Omalizumab, sold under the brand name Xolair, is a medication to treat asthma, nasal polyps, and urticaria (hives).

GD2 is a disialoganglioside expressed on tumors of neuroectodermal origin, including human neuroblastoma and melanoma, with highly restricted expression on normal tissues, principally to the cerebellum and peripheral nerves in humans.

Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective, to induce a specific cell signal, to cause the immune system to attack specific cells, or to bring a drug to a specific cell type.
Abagovomab is a mouse anti-idiotype monoclonal antibody whose variable epitope mirrors a tumour antigen (CA-125) highly expressed in the epithelial ovarian cancer. Abagovomab does not bind directly to CA-125, but it works as a "surrogate" antigen, enabling the immune system to identify and attack tumour cells displaying the CA-125 protein. Through this, it is hoped that the body's immune system may be able to combat any remaining individual tumour cells and thus prevent recurrence of the disease.
Bavituximab (PGN401) is a human-mouse chimeric monoclonal antibody against phosphatidylserine, which is a component of cell membranes that is exposed when a cell is transformed into solid tumor cancer cell or dies, and when cells are infected with hepatitis C. The process of cell death is highly controlled and so there usually no immune response to phosphatidylserine but when bavituximab binds to it, the conjugate appears to stimulate an immune response in humans.

Belimumab, sold under the brand name Benlysta, is a human monoclonal antibody that inhibits B-cell activating factor (BAFF), also known as B-lymphocyte stimulator (BLyS). It is approved in the United States and Canada, and the European Union to treat systemic lupus erythematosus and lupus nephritis.
Metelimumab (CAT-192) is a human IgG4 monoclonal antibody that neutralizes TGF beta 1 which had been chosen for further development for the treatment of diffuse cutaneous systemic sclerosis, also known as scleroderma. It was dropped from further development in favour of fresolimumab, which was being developed by Genzyme as of 2006.
Matuzumab is a humanized monoclonal antibody for the treatment of cancer. It binds to the epidermal growth factor receptor (EGFR) with high affinity. The mouse monoclonal antibody (mAb425) from which matuzumab was developed at the Wistar Institute in Philadelphia, Pennsylvania
Mitumomab (BEC-2) is a mouse anti-BEC-2 monoclonal antibody investigated for the treatment of small cell lung carcinoma in combination with BCG vaccination. Mitumomab attacks tumour cells, while the vaccine is thought to activate the immune system. It was developed by ImClone and Merck.
CD4 immunoadhesin is a recombinant fusion protein consisting of a combination of CD4 and the fragment crystallizable region, similarly known as immunoglobulin. It belongs to the antibody (Ig) gene family. CD4 is a surface receptor for human immunodeficiency virus (HIV). The CD4 immunoadhesin molecular fusion allow the protein to possess key functions from each independent subunit. The CD4 specific properties include the gp120-binding and HIV-blocking capabilities. Properties specific to immunoglobulin are the long plasma half-life and Fc receptor binding. The properties of the protein means that it has potential to be used in AIDS therapy as of 2017. Specifically, CD4 immunoadhesin plays a role in antibody-dependent cell-mediated cytotoxicity (ADCC) towards HIV-infected cells. While natural anti-gp120 antibodies exhibit a response towards uninfected CD4-expressing cells that have a soluble gp120 bound to the CD4 on the cell surface, CD4 immunoadhesin, however, will not exhibit a response. One of the most relevant of these possibilities is its ability to cross the placenta.
Tanox was a biopharmaceutical company based in Houston, Texas. The company was founded by two biomedical research scientists, Nancy T. Chang and Tse Wen Chang in March 1986 with $250,000, which was a large part of their family savings at that time. Both Changs grew up and received college education in chemistry in National Tsing Hua University in Taiwan and obtained Ph.D. degrees from Harvard University. For postdoctoral training, Tse Wen shifted to immunology and did research with Herman N. Eisen at the Center for Cancer Research, M.I.T. The two Changs successively became research managers and worked with a range of monoclonal antibody projects in Centocor, Inc. based in Malvern, Pennsylvania, from 1981 to 1985. The Changs were recruited by Baylor College of Medicine toward the end of 1985 and offered faculty positions in the Division of Molecular Virology. Soon after their arrival, they were encouraged by a high-ranking Baylor official and local business leaders to start a biotech venture in Houston. This was in a period of time when the economy of Houston was in slump as the result of the collapse of the oil industry.
Leronlimab is a humanized monoclonal antibody targeted against the CCR5 receptor found on T lymphocytes of the human immune system. It is being investigated as a potential therapy in the treatment of COVID-19, triple negative breast cancer, and HIV infection. The United States Food and Drug Administration has designated PRO 140 for fast-track approval. In February 2008, the drug entered Phase 2 clinical trials and a phase 3 trial was begun in 2015. In February 2018, Cytodyn Inc reported that the primary endpoint had been achieved in the PRO 140 pivotal combination therapy trial in HIV infection. In 2020 CytoDyn submitted a fast-track biologics license application for treatment of CCR5-tropic HIV-1 Infection.
Milatuzumab is an anti-CD74 humanized monoclonal antibody for the treatment of multiple myeloma non-Hodgkin's lymphoma and chronic lymphocytic leukemia.
Vedolizumab, sold under the brand name Entyvio, is a monoclonal antibody medication developed by Millennium Pharmaceuticals, Inc. for the treatment of ulcerative colitis and Crohn's disease. It binds to integrin α4β7, blocking the α4β7 integrin results in gut-selective anti-inflammatory activity.
CCR5 receptor antagonists are a class of small molecules that antagonize the CCR5 receptor. The C-C motif chemokine receptor CCR5 is involved in the process by which HIV, the virus that causes AIDS, enters cells. Hence antagonists of this receptor are entry inhibitors and have potential therapeutic applications in the treatment of HIV infections.
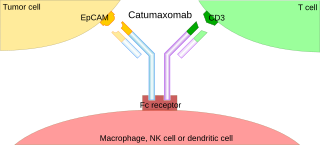
A trifunctional antibody is a monoclonal antibody with binding sites for two different antigens, typically CD3 and a tumor antigen, making it a type of bispecific monoclonal antibody. In addition, its intact Fc-part can bind to an Fc receptor on accessory cells like conventional monospecific antibodies. The net effect is that this type of drug links T cells and monocytes/macrophages, natural killer cells, dendritic cells or other Fc receptor expressing cells to the tumor cells, leading to their destruction.
TOL101, is a murine-monoclonal antibody specific for the human αβ T cell receptor. In 2010 it was an Investigational New Drug under development by Tolera Therapeutics, Inc.
Novimmune is a privately held Swiss pharmaceutical development company focusing on monoclonal antibody therapies for treatment of immune-related diseases. The company was founded in 1998.

Lobucavir is an antiviral drug that shows broad-spectrum activity against herpesviruses, hepatitis B and other hepadnaviruses, HIV/AIDS and cytomegalovirus. It initially demonstrated positive results in human clinical trials against hepatitis B with minimal adverse effects but was discontinued from further development following the discovery of increased risk of cancer associated with long-term use in mice. Although this carcinogenic risk is present in other antiviral drugs, such as zidovudine and ganciclovir that have been approved for clinical use, development was halted by Bristol-Myers Squibb, its manufacturer.

COVID-19 drug development is the research process to develop preventative therapeutic prescription drugs that would alleviate the severity of coronavirus disease 2019 (COVID-19). From early 2020 through 2021, several hundred drug companies, biotechnology firms, university research groups, and health organizations were developing therapeutic candidates for COVID-19 disease in various stages of preclinical or clinical research, with 419 potential COVID-19 drugs in clinical trials, as of April 2021.






