| Polymer science |
|---|
 |

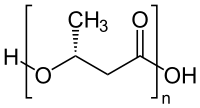
In polymer chemistry, condensation polymers are any kind of polymers whose process of polymerization involves a condensation reaction (i.e. a small molecule, such as water or methanol, is produced as a byproduct). Natural proteins as well as some common plastics such as nylon and PETE are formed in this way. Condensation polymers are formed by polycondensation, when the polymer is formed by condensation reactions between species of all degrees of polymerization, or by condensative chain polymerization, when the polymer is formed by sequential addition of monomers to an active site in a chain reaction. The main alternative forms of polymerization are chain polymerization and polyaddition, both of which give addition polymers.
Contents
Polycondensation: a polymerization in which the growth of polymer chains proceeds by condensation reactions between molecules of all degrees of polymerization. Notes:
- The growth steps are expressed by:
- Px + Py → Px+y + L (x, y = 1, 2, …)
- where Px and Py denote chains of degrees of polymerization x and y, respectively, and L a low-molar-mass by-product.
- The earlier term 'polycondensation' was synonymous with 'condensation polymerization'. The current definitions of polycondensation and condensative chain polymerization were both embraced by the earlier term 'polycondensation'. [1]
Condensation polymerization is a form of step-growth polymerization. Linear polymers are produced from bifunctional monomers, i.e. compounds with two reactive end-groups. Common condensation polymers include polyesters, polyamides such as nylon, polyacetals, and proteins. [2] [3]