
The substantia nigra (SN) is a basal ganglia structure located in the midbrain that plays an important role in reward and movement. Substantia nigra is Latin for "black substance", reflecting the fact that parts of the substantia nigra appear darker than neighboring areas due to high levels of neuromelanin in dopaminergic neurons. Parkinson's disease is characterized by the loss of dopaminergic neurons in the substantia nigra pars compacta.

MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) is a chemical precursor to the neurotoxin MPP+, which causes permanent symptoms of Parkinson's disease by destroying dopaminergic neurons in the substantia nigra of the brain. It has been used to study disease models in various animal studies.
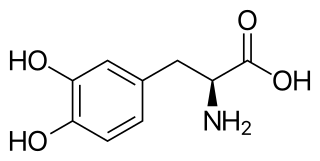
l-DOPA, also known as levodopa and l-3,4-dihydroxyphenylalanine, is an amino acid that is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize l-DOPA, make it via biosynthesis from the amino acid l-tyrosine. l-DOPA is the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), which are collectively known as catecholamines. Furthermore, l-DOPA itself mediates neurotrophic factor release by the brain and CNS. l-DOPA can be manufactured and in its pure form is sold as a psychoactive drug with the INN levodopa; trade names include Sinemet, Pharmacopa, Atamet, and Stalevo. As a drug, it is used in the clinical treatment of Parkinson's disease and dopamine-responsive dystonia.

Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl and Emsam among others, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder. It is provided in the form of a capsule or tablet taken by mouth for Parkinson's disease and as a patch applied to skin for depression.

Cabergoline, sold under the brand name Dostinex among others, is a dopaminergic medication used in the treatment of high prolactin levels, prolactinomas, Parkinson's disease, and for other indications. It is taken by mouth.

Dopaminergic means "related to dopamine" (literally, "working on dopamine"), dopamine being a common neurotransmitter. Dopaminergic substances or actions increase dopamine-related activity in the brain. Dopaminergic brain pathways facilitate dopamine-related activity. For example, certain proteins such as the dopamine transporter (DAT), vesicular monoamine transporter 2 (VMAT2), and dopamine receptors can be classified as dopaminergic, and neurons that synthesize or contain dopamine and synapses with dopamine receptors in them may also be labeled as dopaminergic. Enzymes that regulate the biosynthesis or metabolism of dopamine such as aromatic L-amino acid decarboxylase or DOPA decarboxylase, monoamine oxidase (MAO), and catechol O-methyl transferase (COMT) may be referred to as dopaminergic as well. Also, any endogenous or exogenous chemical substance that acts to affect dopamine receptors or dopamine release through indirect actions (for example, on neurons that synapse onto neurons that release dopamine or express dopamine receptors) can also be said to have dopaminergic effects, two prominent examples being opioids, which enhance dopamine release indirectly in the reward pathways, and some substituted amphetamines, which enhance dopamine release directly by binding to and inhibiting VMAT2.

Entacapone, sold under the brand name Comtan among others, is a medication commonly used in combination with other medications for the treatment of Parkinson's disease. Entacapone together with levodopa and carbidopa allows levodopa to have a longer effect in the brain and reduces Parkinson's disease signs and symptoms for a greater length of time than levodopa and carbidopa therapy alone.
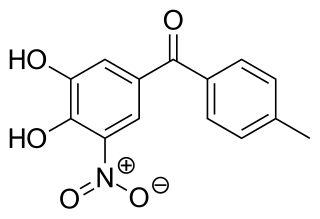
Tolcapone, sold under the brand name Tasmar, is a medication used to treat Parkinson's disease (PD). It is a selective, potent and reversible nitrocatechol-type inhibitor of the enzyme catechol-O-methyltransferase (COMT). It has demonstrated significant liver toxicity, which has led to suspension of marketing authorisations in a number of countries.

A dopamine agonist(DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D2-like and D1-like, and they are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the D2-like family includes D2, D3 and D4 receptors. Dopamine agonists are primarily used in the treatment of Parkinson's disease, and to a lesser extent, in hyperprolactinemia and restless legs syndrome. They are also used off-label in the treatment of clinical depression. The use of dopamine agonists is associated with impulse control disorders and dopamine agonist withdrawal syndrome (DAWS).
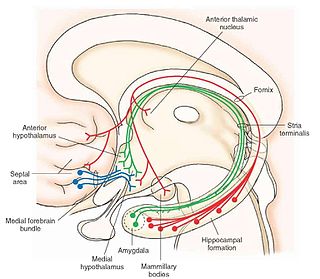
The medial forebrain bundle (MFB), is a neural pathway containing fibers from the basal olfactory regions, the periamygdaloid region and the septal nuclei, as well as fibers from brainstem regions, including the ventral tegmental area and nigrostriatal pathway.

Dihydroergocryptine (DHEC), sold under the brand names Almirid and Cripar among others, is a dopamine agonist of the ergoline group that is used as an antiparkinson agent in the treatment of Parkinson's disease. It is taken by mouth.
Mesocarb is a drug that is currently being developed for Parkinson's disease.

Bornaprine is a synthetic anticholinergic medication that is primarily used to treat Parkinson's disease. Additionally, bornaprine has been used to treat other disorders, including hyperhidrosis.

Tesofensine (NS2330) is a serotonin–noradrenaline–dopamine reuptake inhibitor from the phenyltropane family of drugs, which is being developed for the treatment of obesity. Tesofensine was originally developed by a Danish biotechnology company, NeuroSearch, who transferred the rights to Saniona in 2014.

Dimethocaine, also known as DMC or larocaine, is a compound with a stimulatory effect. This effect resembles that of cocaine, although dimethocaine appears to be less potent. Just like cocaine, dimethocaine is addictive due to its stimulation of the reward pathway in the brain. However, dimethocaine is a legal cocaine replacement in some countries and is even listed by the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) under the category “synthetic cocaine derivatives”. The structure of dimethocaine, being a 4-aminobenzoic acid ester, resembles that of procaine. It is found as a white powder at room temperature.

Remacemide is a drug which acts as a low-affinity NMDA antagonist with sodium channel blocking properties. It has been studied for the treatment of acute ischemic stroke, epilepsy, Huntington's disease, and Parkinson's disease.

Safinamide is a drug used as an add-on treatment for Parkinson's disease with "off" episodes; it has multiple modes of action, including the inhibition of monoamine oxidase B.

BTS 74,398 is a centrally acting stimulant drug which was developed for the treatment of Parkinson's disease. It inhibits the synaptic reuptake of dopamine, serotonin and noradrenaline, making it a triple reuptake inhibitor. It was effective in animal models of Parkinson's disease, but was unsuccessful in human trials.

Norsalsolinol is a chemical compound that is produced naturally in the body through metabolism of dopamine. It has been shown to be a selective dopaminergic neurotoxin, and has been suggested as a possible cause of neurodegenerative conditions such as Parkinson's disease and the brain damage associated with alcoholism, although evidence for a causal relationship is unclear.

UWA-101 is a phenethylamine derivative invented by Dr Matthew Piggott at the University of Western Australia, and researched as a potential treatment for Parkinson's disease. Its chemical structure is very similar to that of the illegal drug MDMA, the only difference being the replacement of the α-methyl group with an α-cyclopropyl group. MDMA has been found in animal studies and reported in unauthorised human self-experiments to be effective in the short-term relief of side-effects of Parkinson's disease therapy, most notably levodopa-induced dyskinesia. However the illegal status of MDMA and concerns about its potential for recreational use, neurotoxicity and potentially dangerous side effects mean that it is unlikely to be investigated for medical use in this application, and so alternative analogues were investigated.



















