B cells, also known as B lymphocytes, are a type of white blood cell of the lymphocyte subtype. They function in the humoral immunity component of the adaptive immune system. B cells produce antibody molecules which may be either secreted or inserted into the plasma membrane where they serve as a part of B-cell receptors. When a naïve or memory B cell is activated by an antigen, it proliferates and differentiates into an antibody-secreting effector cell, known as a plasmablast or plasma cell. In addition, B cells present antigens and secrete cytokines. In mammals, including marsupials B cells mature in the bone marrow, which is at the core of most bones. In birds, B cells mature in the bursa of Fabricius, a lymphoid organ where they were first discovered by Chang and Glick, which is why the B stands for bursa and not bone marrow, as commonly believed.

In immunology, an Fc receptor is a protein found on the surface of certain cells – including, among others, B lymphocytes, follicular dendritic cells, natural killer cells, macrophages, neutrophils, eosinophils, basophils, human platelets, and mast cells – that contribute to the protective functions of the immune system. Its name is derived from its binding specificity for a part of an antibody known as the Fc region. Fc receptors bind to antibodies that are attached to infected cells or invading pathogens. Their activity stimulates phagocytic or cytotoxic cells to destroy microbes, or infected cells by antibody-mediated phagocytosis or antibody-dependent cell-mediated cytotoxicity. Some viruses such as flaviviruses use Fc receptors to help them infect cells, by a mechanism known as antibody-dependent enhancement of infection.

The B-cell receptor (BCR) is a transmembrane protein on the surface of a B cell. A B-cell receptor is composed of a membrane-bound immunoglobulin molecule and a signal transduction moiety. The former forms a type 1 transmembrane receptor protein, and is typically located on the outer surface of these lymphocyte cells. Through biochemical signaling and by physically acquiring antigens from the immune synapses, the BCR controls the activation of the B cell. B cells are able to gather and grab antigens by engaging biochemical modules for receptor clustering, cell spreading, generation of pulling forces, and receptor transport, which eventually culminates in endocytosis and antigen presentation. B cells' mechanical activity adheres to a pattern of negative and positive feedbacks that regulate the quantity of removed antigen by manipulating the dynamic of BCR–antigen bonds directly. Particularly, grouping and spreading increase the relation of antigen with BCR, thereby proving sensitivity and amplification. On the other hand, pulling forces delinks the antigen from the BCR, thus testing the quality of antigen binding.

Growth factor receptor-bound protein 2, also known as Grb2, is an adaptor protein involved in signal transduction/cell communication. In humans, the GRB2 protein is encoded by the GRB2 gene.

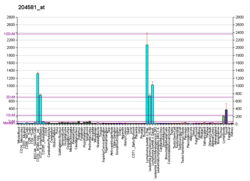
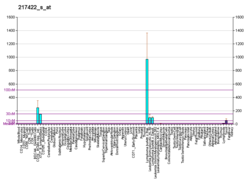
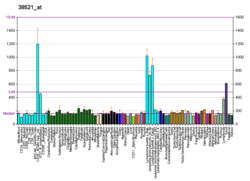
B-lymphocyte antigen CD19, also known as CD19 molecule, B-Lymphocyte Surface Antigen B4, T-Cell Surface Antigen Leu-12 and CVID3 is a transmembrane protein that in humans is encoded by the gene CD19. In humans, CD19 is expressed in all B lineage cells. Contrary to some early doubts, human plasma cells do express CD19, as confirmed by others. CD19 plays two major roles in human B cells: on the one hand, it acts as an adaptor protein to recruit cytoplasmic signaling proteins to the membrane; on the other, it works within the CD19/CD21 complex to decrease the threshold for B cell receptor signaling pathways. Due to its presence on all B cells, it is a biomarker for B lymphocyte development, lymphoma diagnosis and can be utilized as a target for leukemia immunotherapies.
Siglecs(Sialic acid-binding immunoglobulin-type lectins) are cell surface proteins that bind sialic acid. They are found primarily on the surface of immune cells and are a subset of the I-type lectins. There are 14 different mammalian Siglecs, providing an array of different functions based on cell surface receptor-ligand interactions.

Lymphocyte cytosolic protein 2, also known as LCP2 or SLP-76, is a signal-transducing adaptor protein expressed in T cells and myeloid cells and is important in the signaling of T-cell receptors (TCRs). As an adaptor protein, SLP-76 does not have catalytic functions, primarily binding other signaling proteins to form larger signaling complexes. It is a key component of the signaling pathways of receptors with immunoreceptor tyrosine-based activation motifs (ITAMs) such as T-cell receptors, its precursors, and receptors for the Fc regions of certain antibodies. SLP-76 is expressed in T-cells and related lymphocytes like natural killer cells.
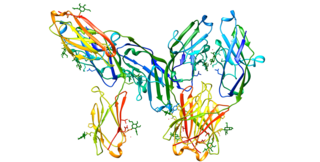
CD33 or Siglec-3 is a transmembrane receptor expressed on cells of myeloid lineage. It is usually considered myeloid-specific, but it can also be found on some lymphoid cells.
An immunoreceptor tyrosine-based inhibitory motif (ITIM), is a conserved sequence of amino acids that is found intracellularly in the cytoplasmic domains of many inhibitory receptors of the non-catalytic tyrosine-phosphorylated receptor family found on immune cells. These immune cells include T cells, B cells, NK cells, dendritic cells, macrophages and mast cells. ITIMs have similar structures of S/I/V/LxYxxI/V/L, where x is any amino acid, Y is a tyrosine residue that can be phosphorylated, S is the amino acid serine, I is the amino acid isoleucine, and V is the amino acid valine. ITIMs recruit SH2 domain-containing phosphatases, which inhibit cellular activation. ITIM-containing receptors often serve to target immunoreceptor tyrosine-based activation motif (ITAM)-containing receptors, resulting in an innate inhibition mechanism within cells. ITIM bearing receptors have important role in regulation of immune system allowing negative regulation at different levels of the immune response.
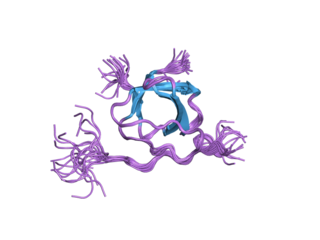
Tyrosine-protein kinase Lyn is a protein that in humans is encoded by the LYN gene.
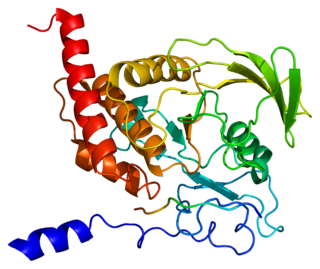
Tyrosine-protein phosphatase non-receptor type 6, also known as Src homology region 2 domain-containing phosphatase-1 (SHP-1), is an enzyme that in humans is encoded by the PTPN6 gene.

Src homology 2 (SH2) domain containing inositol polyphosphate 5-phosphatase 1(SHIP1) is an enzyme with phosphatase activity. SHIP1 is structured by multiple domain and is encoded by the INPP5D gene in humans. SHIP1 is expressed predominantly by hematopoietic cells but also, for example, by osteoblasts and endothelial cells. This phosphatase is important for the regulation of cellular activation. Not only catalytic but also adaptor activities of this protein are involved in this process. Its movement from the cytosol to the cytoplasmic membrane, where predominantly performs its function, is mediated by tyrosine phosphorylation of the intracellular chains of cell surface receptors that SHIP1 binds. Insufficient regulation of SHIP1 leads to different pathologies.

B-cell linker (BLNK) protein is expressed in B cells and macrophages and plays a large role in B cell receptor signaling. Like all adaptor proteins, BLNK has no known intrinsic enzymatic activity. Its function is to temporally and spatially coordinate and regulate downstream signaling effectors in B cell receptor (BCR) signaling, which is important in B cell development. Binding of these downstream effectors is dependent on BLNK phosphorylation. BLNK is encoded by the BLNK gene and is also known as SLP-65, BASH, and BCA.

Leukocyte immunoglobulin-like receptor subfamily B member 4 is a protein that in humans is encoded by the LILRB4 gene.
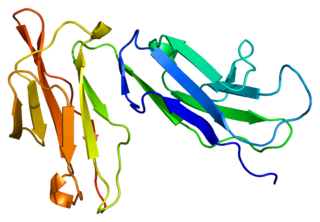
Fc fragment of IgG receptor IIb is a low affinity inhibitory receptor for the Fc region of immunoglobulin gamma (IgG). FCGR2B participates in the phagocytosis of immune complexes and in the regulation of antibody production by B lymphocytes.

Sialic acid-binding Ig-like lectin 8 is a protein that in humans is encoded by the SIGLEC8 gene. This gene is located on chromosome 19q13.4, about 330 kb downstream of the SIGLEC9 gene. Within the siglec family of transmembrane proteins, Siglec-8 belongs to the CD33-related siglec subfamily, a subfamily that has undergone rapid evolution.

Paired immunoglobulin-like type 2 receptor beta is a protein that in humans is encoded by the PILRB gene.

Sialic acid-binding Ig-like lectin 10 is a protein that in humans is encoded by the SIGLEC10 gene. Siglec-G is often referred to as the murine paralog of human Siglec-10

Cluster of differentiation CD79A also known as B-cell antigen receptor complex-associated protein alpha chain and MB-1 membrane glycoprotein, is a protein that in humans is encoded by the CD79A gene.

Sialic acid-binding Ig-like lectin 6 is a protein that in humans is encoded by the SIGLEC6 gene. The gene was originally named CD33L (CD33-like) due to similarities between these genes but later became known as OB-BP1 due to its ability to bind to this factor and, finally, SIGLEC6 as the sixth member of the SIGLEC family of receptors to be identified. The protein has also been given the CD designation CD327.