
Alkaloids are a class of basic, naturally occurring organic compounds that contain at least one nitrogen atom. This group also includes some related compounds with neutral and even weakly acidic properties. Some synthetic compounds of similar structure may also be termed alkaloids. In addition to carbon, hydrogen and nitrogen, alkaloids may also contain oxygen or sulfur. More rarely still, they may contain elements such as phosphorus, chlorine, and bromine.

Caffeine is a central nervous system (CNS) stimulant of the methylxanthine class. It is mainly used recreationally, as a eugeroic (wakefulness promoter) or as a mild cognitive enhancer to increase alertness and attentional performance. Caffeine acts by blocking binding of adenosine to the adenosine A1 receptor, which enhances release of the neurotransmitter acetylcholine. Caffeine has a three-dimensional structure similar to that of adenosine, which allows it to bind and block its receptors. Caffeine also increases cyclic AMP levels through nonselective inhibition of phosphodiesterase.

Theophylline, also known as 1,3-dimethylxanthine, is a drug that inhibits phosphodiesterase and blocks adenosine receptors. It is used to treat chronic obstructive pulmonary disease (COPD) and asthma. Its pharmacology is similar to other methylxanthine drugs. Trace amounts of theophylline are naturally present in tea, coffee, chocolate, yerba maté, guarana, and cola.

Stimulants is an overarching term that covers many drugs including those that increase the activity of the central nervous system and the body, drugs that are pleasurable and invigorating, or drugs that have sympathomimetic effects. Stimulants are widely used throughout the world as prescription medicines as well as without a prescription as performance-enhancing or recreational drugs. Among narcotics, stimulants produce a noticeable crash or comedown at the end of their effects. The most frequently prescribed stimulants as of 2013 were lisdexamfetamine (Vyvanse), methylphenidate (Ritalin), and amphetamine (Adderall). It was estimated in 2015 that the percentage of the world population that had used cocaine during a year was 0.4%. For the category "amphetamines and prescription stimulants" the value was 0.7%, and for MDMA 0.4%.

Guaraná is a climbing plant in the family Sapindaceae, native to the Amazon basin and especially common in Brazil. Guaraná has large leaves and clusters of flowers, and is best known for the seeds from its fruits, which are about the size of a coffee bean.
Caffeinism is a state of intoxication caused by excessive consumption of caffeine. This intoxication covers a variety of unpleasant physical and mental symptoms associated with the consumption of excessive amounts of caffeine.
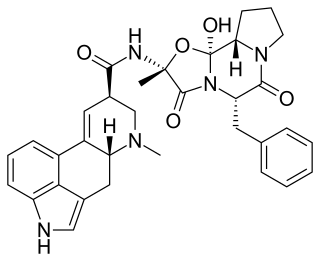
Ergotamine, sold under the brand names Cafergot and Ergomar among others, is an ergopeptine and part of the ergot family of alkaloids; it is structurally and biochemically closely related to ergoline. It possesses structural similarity to several neurotransmitters, and has biological activity as a vasoconstrictor.

Desipramine, sold under the brand name Norpramin among others, is a tricyclic antidepressant (TCA) used in the treatment of depression. It acts as a relatively selective norepinephrine reuptake inhibitor, though it does also have other activities such as weak serotonin reuptake inhibitory, α1-blocking, antihistamine, and anticholinergic effects. The drug is not considered a first-line treatment for depression since the introduction of selective serotonin reuptake inhibitor (SSRI) antidepressants, which have fewer side effects and are safer in overdose.

Metenolone enanthate, or methenolone enanthate, sold under the brand names Primobolan Depot and Nibal Injection, is an androgen and anabolic steroid (AAS) medication which is used mainly in the treatment of anemia due to bone marrow failure. It is given by injection into muscle. Although it was widely used in the past, the drug has mostly been discontinued and hence is now mostly only available on the black market. A related drug, metenolone acetate, is taken by mouth.
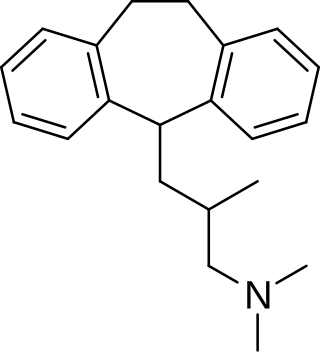
Butriptyline, sold under the brand name Evadyne among others, is a tricyclic antidepressant (TCA) that has been used in the United Kingdom and several other European countries for the treatment of depression but appears to no longer be marketed. Along with trimipramine, iprindole, and amoxapine, it has been described as an "atypical" or "second-generation" TCA due to its relatively late introduction and atypical pharmacology. It was very little-used compared to other TCAs, with the number of prescriptions dispensed only in the thousands.

Lynestrenol, sold under the brand names Exluton and Ministat among others, is a progestin medication which is used in birth control pills and in the treatment of gynecological disorders. The medication is available both alone and in combination with an estrogen. It is taken by mouth.
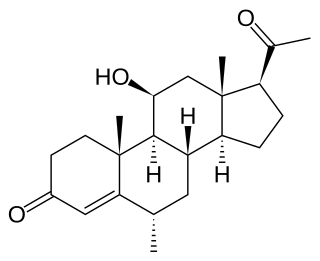
Medrysone, also known as hydroxymethylprogesterone, methylhydroxyprogesterone, or hydroxymesterone, as well as 6α-methyl-11β-hydroxyprogesterone or 6α-methyl-11β-hydroxypregn-4-ene-3,20-dione, is a synthetic glucocorticoid that is or has been used in the treatment of inflammatory eye diseases. It has been discontinued in the United States. Although it is very similar in structure to progesterone, neither progestogenic nor androgenic activity has been demonstrated for or attributed to medrysone.
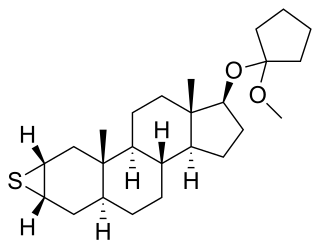
Mepitiostane, sold under the brand name Thioderon, is an orally active antiestrogen and anabolic–androgenic steroid (AAS) of the dihydrotestosterone (DHT) group which is marketed in Japan as an antineoplastic agent for the treatment of breast cancer. It is a prodrug of epitiostanol. The drug was patented and described in 1968.

Acecarbromal (INN), also known as acetylcarbromal and acetyladalin, is a hypnotic and sedative drug of the ureide (acylurea) group discovered by Bayer in 1917 that was formerly marketed in the United States and Europe. It is also used in combination with extract of quebracho and vitamin E as a treatment for erectile dysfunction under the brand name Afrodor in Europe. Acecarbromal is structurally related to the barbiturates, which are basically cyclized ureas. Prolonged use is not recommended as it can cause bromine poisoning.
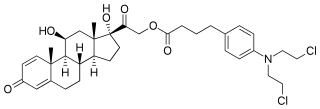
Prednimustine, sold under the brand names Mostarina and Sterecyst, is a medication which is used in chemotherapy in the treatment of leukemias and lymphomas. It is the ester formed from two other drugs, prednisolone and chlorambucil. Rarely, it has been associated with myoclonus.

Oxaflozane (INN) (brand name Conflictan) is an antidepressant and anxiolytic drug that was introduced by Solvay in France in 1982 for the treatment of depression but has since been discontinued. It is a prodrug of flumexadol (N-dealkyloxaflozane; 2-(3-trifluoromethylphenyl)morpholine; CERM-1841 or 1841-CERM), which is reported to act as an agonist of the serotonin 5-HT1A (pKi = 7.1) and 5-HT2C (pKi = 7.5) receptors and, to a much lesser extent, of the 5-HT2A (pKi = 6.0) receptor. In addition to its serotonergic properties, oxaflozane may also produce anticholinergic side effects at high doses, namely in overdose.

Paroxypropione, also known as paraoxypropiophenone, is a synthetic nonsteroidal estrogen which has been used medically as an antigonadotropin in Spain and Italy but appears to no longer be marketed. It was first synthesized in 1902. The antigonadotropic properties of the drug were discovered in 1951 and it entered clinical use shortly thereafter.
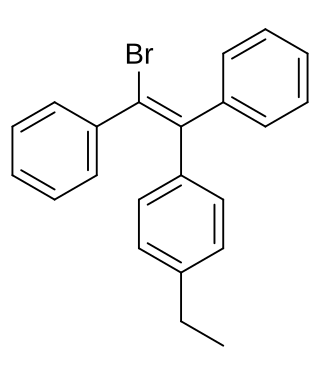
Broparestrol (INN), also known as α-bromo-α,β-diphenyl-β-p-ethylphenylethylene (BDPE), is a synthetic, nonsteroidal selective estrogen receptor modulator (SERM) of the triphenylethylene group that has been used in Europe as a dermatological agent and for the treatment of breast cancer. The drug is described as slightly estrogenic and potently antiestrogenic, and inhibits mammary gland development and suppresses prolactin levels in animals. It is structurally related to clomifene and diethylstilbestrol. Broparestrol is a mixture of E- and Z- isomers, both of which are active and are similarly antiestrogenic but, unlike broparestrol, were never marketed.
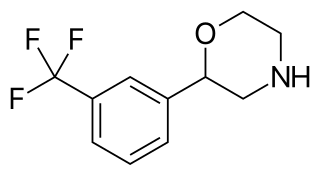
Flumexadol (INN) is a drug described and researched as a non-opioid analgesic which was never marketed. It has been found to act as an agonist of the serotonin 5-HT1A and 5-HT2C receptors and, to a much lesser extent, of the 5-HT2A receptor. According to Nilsson (2006) in a paper on 5-HT2C receptor agonists as potential anorectics, "The (+)-enantiomer of this compound showed [...] affinity for the 5-HT2C receptor (Ki) 25 nM) [...] and was 40-fold selective over the 5-HT2A receptor in receptor binding studies. Curiously, the racemic version [...], also known as 1841 CERM, was originally reported to possess analgesic properties while no association with 5-HT2C receptor activity was mentioned." It is implied that flumexadol might be employable as an anorectic in addition to analgesic. Though flumexadol itself has never been approved for medical use, oxaflozane is a prodrug of the compound that was formerly used clinically in France as an antidepressant and anxiolytic agent.

Metenolone acetate, or methenolone acetate, sold under the brand names Primobolan and Nibal, is an androgen and anabolic steroid (AAS) medication which is used mainly in the treatment of anemia due to bone marrow failure. It is taken by mouth. Although it was widely used in the past, the drug has mostly been discontinued and hence is now mostly no longer available. A related drug, metenolone enanthate, is given by injection into muscle.




















