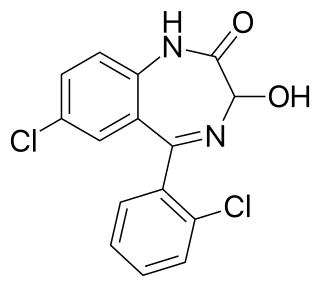
Benzodiazepines, sometimes called "benzos", are a class of psychoactive drugs whose core chemical structure is the fusion of a benzene ring and a diazepine ring. The first such drug, chlordiazepoxide (Librium), was discovered accidentally by Leo Sternbach in 1955, and made available in 1960 by Hoffmann–La Roche, which, since 1963, has also marketed the benzodiazepine diazepam (Valium). In 1977 benzodiazepines were globally the most prescribed medications. They are in the family of drugs commonly known as minor tranquilizers.

Hypnotic, or soporific drugs, commonly known as sleeping pills, are a class of psychoactive drugs whose primary function is to induce sleep and for the treatment of insomnia (sleeplessness), or for surgical anesthesia.

Nonbenzodiazepines (,) sometimes referred to colloquially as Z-drugs, are a class of psychoactive drugs that are very benzodiazepine-like in nature. They are used in the treatment of sleep problems.

Alpidem (Ananxyl) is an anxiolytic drug from the imidazopyridine family, related to the more well known sleeping medication zolpidem. Unlike zolpidem however, alpidem does not produce sedative effects at normal doses, and is instead used specifically for the treatment of anxiety.

Pagoclone is an anxiolytic agent from the cyclopyrrolone family, related to better-known drugs such as the sleeping medication zopiclone. It was synthesized by a French team working for Rhone-Poulenc & Rorer S.A. Pagoclone belongs to the class of nonbenzodiazepines, which have similar effects to the older benzodiazepine group, but with quite different chemical structures. It was never commercialised.
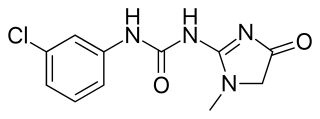
Fenobam is an imidazole derivative developed by McNeil Laboratories in the late 1970s as a novel anxiolytic drug with an at-the-time-unidentified molecular target in the brain. Subsequently, it was determined that fenobam acts as a potent and selective negative allosteric modulator of the metabotropic glutamate receptor subtype mGluR5, and it has been used as a lead compound for the development of a range of newer mGluR5 antagonists.

Etifoxine is an anxiolytic and anticonvulsant drug developed by Hoechst in the 1960s. It is sold in approximately 40 countries for anxiety disorders, without the sedation and ataxia associated with benzodiazepine drugs. It has similar anxiolytic effects to benzodiazepine drugs, but is structurally distinct, although it has structural elements in common with them. Studies suggest is as effective as lorazepam as an anxiolytic, but has fewer side effects. Etifoxine is not approved by the U.S. Food and Drug Administration or the European Medicines Agency.

Panadiplon (U-78875) is an anxiolytic drug with a novel chemical structure that is not closely related to other drugs of this type. It has a similar pharmacological profile to the benzodiazepine family of drugs, but with mainly anxiolytic properties and relatively little sedative or amnestic effect, and so is classified as a nonbenzodiazepine anxiolytic.

Pazinaclone (DN-2327) is a sedative and anxiolytic drug in the cyclopyrrolone family of drugs. Some other cyclopyrrolone drugs include zopiclone and eszopiclone.
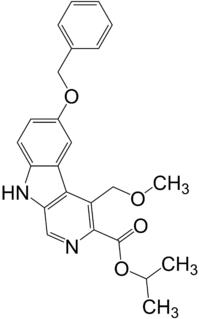
Abecarnil (ZK-112,119) is an anxiolytic drug from the β-Carboline family. It is one of a relatively recently developed class of medicines known as the nonbenzodiazepines, which have similar effects to the older benzodiazepine group, but with quite different chemical structures. It is a partial agonist acting selectively at the benzodiazepine site of the GABAA receptor.

Pipequaline (INN) is an anxiolytic drug that was never marketed. It possesses a novel chemical structure that is not closely related to other drugs of this type. The drug has a similar pharmacological profile to the benzodiazepine family of drugs, but with mainly anxiolytic properties and very little sedative, amnestic or anticonvulsant effects, and so is classified as a nonbenzodiazepine anxiolytic.

L-838,417 is an anxiolytic drug used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. The compound was developed by Merck, Sharp and Dohme.

SL651498 is an anxiolytic and anticonvulsant drug used in scientific research, with a chemical structure most closely related to β-carboline derivatives such as abecarnil and gedocarnil. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.

Y-23684 is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.
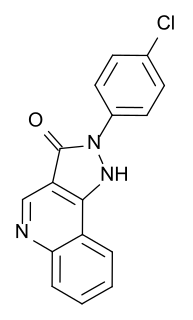
CGS-9896 is an anxiolytic drug used in scientific research. It has similar effects to benzodiazepine drugs but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.

CGS-20625 is an anxiolytic drug used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. It produces anxiolytic and anticonvulsant effects, but with no sedative effects even at high doses, and no significant muscle relaxant effects. It is orally active in humans, but with relatively low bioavailability.

Tracazolate (ICI-136,753) is an anxiolytic drug which is used in scientific research. It is a pyrazolopyridine derivative, most closely related to pyrazolopyrimidine drugs such as zaleplon, and is one of a structurally diverse group of drugs known as the nonbenzodiazepines which act at the same receptor targets as benzodiazepines but have distinct chemical structures.

ELB-139 (LS-191,811) is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic.

TPA-023 (MK-0777) is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. It is a subtype-selective, mixed agonist-antagonist at GABAA receptors, which acts as a partial agonist at the α2 and α3 subtypes, but as a silent antagonist at α1 and α5 subtypes. It has primarily anxiolytic and anticonvulsant effects in animal tests, but with no sedative effects even at 50 times the effective anxiolytic dose.

TP-13 is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. It is a subtype-selective partial agonist at GABAA receptors, binding selectively to GABAA receptor complexes bearing α2 and α3 subunits. It has modest anticonvulsant activity although less than that of diazepam, and its main effect is likely to be selective anxiolytic action, as seen with other related α2/3-preferring agonists such as L-838,417.




















