
Nicotinic acetylcholine receptors, or nAChRs, are receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral nervous system, muscle, and many other tissues of many organisms. At the neuromuscular junction they are the primary receptor in muscle for motor nerve-muscle communication that controls muscle contraction. In the peripheral nervous system: (1) they transmit outgoing signals from the presynaptic to the postsynaptic cells within the sympathetic and parasympathetic nervous system, and (2) they are the receptors found on skeletal muscle that receive acetylcholine released to signal for muscular contraction. In the immune system, nAChRs regulate inflammatory processes and signal through distinct intracellular pathways. In insects, the cholinergic system is limited to the central nervous system.

Mecamylamine is a non-selective, non-competitive antagonist of the nicotinic acetylcholine receptors (nAChRs) that was introduced in the 1950s as an antihypertensive drug. In the United States, it was voluntarily withdrawn from the market in 2009 but was brought to market in 2013 as Vecamyl and eventually was marketed by Turing Pharmaceuticals.
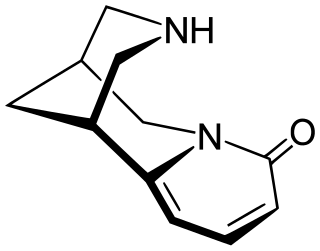
Cytisine, also known as baptitoxine, cytisinicline, or sophorine, is an alkaloid that occurs naturally in several plant genera, such as Laburnum and Cytisus of the family Fabaceae. It has been used medically to help with smoking cessation. Although widely used for smoking cessation in Eastern Europe, cytisine remains relatively unknown beyond it. However, it has been found effective in several randomized clinical trials, including some in the United States and a large one in New Zealand, and is being investigated in additional trials in the United States and a non-inferiority trial in Australia in which it is being compared head-to-head with the smoking cessation aid varenicline. It has also been used entheogenically via mescalbeans by some Native American groups, historically in the Rio Grande Valley predating even peyote.
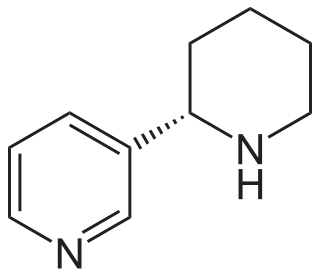
Anabasine is a pyridine and piperidine alkaloid found in the Tree Tobacco plant, as well as in the close relative of the common tobacco plant. It is a structural isomer of, and chemically similar to, nicotine. Its principal (historical) industrial use is as an insecticide.

Methyllycaconitine (MLA) is a diterpenoid alkaloid found in many species of Delphinium (larkspurs). In common with many other diterpenoid alkaloids, it is toxic to animals, although the acute toxicity varies with species. Early research was focused on identifying, and characterizing the properties of methyllycaconitine as one of the principal toxins in larkspurs responsible for livestock poisoning in the mountain rangelands of North America. Methyllycaconitine has been explored as a possible therapeutic agent for the treatment of spastic paralysis, and it has been shown to have insecticidal properties. Most recently, it has become an important molecular probe for studying the pharmacology of the nicotinic acetylcholine receptor.

Anatoxin-a, also known as Very Fast Death Factor (VFDF), is a secondary, bicyclic amine alkaloid and cyanotoxin with acute neurotoxicity. It was first discovered in the early 1960s in Canada, and was isolated in 1972. The toxin is produced by multiple genera of cyanobacteria and has been reported in North America, South America, Central America, Europe, Africa, Asia, and Oceania. Symptoms of anatoxin-a toxicity include loss of coordination, muscular fasciculations, convulsions and death by respiratory paralysis. Its mode of action is through the nicotinic acetylcholine receptor (nAchR) where it mimics the binding of the receptor's natural ligand, acetylcholine. As such, anatoxin-a has been used for medicinal purposes to investigate diseases characterized by low acetylcholine levels. Due to its high toxicity and potential presence in drinking water, anatoxin-a poses a threat to animals, including humans. While methods for detection and water treatment exist, scientists have called for more research to improve reliability and efficacy. Anatoxin-a is not to be confused with guanitoxin, another potent cyanotoxin that has a similar mechanism of action to that of anatoxin-a and is produced by many of the same cyanobacteria genera, but is structurally unrelated.

Anthony's poison arrow frog is a species of poison dart frog in the family Dendrobatidae. The species is endemic to Ecuador and Peru.
A nicotinic agonist is a drug that mimics the action of acetylcholine (ACh) at nicotinic acetylcholine receptors (nAChRs). The nAChR is named for its affinity for nicotine.

Pumiliotoxin 251D is a toxic organic compound. It is found in the skin of poison frogs from the genera Dendrobates, Epipedobates, Minyobates, and Phyllobates and toads from the genus Melanophryniscus. Its name comes from the pumiliotoxin family (PTXs) and its molecular mass of 251 daltons. When the toxin enters the bloodstream through cuts in the skin or by ingestion, it can cause hyperactivity, convulsions, cardiac arrest and ultimately death. It is especially toxic to arthropods, even at low concentrations.
The alpha-4 beta-2 nicotinic receptor, also known as the α4β2 receptor, is a type of nicotinic acetylcholine receptor implicated in learning, consisting of α4 and β2 subunits. It is located in the brain, where activation yields post- and presynaptic excitation, mainly by increased Na+ and K+ permeability.
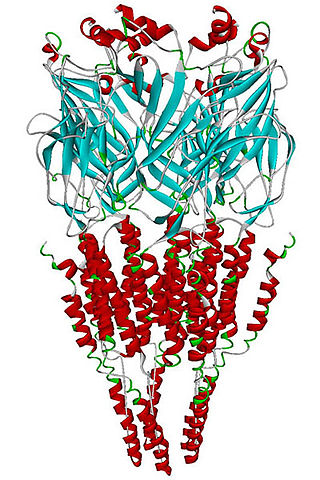
The alpha-7 nicotinic receptor, also known as the α7 receptor, is a type of nicotinic acetylcholine receptor implicated in long-term memory, consisting entirely of α7 subunits. As with other nicotinic acetylcholine receptors, functional α7 receptors are pentameric [i.e., (α7)5 stoichiometry].

Tebanicline is a potent synthetic nicotinic (non-opioid) analgesic drug developed by Abbott. It was developed as a less toxic analog of the potent poison dart frog-derived compound epibatidine, which is about 200 times stronger than morphine as an analgesic, but produces extremely dangerous toxic side effects. Like epibatidine, tebanicline showed potent analgesic activity against neuropathic pain in both animal and human trials, but with far less toxicity than its parent compound. It acts as a partial agonist at neuronal nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes.

Epiboxidine is a chemical compound which acts as a partial agonist at neural nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes. It was developed as a less toxic analogue of the potent frog-derived alkaloid epibatidine, which is around 200 times stronger than morphine as an analgesic but produces extremely dangerous toxic nicotinic side effects.

ABT-418 is a drug developed by Abbott, that has nootropic, neuroprotective and anxiolytic effects, and has been researched for treatment of both Alzheimer's disease and ADHD. It acts as an agonist at neural nicotinic acetylcholine receptors, subtype-selective binding with high affinity to the α4β2, α7/5-HT3, and α2β2 nicotinic acetylcholine receptors but not α3β4 receptors ABT-418 was reasonably effective for both applications and fairly well tolerated, but produced some side effects, principally nausea, and it is unclear whether ABT-418 itself will proceed to clinical development or if another similar drug will be used instead.

Histrionicotoxins are a group of related toxins found in the skin of poison frogs from the family Dendrobatidae, notably Oophaga histrionica, which are native to Colombia. It is likely that, as with other poison frog alkaloids, histrionicotoxins are not manufactured by the amphibians, but absorbed from insects in their diet and stored in glands in their skin. They are notably less toxic than other alkaloids found in poison frogs, yet their distinct structure acts as a neurotoxin by non-competitive inhibition of nicotinic acetylcholine receptors.

A-84543 is a drug developed by Abbott, which acts as an agonist at neural nicotinic acetylcholine receptors with high selectivity for the α4β2 subtype. It is widely used in scientific research into the structure and function of this receptor subtype and has been the lead compound for the development of a large family of related derivatives.
The alpha-3 beta-4 nicotinic receptor, also known as the α3β4 receptor and the ganglion-type nicotinic receptor, is a type of nicotinic acetylcholine receptor, consisting of α3 and β4 subunits. It is located in the autonomic ganglia and adrenal medulla, where activation yields post- and/or presynaptic excitation, mainly by increased Na+ and K+ permeability.

RJR-2429 is a drug that acts as an agonist at neural nicotinic acetylcholine receptors, binding to both the α3β4 and the α4β2 subtypes. RJR-2429 is stronger than nicotine but weaker than epibatidine in most assays, and with high affinity for both α3β4 and α4β2 subtypes, as well as the less studied α1βγδ subtype.

Phantasmidine is a toxic substance derived from the Ecuadorian poisonous frog Anthony's poison arrow frog, more commonly known as the “phantasmal poison frog”. It is a nicotinic agonist, meaning it binds to nicotinic receptors in the body and mimics the effects of the neurotransmitter acetylcholine. This causes the stimulation of the body's parasympathetic nervous system, which induces many inhibitory behaviors in the body such as decreased heart rate.
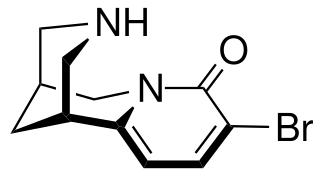
3-Bromocytisine is a derivative of the toxic alkaloid cytisine that acts as a highly potent agonist at neural nicotinic acetylcholine receptors, binding primarily to the α4β2 and α7 subtypes. 3-Bromocytisine is a full agonist at the α7 subtype while it is only a partial agonist at α4β2, but has an extremely strong binding affinity at α4β2 with 200-fold selectivity for α4β2 over α7. In animal studies 3-bromocytisine stimulates the release of dopamine and noradrenaline and increases locomotor activity.




















