
Triazolam, sold under the brand name Halcion among others, is a central nervous system (CNS) depressant tranquilizer of the triazolobenzodiazepine (TBZD) class, which are benzodiazepine (BZD) derivatives. It possesses pharmacological properties similar to those of other benzodiazepines, but it is generally only used as a sedative to treat severe insomnia. In addition to the hypnotic properties, triazolam's amnesic, anxiolytic, sedative, anticonvulsant, and muscle relaxant properties are pronounced as well.
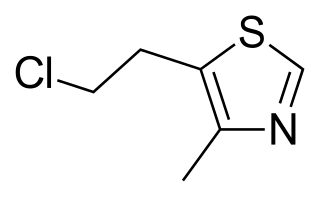
Clomethiazole is a sedative and hypnotic originally developed by Hoffmann-La Roche in the 1930s. The drug is used in treating and preventing symptoms of acute alcohol withdrawal.

Beclamide is a drug that possesses anticonvulsant activity. It is no longer used.

Camazepam is a benzodiazepine psychoactive drug, marketed under the brand names Albego, Limpidon and Paxor. It is the dimethyl carbamate ester of temazepam, a metabolite of diazepam. While it possesses anxiolytic, anticonvulsant, skeletal muscle relaxant and hypnotic properties it differs from other benzodiazepines in that its anxiolytic properties are particularly prominent but has comparatively limited anticonvulsant, hypnotic and skeletal muscle relaxant properties.
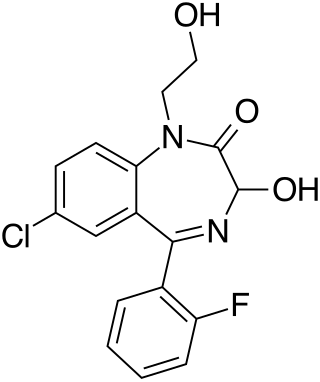
Doxefazepam is a benzodiazepine medication It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties. It is used therapeutically as a hypnotic. According to Babbini and colleagues in 1975, this derivative of flurazepam was between 2 and 4 times more potent than the latter while at the same time being half as toxic in laboratory animals.

Tricyclics are cyclic chemical compounds that contain three fused rings of atoms.

Hexethal (Ortal) is a barbiturate derivative invented in the 1940s. It has sedative, anxiolytic, muscle relaxant, and anticonvulsant properties, and was used primarily as an anaesthetic in veterinary medicine.
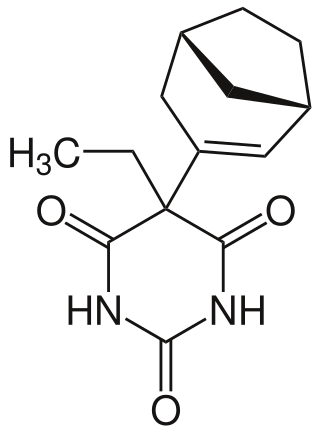
Reposal is a barbiturate derivative invented in the 1960s in Denmark. It has sedative, hypnotic and anticonvulsant properties, and was used primarily for the treatment of insomnia.
Thialbarbital (Intranarcon) is a barbiturate derivative invented in the 1960s. It has sedative effects, and was used primarily for induction in surgical anaesthesia. Thialbarbital is short acting and has less of a tendency to induce respiratory depression than other barbiturate derivatives such as pentobarbital.

Methylpentynol is a tertiary hexynol with hypnotic/sedative and anticonvulsant effects and an exceptionally low therapeutic index. It was discovered by Bayer in 1913 and was used shortly thereafter for the treatment of insomnia, but its use was quickly phased out in response to newer drugs with far more favorable safety profiles.

Imidazenil is an experimental anxiolytic drug which is derived from the benzodiazepine family, and is most closely related to other imidazobenzodiazepines such as midazolam, flumazenil, and bretazenil.

QH-II-66 (QH-ii-066) is a sedative drug which is a benzodiazepine derivative. It produces some of the same effects as other benzodiazepines, but is much more selective than most other drugs of this class and so produces somewhat less sedation and ataxia than other related drugs such as diazepam and triazolam, although it still retains anticonvulsant effects.

CL-218,872 is a sedative and hypnotic drug used in scientific research. It has similar effects to sedative-hypnotic benzodiazepine drugs such as triazolam, but is structurally distinct and so is classed as a nonbenzodiazepine hypnotic.

Y-23684 is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.
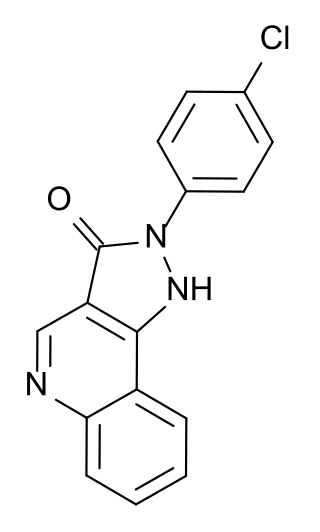
CGS-9896 is an anxiolytic drug used in scientific research. It has similar effects to benzodiazepine drugs but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.

Wogonin is an O-methylated flavone, a flavonoid-like chemical compound which is found in Scutellaria baicalensis.
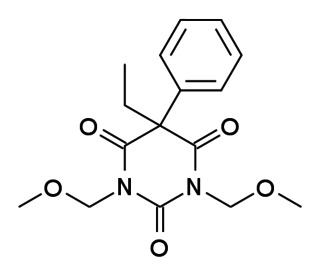
Eterobarb (Antilon) is a barbiturate derivative. It has mainly anticonvulsant action with less sedative effects than the closely related compound phenobarbital. It saw reasonable success in clinical trials, but is not in widespread medical use.

HA-966 or (±) 3-Amino-1-hydroxy-pyrrolidin-2-one is a molecule used in scientific research as a glycine receptor and NMDA receptor antagonist / low efficacy partial agonist. It has neuroprotective and anticonvulsant, anxiolytic, antinociceptive and sedative / hypnotic effects in animal models. Pilot human clinical trials in the early 1960s showed that HA-966 appeared to benefit patients with tremors of extrapyramidal origin.
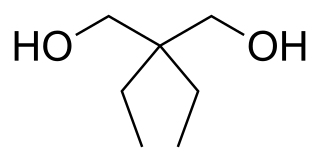
Prenderol (Diethylpropanediol) is a simple alkyl diol which has sedative, anticonvulsant and muscle relaxant effects. It is closely related in structure to meprobamate and numerous other alkyl alcohols and diols with generally comparable activity.
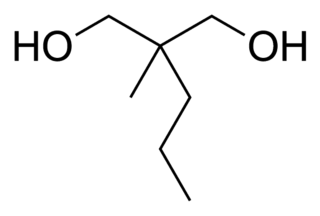
2-Methyl-2-propyl-1,3-propanediol (MPP) is a simple alkyl diol which has sedative, anticonvulsant and muscle relaxant effects. It is both a synthetic precursor to, and an active metabolite of the tranquilizers meprobamate and carisoprodol, as well as other derivatives.



















