Carboxy-lyases, also known as decarboxylases, are carbon–carbon lyases that add or remove a carboxyl group from organic compounds. These enzymes catalyze the decarboxylation of amino acids, beta-keto acids and alpha-keto acids.

Phosphopentose epimerase encoded by the RPE gene is a metalloprotein that catalyzes the interconversion between D-ribulose 5-phosphate and D-xylulose 5-phosphate.
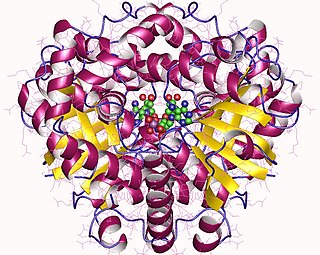
In enzymology, a L-gulonate 3-dehydrogenase (EC 1.1.1.45) is an enzyme that catalyzes the chemical reaction

In enzymology, a 3-dehydro-L-gulonate 2-dehydrogenase (EC 1.1.1.130) is an enzyme that catalyzes the chemical reaction:
In enzymology, a L-ribulose-5-phosphate 3-epimerase is an enzyme that catalyzes the chemical reaction
The enzyme 2-dehydro-3-deoxy-6-phosphogalactonate aldolase catalyzes the chemical reaction

The enzyme 2-dehydro-3-deoxyglucarate aldolase catalyzes the chemical reaction

In enzymology, an aspartate 4-decarboxylase (EC 4.1.1.12) is an enzyme that catalyzes the chemical reaction
The enzyme branched-chain-2-oxoacid decarboxylase (EC 4.1.1.72) catalyzes the chemical reaction
The enzyme dehydro-L-gulonate decarboxylase (EC 4.1.1.34) catalyzes the chemical reaction
The enzyme hydroxyglutamate decarboxylase (EC 4.1.1.16) catalyzes the chemical reaction

The enzyme phenylalanine decarboxylase (EC 4.1.1.53) catalyzes the chemical reaction
The enzyme phosphopantothenoylcysteine decarboxylase (EC 4.1.1.36) catalyzes the chemical reaction
The enzyme threonine-phosphate decarboxylase (EC 4.1.1.81) catalyzes the chemical reaction
The enzyme tyrosine decarboxylase (EC 4.1.1.25) catalyzes the chemical reaction
In enzymology, a valine decarboxylase (EC 4.1.1.14) is an enzyme that catalyzes the chemical reaction
The enzyme 2-dehydro-3-deoxy-L-arabinonate dehydratase (EC 4.2.1.43) catalyzes the chemical reaction
The enzyme L-arabinonate dehydratase (EC 4.2.1.25) catalyzes the chemical reaction
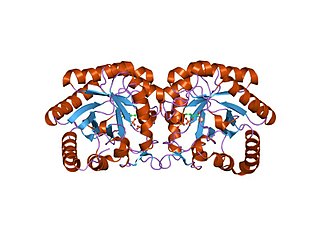
3-Deoxy-D-arabinoheptulosonate 7-phosphate (DAHP) synthase is the first enzyme in a series of metabolic reactions known as the shikimate pathway, which is responsible for the biosynthesis of the amino acids phenylalanine, tyrosine, and tryptophan. Since it is the first enzyme in the shikimate pathway, it controls the amount of carbon entering the pathway. Enzyme inhibition is the primary method of regulating the amount of carbon entering the pathway. Forms of this enzyme differ between organisms, but can be considered DAHP synthase based upon the reaction that is catalyzed by this enzyme.
The PTS L-Ascorbate (L-Asc) Family includes porters specific for L-ascorbate, and is part of the PTS-AG superfamily. A single PTS permease of the L-Asc family of PTS permeases has been functionally characterized. This is the SgaTBA system, renamed UlaABC by Yew and Gerlt.







