Wilhelm Eduard Weber was a German physicist and, together with Carl Friedrich Gauss, inventor of the first electromagnetic telegraph.

The Physikalisch-Technische Bundesanstalt (PTB) is the national metrology institute of the Federal Republic of Germany, with scientific and technical service tasks. It is a higher federal authority and a public-law institution directly under federal government control, without legal capacity, under the auspices of the Federal Ministry for Economic Affairs and Climate Action.

Walther Wilhelm Georg Bothe was a German nuclear physicist known for the development of coincidence methods to study particle physics.

Max Theodor Felix von Laue was a German physicist who received the Nobel Prize in Physics in 1914 for his discovery of the diffraction of X-rays by crystals.
Walther Gerlach was a German physicist who co-discovered, through laboratory experiment, spin quantization in a magnetic field, the Stern–Gerlach effect. The experiment was conceived by Otto Stern in 1921 and successfully conducted first by Gerlach in early 1922.
Electrochemistry, a branch of chemistry, went through several changes during its evolution from early principles related to magnets in the early 16th and 17th centuries, to complex theories involving conductivity, electric charge and mathematical methods. The term electrochemistry was used to describe electrical phenomena in the late 19th and 20th centuries. In recent decades, electrochemistry has become an area of current research, including research in batteries and fuel cells, preventing corrosion of metals, the use of electrochemical cells to remove refractory organics and similar contaminants in wastewater electrocoagulation and improving techniques in refining chemicals with electrolysis and electrophoresis.
The molar conductivity of an electrolyte solution is defined as its conductivity divided by its molar concentration.
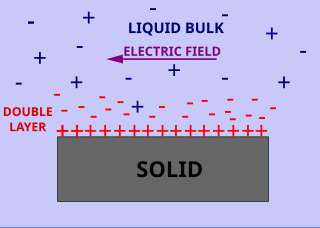
Surface conductivity is an additional conductivity of an electrolyte in the vicinity of the charged interfaces. Surface and volume conductivity of liquids correspond to the electrically driven motion of ions in an electric field. A layer of counter ions of the opposite polarity to the surface charge exists close to the interface. It is formed due to attraction of counter-ions by the surface charges. This layer of higher ionic concentration is a part of the interfacial double layer. The concentration of the ions in this layer is higher as compared to the ionic strength of the liquid bulk. This leads to the higher electric conductivity of this layer.

Louis Winslow Austin was an American physicist known for his research on long-range radio transmissions.

Rudolf Hermann Arndt Kohlrausch was a German physicist.

Eduard August Grüneisen was a German physicist. He is best known for the Grüneisen parameter, the Mie–Grüneisen equation of state and the Bloch–Grüneisen temperature. He served as director of the Physics Department at the University of Marburg for 20 years, and was editor of Annalen der Physik together with Max Planck.

Margaret Eliza Maltby was an American physicist notable for her measurement of high electrolytic resistances and the conductivity of very dilute solutions. Maltby was the first woman to earn a Bachelor of Science degree from the Massachusetts Institute of Technology, and the first woman to earn a Ph.D. in physics from any German university.

Solid-state ionics is the study of ionic-electronic mixed conductor and fully ionic conductors and their uses. Some materials that fall into this category include inorganic crystalline and polycrystalline solids, ceramics, glasses, polymers, and composites. Solid-state ionic devices, such as solid oxide fuel cells, can be much more reliable and long-lasting, especially under harsh conditions, than comparable devices with fluid electrolytes.

Conductivity or specific conductance of an electrolyte solution is a measure of its ability to conduct electricity. The SI unit of conductivity is siemens per meter (S/m).
Beta dispersion is the phenomenon associated with the ability of a biological cell membrane to filter out low frequency currents and allow high frequency currents to pass through. It was originally hypothesized by Rudolf Höber in 1910 and confirmed through a series of experiments between 1910 and 1913.
Conductometry is a measurement of electrolytic conductivity to monitor a progress of chemical reaction. Conductometry has notable application in analytical chemistry, where conductometric titration is a standard technique. In usual analytical chemistry practice, the term conductometry is used as a synonym of conductometric titration while the term conductimetry is used to describe non-titrative applications. Conductometry is often applied to determine the total conductance of a solution or to analyze the end point of titrations that include ions.
In chemistry, ion transport number, also called the transference number, is the fraction of the total electric current carried in an electrolyte by a given ionic species i:

Eduard Riecke was a German experimental physicist.

Wilhelm von Beetz was a German physicist, known for his studies of electrical conductivity properties.

Alexander Alexandrovich Eichenwald was a Russian experimental physicist who worked on electrodynamics. He conducted experiments on electromagnetism, electrical fields, and on the construction of instruments to measure magnetic fields. His most famous experiment, following those of Wilhelm Röntgen, examined the predictions of James Clerk Maxwell. Named after them as the Röntgen-Eichenwald experiment, this demonstrated that the movement of static charges was no different from electric currents in that they produced an electromagnetic field.