An anxiolytic is a medication or other intervention that reduces anxiety. This effect is in contrast to anxiogenic agents which increase anxiety. Anxiolytic medications are used for the treatment of anxiety disorders and their related psychological and physical symptoms.

GABA is the chief inhibitory neurotransmitter in the developmentally mature mammalian central nervous system. Its principal role is reducing neuronal excitability throughout the nervous system.

Phenelzine, sold under the brand name Nardil, among others, is a non-selective and irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine family which is primarily used as an antidepressant and anxiolytic to treat depression and anxiety. Along with tranylcypromine and isocarboxazid, phenelzine is one of the few non-selective and irreversible MAOIs still in widespread clinical use.

Tiagabine is an anticonvulsant medication produced by Cephalon that is used in the treatment of epilepsy. The drug is also used off-label in the treatment of anxiety disorders and panic disorder.
Cross-tolerance is a phenomenon that occurs when tolerance to the effects of a certain drug produces tolerance to another drug. It often happens between two drugs with similar functions or effects—for example, acting on the same cell receptor or affecting the transmission of certain neurotransmitters. Cross-tolerance has been observed with pharmaceutical drugs such as anti-anxiety agents and illicit substances, and sometimes the two of them together. Often, a person who uses one drug can be tolerant to a drug that has a completely different function. This phenomenon allows one to become tolerant to a drug that they have never used before.
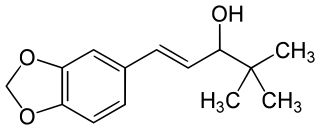
Stiripentol, sold under the brand name Diacomit, is an anticonvulsant medication used for the treatment of Dravet syndrome - a serious genetic brain disorder.

Succinic semialdehyde dehydrogenase deficiency (SSADHD) is a rare autosomal recessive disorder of the degradation pathway of the inhibitory neurotransmitter γ-aminobutyric acid, or GABA. The disorder has been identified in approximately 350 families, with a significant proportion being consanguineous families. The first case was identified in 1981 and published in a Dutch clinical chemistry journal that highlighted a number of neurological conditions such as delayed intellectual, motor, speech, and language as the most common manifestations. Later cases reported in the early 1990s began to show that hypotonia, hyporeflexia, seizures, and a nonprogressive ataxia were frequent clinical features as well.
Neurotransmitter transporters are a class of membrane transport proteins that span the cellular membranes of neurons. Their primary function is to carry neurotransmitters across these membranes and to direct their further transport to specific intracellular locations. There are more than twenty types of neurotransmitter transporters.

γ-Amino-β-hydroxybutyric acid (GABOB), also known as β-hydroxy-γ-aminobutyric acid (β-hydroxy-GABA), and sold under the brand name Gamibetal among others, is an anticonvulsant which is used for the treatment of epilepsy in Europe, Japan, and Mexico. It is a GABA analogue, or an analogue of the neurotransmitter γ-aminobutyric acid (GABA), and has been found to be an endogenous metabolite of GABA.
In enzymology, a diaminobutyrate acetyltransferase (EC 2.3.1.178) is an enzyme that catalyzes the chemical reaction
Reuptake inhibitors (RIs) are a type of reuptake modulators. It is a drug that inhibits the plasmalemmal transporter-mediated reuptake of a neurotransmitter from the synapse into the pre-synaptic neuron. This leads to an increase in extracellular concentrations of the neurotransmitter and an increase in neurotransmission. Various drugs exert their psychological and physiological effects through reuptake inhibition, including many antidepressants and psychostimulants.

Gabaculine is a naturally occurring neurotoxin first isolated from the bacteria Streptomyces toyacaensis, which acts as a potent and irreversible GABA transaminase inhibitor, and also a GABA reuptake inhibitor. Gabaculine is also known as 3-amino-2,3-dihydrobenzoic acid hydrochloride and 5-amino cyclohexa-1,3 dienyl carboxylic acid. Gabaculine increased GABA levels in the brain and had an effect on convulsivity in mice.
GABA transporters are a family of neurotransmitter / sodium symporters, belonging to the solute carrier 6 (SLC6) family. They are found in various regions of the brain in different cell types, such as neurons and astrocytes.

A GABA reuptake inhibitor (GRI) is a type of drug which acts as a reuptake inhibitor for the neurotransmitter gamma-Aminobutyric acid (GABA) by blocking the action of the gamma-Aminobutyric acid transporters (GATs). This in turn leads to increased extracellular concentrations of GABA and therefore an increase in GABAergic neurotransmission. Gamma-aminobutyric acid (GABA) is an amino acid that functions as the predominant inhibitory neurotransmitter within the central nervous system, playing a crucial role in modulating neuronal activity in both the brain and spinal cord. While GABA predominantly exerts inhibitory actions in the adult brain, it has an excitatory role during developmental stages. When the neuron receives the action potential, GABA is released from the pre-synaptic cell to the synaptic cleft. After the action potential transmission, GABA is detected on the dendritic side, where specific receptors collectively contribute to the inhibitory outcome by facilitating GABA transmitter uptake. Facilitated by specific enzymes, GABA binds to post-synaptic receptors, with GABAergic neurons playing a key role in system regulation. The inhibitory effects of GABA diminish when presynaptic neurons reabsorb it from the synaptic cleft for recycling by GABA transporters (GATs). The reuptake mechanism is crucial for maintaining neurotransmitter levels and synaptic functioning. Gamma-aminobutyric acid Reuptake Inhibitors (GRIs) hinder the functioning of GATs, preventing GABA reabsorption in the pre-synaptic cell. This results in increased GABA levels in the extracellular environment, leading to elevated GABA-mediated synaptic activity in the brain.
In pharmacology, a GABA transaminase inhibitor is an enzyme inhibitor that acts upon GABA transaminase. Inhibition of GABA transaminase enzymes reduces the degradation of GABA, leading to increased neuronal GABA concentrations.
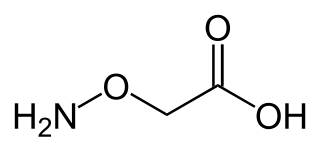
Aminooxyacetic acid, often abbreviated AOA or AOAA, is a compound that inhibits 4-aminobutyrate aminotransferase (GABA-T) activity in vitro and in vivo, leading to less gamma-aminobutyric acid (GABA) being broken down. Subsequently, the level of GABA is increased in tissues. At concentrations high enough to fully inhibit 4-aminobutyrate aminotransferase activity, aminooxyacetic acid is indicated as a useful tool to study regional GABA turnover in rats.

CI-966 (developmental code name) is a central nervous system depressant acting as a GABA reuptake inhibitor, specifically a highly potent and selective blocker of the GABA transporter 1 (GAT-1) (IC50 = 0.26 μM), and hence indirect and non-selective GABA receptor full agonist. It was investigated as a potential anticonvulsant, anxiolytic, and neuroprotective therapeutic but was discontinued during clinical development due to the incidence of severe adverse effects at higher doses and hence was never marketed.
An excitatory amino acid reuptake inhibitor (EAARI) is a type of drug which inhibits the reuptake of the excitatory neurotransmitters glutamate and aspartate by blocking one or more of the excitatory amino acid transporters (EAATs).
A neurosteroidogenesis inhibitor is a drug that inhibits the production of endogenous neurosteroids. Neurosteroids include the excitatory neurosteroids pregnenolone sulfate, dehydroepiandrosterone (DHEA), and dehydroepiandrosterone sulfate (DHEA-S), and the inhibitory neurosteroids allopregnanolone, tetrahydrodeoxycorticosterone (THDOC), and 3α-androstanediol, among others. By inhibiting the synthesis of endogenous neurosteroids, neurosteroidogenesis inhibitors have effects in the central nervous system.

A GABA analogue is a compound which is an analogue or derivative of the neurotransmitter gamma-Aminobutyric acid (GABA).












