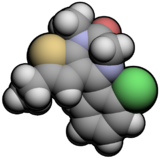
Diazepam, first marketed as Valium, is a medicine of the benzodiazepine family that acts as an anxiolytic. It is commonly used to treat a range of conditions, including anxiety, seizures, alcohol withdrawal syndrome, muscle spasms, insomnia, and restless legs syndrome. It may also be used to cause memory loss during certain medical procedures. It can be taken orally, as a suppository inserted into the rectum, intramuscularly, intravenously or used as a nasal spray. When injected intravenously, effects begin in one to five minutes and last up to an hour. Orally, effects begin after 15 to 60 minutes.

Alprazolam, sold under the brand name Xanax, is a fast-acting, potent tranquilizer of moderate duration within the triazolobenzodiazepine group of chemicals called benzodiazepines. Alprazolam is most commonly used in management of anxiety disorders, specifically panic disorder or generalized anxiety disorder (GAD). Other uses include the treatment of chemotherapy-induced nausea, together with other treatments. GAD improvement occurs generally within a week. Alprazolam is generally taken orally.
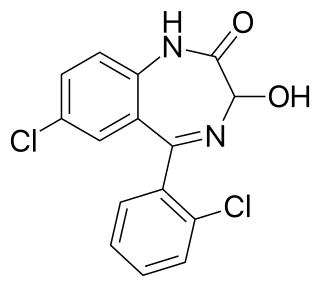
Lorazepam, sold under the brand name Ativan among others, is a benzodiazepine medication. It is used to treat anxiety disorders, trouble sleeping, severe agitation, active seizures including status epilepticus, alcohol withdrawal, and chemotherapy-induced nausea and vomiting. It is also used during surgery to interfere with memory formation and to sedate those who are being mechanically ventilated. It is also used, along with other treatments, for acute coronary syndrome due to cocaine use. It can be given by mouth or as an injection into a muscle or vein. When given by injection, onset of effects is between one and thirty minutes and effects last for up to a day.

Zolpidem, sold under the brand name Ambien among others, is a medication primarily used for the short-term treatment of sleeping problems. Guidelines recommend that it be used only after cognitive behavioral therapy for insomnia and behavioral changes, such as sleep hygiene, have been tried. It decreases the time to sleep onset by about fifteen minutes and at larger doses helps people stay asleep longer. It is taken by mouth and is available in conventional tablets, sublingual tablets, or oral spray.

Clonazepam, sold under the brand names Klonopin and Rivotril, is a medication used to prevent and treat anxiety disorders, seizures, bipolar mania, agitation associated with psychosis, OCD and akathisia. It is a tranquilizer of the benzodiazepine class. It possesses anxiolytic, anticonvulsant, sedative, hypnotic, and skeletal muscle relaxant properties. It is typically taken by mouth. Effects begin within one hour and last between six and twelve hours.

Zopiclone, sold under the brand name Imovane among others, is a nonbenzodiazepine used to treat difficulty sleeping. Zopiclone is molecularly distinct from benzodiazepine drugs and is classed as a cyclopyrrolone. However, zopiclone increases the normal transmission of the neurotransmitter gamma-aminobutyric acid (GABA) in the central nervous system, via modulating GABAA receptors similarly to the way benzodiazepine drugs do.

Nitrazepam, sold under the brand name Mogadon among others, is a hypnotic drug of the benzodiazepine class used for short-term relief from severe, disabling anxiety and insomnia. It also has sedative (calming) properties, as well as amnestic, anticonvulsant, and skeletal muscle relaxant effects.

Bromazepam, sold under many brand names, is a benzodiazepine. It is mainly an anti-anxiety agent with similar side effects to diazepam (Valium). In addition to being used to treat anxiety or panic states, bromazepam may be used as a premedicant prior to minor surgery. Bromazepam typically comes in doses of 3 mg and 6 mg tablets.

Oxazepam is a short-to-intermediate-acting benzodiazepine. Oxazepam is used for the treatment of anxiety and insomnia and in the control of symptoms of alcohol withdrawal syndrome.

Quazepam, sold under brand name Doral among others, is a relatively long-acting benzodiazepine derivative drug developed by the Schering Corporation in the 1970s. Quazepam is used for the treatment of insomnia including sleep induction and sleep maintenance. Quazepam induces impairment of motor function and has relatively selective hypnotic and anticonvulsant properties with considerably less overdose potential than other benzodiazepines. Quazepam is an effective hypnotic which induces and maintains sleep without disruption of the sleep architecture.

Clorazepate, sold under the brand name Tranxene among others, is a benzodiazepine medication. It possesses anxiolytic, anticonvulsant, sedative, hypnotic, and skeletal muscle relaxant properties. Clorazepate is an unusually long-lasting benzodiazepine and serves as a majoritive prodrug for the equally long-lasting desmethyldiazepam, which is rapidly produced as an active metabolite. Desmethyldiazepam is responsible for most of the therapeutic effects of clorazepate.

Loprazolam (triazulenone) marketed under many brand names is a benzodiazepine medication. It possesses anxiolytic, anticonvulsant, hypnotic, sedative and skeletal muscle relaxant properties. It is licensed and marketed for the short-term treatment of moderately-severe insomnia.

Prazepam is a benzodiazepine derivative drug developed by Warner-Lambert in the 1960s. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties. Prazepam is a prodrug for desmethyldiazepam which is responsible for the therapeutic effects of prazepam.

Etizolam is a thienodiazepine derivative which is a benzodiazepine analog. The etizolam molecule differs from a benzodiazepine in that the benzene ring has been replaced by a thiophene ring and triazole ring has been fused, making the drug a thienotriazolodiazepine.
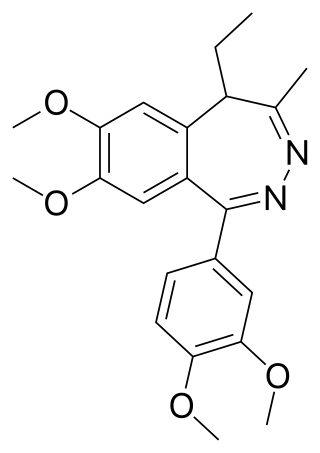
Tofisopam is an anxiolytic that is marketed in several European countries. Chemically, it is a 2,3-benzodiazepine. Unlike other anxiolytic benzodiazepines however, tofisopam does not have anticonvulsant, sedative, skeletal muscle relaxant, motor skill-impairing or amnestic properties. While it may not be an anticonvulsant in and of itself, it has been shown to enhance the anticonvulsant action of classical 1,4-benzodiazepines and muscimol, but not sodium valproate, carbamazepine, phenobarbital, or phenytoin. Tofisopam is indicated for the treatment of anxiety and alcohol withdrawal, and is prescribed in a dosage of 50–300 mg per day divided into three doses. Peak plasma levels are attained two hours after an oral dose. Tofisopam is not reported as causing dependence to the same extent as other benzodiazepines, but is still recommended to be prescribed for a maximum of 12 weeks.

Lormetazepam, sold under the brand name Noctamid among others, is a drug which is a short to intermediate acting 3-hydroxy benzodiazepine derivative and temazepam analogue. It possesses hypnotic, anxiolytic, anticonvulsant, sedative, and skeletal muscle relaxant properties.
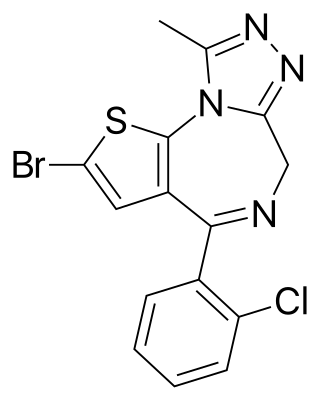
Brotizolam is a sedative-hypnotic thienotriazolodiazepine drug which is a benzodiazepine analog. It possesses anxiolytic, anticonvulsant, hypnotic, sedative and skeletal muscle relaxant properties, and is considered to be similar in effect to other short-acting hypnotic benzodiazepines such as triazolam or midazolam. It is used in the short-term treatment of severe insomnia. Brotizolam is a highly potent and short-acting hypnotic, with a typical dose ranging from 0.125 to 0.25 milligrams, which is rapidly eliminated with an average half-life of 4.4 hours.

Delorazepam, also known as chlordesmethyldiazepam and nordiclazepam, is a drug which is a benzodiazepine and a derivative of desmethyldiazepam. It is marketed in Italy, where it is available under the trade name EN and Dadumir. Delorazepam (chlordesmethyldiazepam) is also an active metabolite of the benzodiazepine drugs diclazepam and cloxazolam. Adverse effects may include hangover type effects, drowsiness, behavioural impairments and short-term memory impairments. Similar to other benzodiazepines delorazepam has anxiolytic, skeletal muscle relaxant, hypnotic and anticonvulsant properties.
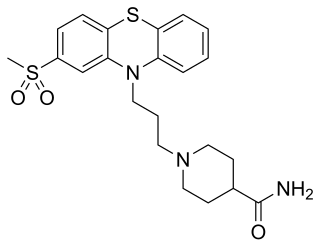
Metopimazine, sold under the brand names Vogalen and Vogalene, is an antiemetic of the phenothiazine group which is used to treat nausea and vomiting. It is marketed in Europe, Canada, and South America. As of August 2020, metopimazine has been repurposed and is additionally under development for use in the United States for the treatment of gastroparesis.
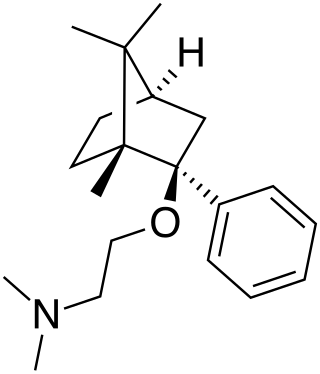
Deramciclane (EGIS-3886) is a non-benzodiazepine-type anxiolytic drug to treat various types of anxiety disorders. Deramciclane is a unique alternative to current anxiolytics on the market because it has a novel chemical structure and target. It acts as an antagonist at the 5-HT2A receptor, as an inverse agonist at the 5-HT2C receptor, and as a GABA reuptake inhibitor. The two serotonin receptors are G protein-coupled receptors and are two of the main excitatory serotonin receptor types. Their excitation has been implicated in anxiety and mood. Deramciclane does not affect CYP3A4 activity in metabolizing other drugs, but it is a weak inhibitor of CYP2D6. Some studies also show the drug to have moderate affinity to dopamine D2 receptors and low affinity to dopamine receptor D1. Researchers are looking for alternatives to benzodiazepines for anxiolytic use because benzodiazepine drugs have sedative and muscle relaxant side effects.