The nephron is the minute or microscopic structural and functional unit of the kidney. It is composed of a renal corpuscle and a renal tubule. The renal corpuscle consists of a tuft of capillaries called a glomerulus and a cup-shaped structure called Bowman's capsule. The renal tubule extends from the capsule. The capsule and tubule are connected and are composed of epithelial cells with a lumen. A healthy adult has 1 to 1.5 million nephrons in each kidney. Blood is filtered as it passes through three layers: the endothelial cells of the capillary wall, its basement membrane, and between the foot processes of the podocytes of the lining of the capsule. The tubule has adjacent peritubular capillaries that run between the descending and ascending portions of the tubule. As the fluid from the capsule flows down into the tubule, it is processed by the epithelial cells lining the tubule: water is reabsorbed and substances are exchanged ; first with the interstitial fluid outside the tubules, and then into the plasma in the adjacent peritubular capillaries through the endothelial cells lining that capillary. This process regulates the volume of body fluid as well as levels of many body substances. At the end of the tubule, the remaining fluid—urine—exits: it is composed of water, metabolic waste, and toxins.

The collecting duct system of the kidney consists of a series of tubules and ducts that physically connect nephrons to a minor calyx or directly to the renal pelvis. The collecting duct participates in electrolyte and fluid balance through reabsorption and excretion, processes regulated by the hormones aldosterone and vasopressin.
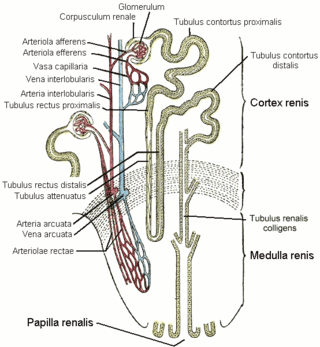
The distal convoluted tubule (DCT) is a portion of kidney nephron between the loop of Henle and the collecting tubule.

Renal physiology is the study of the physiology of the kidney. This encompasses all functions of the kidney, including maintenance of acid-base balance; regulation of fluid balance; regulation of sodium, potassium, and other electrolytes; clearance of toxins; absorption of glucose, amino acids, and other small molecules; regulation of blood pressure; production of various hormones, such as erythropoietin; and activation of vitamin D.

The proximal tubule is the segment of the nephron in kidneys which begins from the renal pole of the Bowman's capsule to the beginning of loop of Henle. At this location, the glomerular parietal epithelial cells (PECs) lining bowman’s capsule abruptly transition to proximal tubule epithelial cells (PTECs). The proximal tubule can be further classified into the proximal convoluted tubule (PCT) and the proximal straight tubule (PST).

In the kidney, the loop of Henle is the portion of a nephron that leads from the proximal convoluted tubule to the distal convoluted tubule. Named after its discoverer, the German anatomist Friedrich Gustav Jakob Henle, the loop of Henle's main function is to create a concentration gradient in the medulla of the kidney.
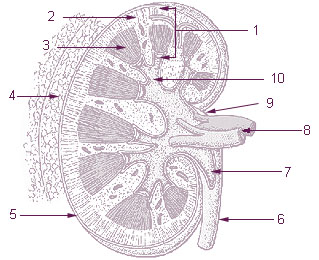
The renal medulla is the innermost part of the kidney. The renal medulla is split up into a number of sections, known as the renal pyramids. Blood enters into the kidney via the renal artery, which then splits up to form the segmental arteries which then branch to form interlobar arteries. The interlobar arteries each in turn branch into arcuate arteries, which in turn branch to form interlobular arteries, and these finally reach the glomeruli. At the glomerulus the blood reaches a highly disfavourable pressure gradient and a large exchange surface area, which forces the serum portion of the blood out of the vessel and into the renal tubules. Flow continues through the renal tubules, including the proximal tubule, the loop of Henle, through the distal tubule and finally leaves the kidney by means of the collecting duct, leading to the renal pelvis, the dilated portion of the ureter.
Hyperchloremia is an electrolyte disturbance in which there is an elevated level of chloride ions in the blood. The normal serum range for chloride is 96 to 106 mEq/L, therefore chloride levels at or above 110 mEq/L usually indicate kidney dysfunction as it is a regulator of chloride concentration. As of now there are no specific symptoms of hyperchloremia; however, it can be influenced by multiple abnormalities that cause a loss of electrolyte-free fluid, loss of hypotonic fluid, or increased administration of sodium chloride. These abnormalities are caused by diarrhea, vomiting, increased sodium chloride intake, renal dysfunction, diuretic use, and diabetes. Hyperchloremia should not be mistaken for hyperchloremic metabolic acidosis as hyperchloremic metabolic acidosis is characterized by two major changes: a decrease in blood pH and bicarbonate levels, as well as an increase in blood chloride levels. Instead those with hyperchloremic metabolic acidosis are usually predisposed to hyperchloremia.
An osmotic diuretic is a type of diuretic that inhibits reabsorption of water and sodium (Na). They are pharmacologically inert substances that are given intravenously. They increase the osmolarity of blood and renal filtrate. This fluid eventually becomes urine.

Renal tubular acidosis (RTA) is a medical condition that involves an accumulation of acid in the body due to a failure of the kidneys to appropriately acidify the urine. In renal physiology, when blood is filtered by the kidney, the filtrate passes through the tubules of the nephron, allowing for exchange of salts, acid equivalents, and other solutes before it drains into the bladder as urine. The metabolic acidosis that results from RTA may be caused either by insufficient secretion of hydrogen ions into the latter portions of the nephron or by failure to reabsorb sufficient bicarbonate ions from the filtrate in the early portion of the nephron. Although a metabolic acidosis also occurs in those with chronic kidney disease, the term RTA is reserved for individuals with poor urinary acidification in otherwise well-functioning kidneys. Several different types of RTA exist, which all have different syndromes and different causes. RTA is usually an incidental finding based on routine blood draws that show abnormal results. Clinically, patients may present with vague symptoms such as dehydration, mental status changes, or delayed growth in adolescents.

In renal physiology, reabsorption or tubular reabsorption is the process by which the nephron removes water and solutes from the tubular fluid (pre-urine) and returns them to the circulating blood. It is called reabsorption (and not absorption) because these substances have already been absorbed once (particularly in the intestines) and the body is reclaiming them from a postglomerular fluid stream that is on its way to becoming urine (that is, they will soon be lost to the urine unless they are reabsorbed from the tubule into the peritubular capillaries. This happens as a result of sodium transport from the lumen into the blood by the Na+/K+ATPase in the basolateral membrane of the epithelial cells. Thus, the glomerular filtrate becomes more concentrated, which is one of the steps in forming urine. Nephrons are divided into five segments, with different segments responsible for reabsorbing different substances. Reabsorption allows many useful solutes (primarily glucose and amino acids), salts and water that have passed through Bowman's capsule, to return to the circulation. These solutes are reabsorbed isotonically, in that the osmotic potential of the fluid leaving the proximal convoluted tubule is the same as that of the initial glomerular filtrate. However, glucose, amino acids, inorganic phosphate, and some other solutes are reabsorbed via secondary active transport through cotransport channels driven by the sodium gradient.
Sodium-dependent glucose cotransporters are a family of glucose transporter found in the intestinal mucosa (enterocytes) of the small intestine (SGLT1) and the proximal tubule of the nephron. They contribute to renal glucose reabsorption. In the kidneys, 100% of the filtered glucose in the glomerulus has to be reabsorbed along the nephron. If the plasma glucose concentration is too high (hyperglycemia), glucose passes into the urine (glucosuria) because SGLT are saturated with the filtered glucose.
In physiology, transport maximum refers to the point at which increase in concentration of a substance does not result in an increase in movement of a substance across a cell membrane.

Renal glycosuria is a rare condition in which the simple sugar glucose is excreted in the urine despite normal or low blood glucose levels. With normal kidney (renal) function, glucose is excreted in the urine only when there are abnormally elevated levels of glucose in the blood. However, in those with renal glycosuria, glucose is abnormally elevated in the urine due to improper functioning of the renal tubules, which are primary components of nephrons, the filtering units of the kidneys.

The sodium/glucose cotransporter 2 (SGLT2) is a protein that in humans is encoded by the SLC5A2 gene.
Renal glucose reabsorption is the part of kidney (renal) physiology that deals with the retrieval of filtered glucose, preventing it from disappearing from the body through the urine.
In physiology, splay is the difference between urine threshold and saturation, or TM, where saturation is the exhausted supply of renal reabsorption carriers. In simpler terms, splay is the concentration difference between a substance's maximum renal reabsorption vs. appearance in the urine. Splay is usually used in reference to glucose; other substances, such as phosphate, have virtually no splay at all.
Fanconi syndrome or Fanconi's syndrome is a syndrome of inadequate reabsorption in the proximal renal tubules of the kidney. The syndrome can be caused by various underlying congenital or acquired diseases, by toxicity, or by adverse drug reactions. It results in various small molecules of metabolism being passed into the urine instead of being reabsorbed from the tubular fluid. Fanconi syndrome affects the proximal tubules, namely, the proximal convoluted tubule (PCT), which is the first part of the tubule to process fluid after it is filtered through the glomerulus, and the proximal straight tubule, which leads to the descending limb of loop of Henle.

A diuretic is any substance that promotes diuresis, the increased production of urine. This includes forced diuresis. A diuretic tablet is sometimes colloquially called a water tablet. There are several categories of diuretics. All diuretics increase the excretion of water from the body, through the kidneys. There exist several classes of diuretic, and each works in a distinct way. Alternatively, an antidiuretic, such as vasopressin, is an agent or drug which reduces the excretion of water in urine.
Gliflozins are a class of drugs in the treatment of type 2 diabetes (T2D). They act by inhibiting sodium/glucose cotransporter 2 (SGLT-2), and are therefore also called SGLT-2 inhibitors. The efficacy of the drug is dependent on renal excretion and prevents glucose from going into blood circulation by promoting glucosuria. The mechanism of action is insulin independent.