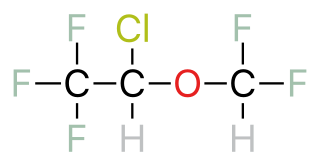General anaesthetics are often defined as compounds that induce a loss of consciousness in humans or loss of righting reflex in animals. Clinical definitions are also extended to include an induced coma that causes lack of awareness to painful stimuli, sufficient to facilitate surgical applications in clinical and veterinary practice. General anaesthetics do not act as analgesics and should also not be confused with sedatives. General anaesthetics are a structurally diverse group of compounds whose mechanisms encompass multiple biological targets involved in the control of neuronal pathways. The precise workings are the subject of some debate and ongoing research.

Anesthesia or anaesthesia is a state of controlled, temporary loss of sensation or awareness that is induced for medical or veterinary purposes. It may include some or all of analgesia, paralysis, amnesia, and unconsciousness. An individual under the effects of anesthetic drugs is referred to as being anesthetized.

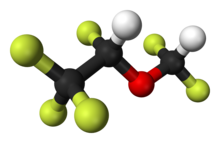
Halothane, sold under the brand name Fluothane among others, is a general anaesthetic. It can be used to induce or maintain anaesthesia. One of its benefits is that it does not increase the production of saliva, which can be particularly useful in those who are difficult to intubate. It is given by inhalation.
Sedation is the reduction of irritability or agitation by administration of sedative drugs, generally to facilitate a medical procedure or diagnostic procedure. Examples of drugs which can be used for sedation include isoflurane, diethyl ether, propofol, etomidate, ketamine, pentobarbital, lorazepam and midazolam.

Isoflurane, sold under the brand name Forane among others, is a general anesthetic. It can be used to start or maintain anesthesia; however, other medications are often used to start anesthesia, due to airway irritation with isoflurane. Isoflurane is given via inhalation.

Sevoflurane, sold under the brand name Sevorane, among others, is a sweet-smelling, nonflammable, highly fluorinated methyl isopropyl ether used as an inhalational anaesthetic for induction and maintenance of general anesthesia. After desflurane, it is the volatile anesthetic with the fastest onset. While its offset may be faster than agents other than desflurane in a few circumstances, its offset is more often similar to that of the much older agent isoflurane. While sevoflurane is only half as soluble as isoflurane in blood, the tissue blood partition coefficients of isoflurane and sevoflurane are quite similar. For example, in the muscle group: isoflurane 2.62 vs. sevoflurane 2.57. In the fat group: isoflurane 52 vs. sevoflurane 50. As a result, the longer the case, the more similar will be the emergence times for sevoflurane and isoflurane.

General anaesthesia (UK) or general anesthesia (US) is a method of medically inducing loss of consciousness that renders a patient unarousable even with painful stimuli. This effect is achieved by administering either intravenous or inhalational general anaesthetic medications, which often act in combination with an analgesic and neuromuscular blocking agent. Spontaneous ventilation is often inadequate during the procedure and intervention is often necessary to protect the airway. General anaesthesia is generally performed in an operating theater to allow surgical procedures that would otherwise be intolerably painful for a patient, or in an intensive care unit or emergency department to facilitate endotracheal intubation and mechanical ventilation in critically ill patients. Depending on the procedure, general anaesthesia may be optional or required. Regardless of whether a patient may prefer to be unconscious or not, certain pain stimuli could result in involuntary responses from the patient that may make an operation extremely difficult. Thus, for many procedures, general anaesthesia is required from a practical perspective.
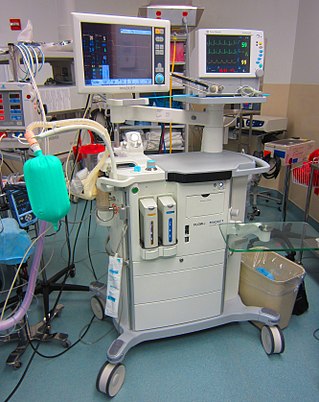
An anaesthetic machine or anesthesia machine is a medical device used to generate and mix a fresh gas flow of medical gases and inhalational anaesthetic agents for the purpose of inducing and maintaining anaesthesia.

An anesthetic or anaesthetic is a drug used to induce anesthesia — in other words, to result in a temporary loss of sensation or awareness. They may be divided into two broad classes: general anesthetics, which result in a reversible loss of consciousness, and local anesthetics, which cause a reversible loss of sensation for a limited region of the body without necessarily affecting consciousness.
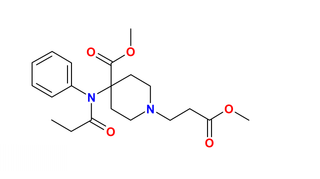
Remifentanil, marketed under the brand name Ultiva is a potent, short-acting synthetic opioid analgesic drug. It is given to patients during surgery to relieve pain and as an adjunct to an anaesthetic. Remifentanil is used for sedation as well as combined with other medications for use in general anesthesia. The use of remifentanil has made possible the use of high-dose opioid and low-dose hypnotic anesthesia, due to synergism between remifentanil and various hypnotic drugs and volatile anesthetics.

Enflurane is a halogenated ether. Developed by Ross Terrell in 1963, it was first used clinically in 1966. It was increasingly used for inhalational anesthesia during the 1970s and 1980s but is no longer in common use.

An inhalational anesthetic is a chemical compound possessing general anesthetic properties that is delivered via inhalation. They are administered through a face mask, laryngeal mask airway or tracheal tube connected to an anesthetic vaporiser and an anesthetic delivery system. Agents of significant contemporary clinical interest include volatile anesthetic agents such as isoflurane, sevoflurane and desflurane, as well as certain anesthetic gases such as nitrous oxide and xenon.
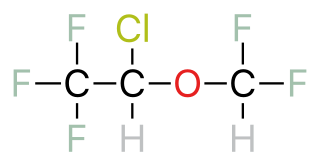
A halogenated ether is a subcategory of a larger group of chemicals known as ethers. An ether is an organic chemical that contains an ether group—an oxygen atom connected to two (substituted) alkyl groups. A good example of an ether is the solvent diethyl ether. What differentiates a halogenated ether from other types of ethers is the substitution (halogenation) of one or more hydrogen atoms with a halogen atom. Halogen atoms include fluorine, chlorine, bromine, and iodine.
Minimum alveolar concentration or MAC is the concentration, often expressed as a percentage by volume, of a vapour in the alveoli of the lungs that is needed to prevent movement in 50% of subjects in response to surgical (pain) stimulus. MAC is used to compare the strengths, or potency, of anaesthetic vapours. The concept of MAC was first introduced in 1965.
Veterinary anesthesia is anesthesia performed on non-human animals by a veterinarian or a Registered Veterinary Technician. Anesthesia is used for a wider range of circumstances in animals than in people, due to animals' inability to cooperate with certain diagnostic or therapeutic procedures. Veterinary anesthesia includes anesthesia of the major species: dogs, cats, horses, cattle, sheep, goats, and pigs, as well as all other animals requiring veterinary care such as birds, pocket pets, and wildlife.

Flurothyl (Indoklon) is a volatile liquid drug from the halogenated ether family, related to inhaled anaesthetic agents such as diethyl ether, but having the opposite effects, acting as a stimulant and convulsant. A clear and stable liquid, it has a mild ethereal odor whose vapors are non-flammable. It is excreted from the body by the lungs in an unchanged state.
Procedural sedation and analgesia (PSA) is a technique in which a sedating/dissociative medication is given, usually along with an analgesic medication, in order to perform non-surgical procedures on a patient. The overall goal is to induce a decreased level of consciousness while maintaining the patient's ability to breathe on their own. Airway protective reflexes are not compromised by this process and therefore endotracheal intubation is not required. PSA is commonly used in the emergency department, in addition to the operating room.
Emergence delirium is a condition in which emergence from general anesthesia is accompanied by psychomotor agitation. Some see a relation to pavor nocturnus while others see a relation to the excitement stage of anesthesia.
Blood–gas partition coefficient, also known as Ostwald coefficient for blood–gas, is a term used in pharmacology to describe the solubility of inhaled general anesthetics in blood. According to Henry's law, the ratio of the concentration in blood to the concentration in gas that is in contact with that blood, when the partial pressure in both compartments is equal, is nearly constant at sufficiently low concentrations. The partition coefficient is defined as this ratio and, therefore, has no units. The concentration of the anesthetic in blood includes the portion that is undissolved in plasma and the portion that is dissolved. The more soluble the inhaled anesthetic is in blood compared to in air, the more it binds to plasma proteins in the blood and the higher the blood–gas partition coefficient.
The following outline is provided as an overview of and topical guide to anesthesia: