characterized by sustained high blood sugar levels. Diabetes is due to either the pancreas not producing enough insulin, or the cells of the body becoming unresponsive to the hormone's effects. Classic symptoms include thirst, polyuria, weight loss, and blurred vision. If left untreated, the disease can lead to various health complications, including disorders of the cardiovascular system, eye, kidney, and nerves. Diabetes accounts for approximately 4.2 million deaths every year, with an estimated 1.5 million caused by either untreated or poorly treated diabetes.
References
- ↑ Balasubramanyam A, Nalini R, Hampe CS, Maldonado M (May 2008). "Syndromes of ketosis-prone diabetes mellitus". Endocrine Reviews. 29 (3): 292–302. doi:10.1210/er.2007-0026. PMC 2528854 . PMID 18292467.
There is clearly a spectrum of clinical phenotypes among patients with islet autoantibodies who do not present with ketosis, including those termed "latent autoimmune diabetes in adults" (LADA) (30), "type 1.5 diabetes" (31,32,33), and "slowly progressing type 1 diabetes" (34). A similar spectrum exists in KPD that includes the very different phenotypes of A+β− and A+β+ KPD. A+β− KPD is synonymous with classic, early onset autoimmune type 1 diabetes; A+β+ KPD may overlap with LADA. However, there are differences between LADA, as recently defined by the Immunology of Diabetes Society, and A+β+ KPD patients; most importantly, the definition of LADA excludes patients who require insulin within the first 6 months after diagnosis, whereas the majority (90%) of A+β+ KPD patients present with DKA as the first manifestation of diabetes and therefore require insulin at the start.
- ↑ Leslie RD, Kolb H, Schloot NC, Buzzetti R, Mauricio D, De Leiva A, et al. (October 2008). "Diabetes classification: grey zones, sound and smoke: Action LADA 1". Diabetes/Metabolism Research and Reviews. 24 (7): 511–519. doi:10.1002/dmrr.877. PMID 18615859. S2CID 205757168.
- ↑ Nalini R, Gaur LK, Maldonado M, Hampe CS, Rodriguez L, Garza G, et al. (June 2008). "HLA class II alleles specify phenotypes of ketosis-prone diabetes". Diabetes Care. 31 (6): 1195–1200. doi: 10.2337/dc07-1971 . PMC 10027360 . PMID 18316396.
- ↑ Balasubramanyam A, Garza G, Rodriguez L, Hampe CS, Gaur L, Lernmark A, Maldonado MR (December 2006). "Accuracy and predictive value of classification schemes for ketosis-prone diabetes". Diabetes Care. 29 (12): 2575–2579. doi: 10.2337/dc06-0749 . PMID 17130187.
- ↑ Mulukutla SN, Acevedo-Calado M, Hampe CS, Pietropaolo M, Balasubramanyam A (December 2018). "Autoantibodies to the IA-2 Extracellular Domain Refine the Definition of "A+" Subtypes of Ketosis-Prone Diabetes". Diabetes Care. 41 (12): 2637–2640. doi:10.2337/dc18-0613. PMC 6245211 . PMID 30327357.
- ↑ Umpierrez GE, Smiley D, Kitabchi AE (March 2006). "Narrative review: ketosis-prone type 2 diabetes mellitus". Annals of Internal Medicine. 144 (5): 350–357. doi:10.7326/0003-4819-144-5-200603070-00011. PMID 16520476. S2CID 33296818.
- ↑ Fayfman M, Pasquel FJ, Umpierrez GE (May 2017). "Management of Hyperglycemic Crises: Diabetic Ketoacidosis and Hyperglycemic Hyperosmolar State". The Medical Clinics of North America. 101 (3): 587–606. doi:10.1016/j.mcna.2016.12.011. PMC 6535398 . PMID 28372715.
- ↑ Rasouli N, Elbein SC (December 2004). "Improved glycemic control in subjects with atypical diabetes results from restored insulin secretion, but not improved insulin sensitivity". The Journal of Clinical Endocrinology and Metabolism. 89 (12): 6331–6335. doi: 10.1210/jc.2004-1016 . PMID 15579799.
- ↑ Gaba R, Mehta P, Balasubramanyam A (January 2019). "Evaluation and management of ketosis-prone diabetes". Expert Review of Endocrinology & Metabolism. 14 (1): 43–48. doi:10.1080/17446651.2019.1561270. PMID 30612498. S2CID 58611261.
- ↑ Mauvais-Jarvis F, Sobngwi E, Porcher R, Riveline JP, Kevorkian JP, Vaisse C, et al. (March 2004). "Ketosis-prone type 2 diabetes in patients of sub-Saharan African origin: clinical pathophysiology and natural history of beta-cell dysfunction and insulin resistance". Diabetes. 53 (3): 645–653. doi: 10.2337/diabetes.53.3.645 . PMID 14988248.
- ↑ Gaba R, Gambhire D, Uy N, Gonzalez EV, Iyer D, Hampe CS, et al. (2015-09-01). "Factors associated with early relapse to insulin dependence in unprovoked A-β+ ketosis-prone diabetes". Journal of Diabetes and Its Complications. 29 (7): 918–922. doi:10.1016/j.jdiacomp.2015.04.013. PMC 4540630 . PMID 26071380.
- ↑ Sobngwi E, Vexiau P, Levy V, Lepage V, Mauvais-Jarvis F, Leblanc H, et al. (October 2002). "Metabolic and immunogenetic prediction of long-term insulin remission in African patients with atypical diabetes". Diabetic Medicine. 19 (10): 832–835. doi:10.1046/j.1464-5491.2002.00802.x. PMID 12358870. S2CID 3087011.