Related Research Articles

An ion source is a device that creates atomic and molecular ions. Ion sources are used to form ions for mass spectrometers, optical emission spectrometers, particle accelerators, ion implanters and ion engines.

Tandem mass spectrometry, also known as MS/MS or MS2, is a technique in instrumental analysis where two or more stages of analysis using one or more mass analyzer are performed with an additional reaction step in between these analyses to increase their abilities to analyse chemical samples. A common use of tandem MS is the analysis of biomolecules, such as proteins and peptides.

In mass spectrometry, matrix-assisted laser desorption/ionization (MALDI) is an ionization technique that uses a laser energy-absorbing matrix to create ions from large molecules with minimal fragmentation. It has been applied to the analysis of biomolecules and various organic molecules, which tend to be fragile and fragment when ionized by more conventional ionization methods. It is similar in character to electrospray ionization (ESI) in that both techniques are relatively soft ways of obtaining ions of large molecules in the gas phase, though MALDI typically produces far fewer multi-charged ions.
Soft laser desorption (SLD) is laser desorption of large molecules that results in ionization without fragmentation. "Soft" in the context of ion formation means forming ions without breaking chemical bonds. "Hard" ionization is the formation of ions with the breaking of bonds and the formation of fragment ions.

Desorption electrospray ionization (DESI) is an ambient ionization technique that can be coupled to mass spectrometry (MS) for chemical analysis of samples at atmospheric conditions. Coupled ionization sources-MS systems are popular in chemical analysis because the individual capabilities of various sources combined with different MS systems allow for chemical determinations of samples. DESI employs a fast-moving charged solvent stream, at an angle relative to the sample surface, to extract analytes from the surfaces and propel the secondary ions toward the mass analyzer. This tandem technique can be used to analyze forensics analyses, pharmaceuticals, plant tissues, fruits, intact biological tissues, enzyme-substrate complexes, metabolites and polymers. Therefore, DESI-MS may be applied in a wide variety of sectors including food and drug administration, pharmaceuticals, environmental monitoring, and biotechnology.
Robert Graham Cooks is the Henry Bohn Hass Distinguished Professor of Chemistry in the Aston Laboratories for Mass Spectrometry at Purdue University. He is an ISI Highly Cited Chemist, with over 1,000 publications and an H-index of 144.
Michael L. Gross is Professor of Chemistry, Medicine, and Immunology, at Washington University in St. Louis. He was formerly Professor of Chemistry at the University of Nebraska-Lincoln from 1968–1994. He is recognized for his contributions to the field of mass spectrometry and ion chemistry. He is credited with the discovery of distonic ions, chemical species containing a radical and an ionic site on different atoms of the same molecule.

Matrix-assisted laser desorption electrospray ionization (MALDESI) was first introduced in 2006 as a novel ambient ionization technique which combines the benefits of electrospray ionization (ESI) and matrix-assisted laser desorption/ionization (MALDI). An infrared (IR) or ultraviolet (UV) laser can be utilized in MALDESI to resonantly excite an endogenous or exogenous matrix. The term 'matrix' refers to any molecule that is present in large excess and absorbs the energy of the laser, thus facilitating desorption of analyte molecules. The original MALDESI design was implemented using common organic matrices, similar to those used in MALDI, along with a UV laser. The current MALDESI source employs endogenous water or a thin layer of exogenously deposited ice as the energy-absorbing matrix where O-H symmetric and asymmetric stretching bonds are resonantly excited by a mid-IR laser.
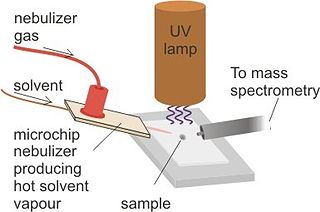
Desorption atmospheric pressure photoionization (DAPPI) is an ambient ionization technique for mass spectrometry that uses hot solvent vapor for desorption in conjunction with photoionization. Ambient Ionization techniques allow for direct analysis of samples without pretreatment. The direct analysis technique, such as DAPPI, eliminates the extraction steps seen in most nontraditional samples. DAPPI can be used to analyze bulkier samples, such as, tablets, powders, resins, plants, and tissues. The first step of this technique utilizes a jet of hot solvent vapor. The hot jet thermally desorbs the sample from a surface. The vaporized sample is then ionized by the vacuum ultraviolet light and consequently sampled into a mass spectrometer. DAPPI can detect a range of both polar and non-polar compounds, but is most sensitive when analyzing neutral or non-polar compounds. This technique also offers a selective and soft ionization for highly conjugated compounds.

Ambient ionization is a form of ionization in which ions are formed in an ion source outside the mass spectrometer without sample preparation or separation. Ions can be formed by extraction into charged electrospray droplets, thermally desorbed and ionized by chemical ionization, or laser desorbed or ablated and post-ionized before they enter the mass spectrometer.

Laser ablation electrospray ionization (LAESI) is an ambient ionization method for mass spectrometry that combines laser ablation from a mid-infrared (mid-IR) laser with a secondary electrospray ionization (ESI) process. The mid-IR laser is used to generate gas phase particles which are then ionized through interactions with charged droplets from the ESI source. LAESI was developed in Professor Akos Vertes lab by Peter Nemes in 2007 and it was marketed commercially by Protea Biosciences, Inc until 2017. Fiber-LAESI for single-cell analysis approach was developed by Bindesh Shrestha in Professor Vertes lab in 2009. LAESI is a novel ionization source for mass spectrometry (MS) that has been used to perform MS imaging of plants, tissues, cell pellets, and even single cells. In addition, LAESI has been used to analyze historic documents and untreated biofluids such as urine and blood. The technique of LAESI is performed at atmospheric pressure and therefore overcomes many of the obstacles of traditional MS techniques, including extensive and invasive sample preparation steps and the use of high vacuum. Because molecules and aerosols are ionized by interacting with an electrospray plume, LAESI's ionization mechanism is similar to SESI and EESI techniques.
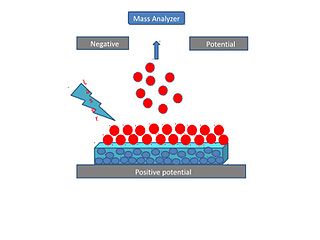
Surface-assisted laser desorption/ionization (SALDI) is a soft laser desorption technique used for mass spectrometry analysis of biomolecules, polymers, and small organic molecules. In its first embodiment Koichi Tanaka used a cobalt/glycerol liquid matrix and subsequent applications included a graphite/glycerol liquid matrix as well as a solid surface of porous silicon. The porous silicon represents the first matrix-free SALDI surface analysis allowing for facile detection of intact molecular ions, these porous silicon surfaces also facilitated the analysis of small molecules at the yoctomole level. At present laser desorption/ionization methods using other inorganic matrices such as nanomaterials are often regarded as SALDI variants. As an example, silicon nanowires as well as Titania nanotube arrays (NTA) have been used as substrates to detect small molecules. SALDI is used to detect proteins and protein-protein complexes. A related method named "ambient SALDI" - which is a combination of conventional SALDI with ambient mass spectrometry incorporating the direct analysis real time (DART) ion source has also been demonstrated. SALDI is considered one of the most important techniques in MS and has many applications.

Atmospheric pressure photoionization (APPI) is a soft ionization method used in mass spectrometry (MS) usually coupled to liquid chromatography (LC). Molecules are ionized using a vacuum ultraviolet (VUV) light source operating at atmospheric pressure, either by direct absorption followed by electron ejection or through ionization of a dopant molecule that leads to chemical ionization of target molecules. The sample is usually a solvent spray that is vaporized by nebulization and heat. The benefit of APPI is that it ionizes molecules across a broad range of polarity and is particularly useful for ionization of low polarity molecules for which other popular ionization methods such as electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) are less suitable. It is also less prone to ion suppression and matrix effects compared to ESI and APCI and typically has a wide linear dynamic range. The application of APPI with LC/MS is commonly used for analysis of petroleum compounds, pesticides, steroids, and drug metabolites lacking polar functional groups and is being extensively deployed for ambient ionization particularly for explosives detection in security applications.

Robert Reed Squires was an American chemist known for his work in gas phase ion chemistry and flowing afterglow mass spectrometry.

Desorption/ionization on silicon (DIOS) is a soft laser desorption method used to generate gas-phase ions for mass spectrometry analysis. DIOS is considered the first surface-based surface-assisted laser desorption/ionization (SALDI-MS) approach. Prior approaches were accomplished using nanoparticles in a matrix of glycerol, while DIOS is a matrix-free technique in which a sample is deposited on a nanostructured surface and the sample desorbed directly from the nanostructured surface through the adsorption of laser light energy. DIOS has been used to analyze organic molecules, metabolites, biomolecules and peptides, and, ultimately, to image tissues and cells.
Mahdi Muhammad Abu-Omar is a Palestinian-American chemist, currently the Duncan and Suzanne Mellichamp Professor of Green Chemistry in the Departments of Chemistry & Biochemistry and Chemical Engineering at University of California, Santa Barbara.
In mass spectrometry, a matrix is a compound that promotes the formation of ions. Matrix compounds are used in matrix-assisted laser desorption/ionization (MALDI), matrix-assisted ionization (MAI), and fast atom bombardment (FAB).

Laser diode thermal desorption (LDTD) is an ionization technique that is coupled to mass spectrometry to analyze samples with atmospheric pressure chemical ionization (APCI). It uses a laser to thermally desorb analytes that are deposited on a stainless steel sheet sample holder, called LazWell. The coupling of LDTD and APCI is considered to be a soft-ionization technique. With LDTD-APCI, it is possible to analyze samples in forensics, pharmaceuticals, environment, food and clinical studies. LDTD is suitable for small molecules between 0 and 1200 Da and some peptides such as cyclosporine.

Julia Laskin is the William F. and Patty J. Miller Professor of Analytical Chemistry at Purdue University. Her research is focused on the fundamental understanding of ion-surface collisions, understanding of phenomena underlying chemical analysis of large molecules in complex heterogeneous environments, and the development of new instrumentation and methods in preparative and imaging mass spectrometry.
References
- 1 2 3 Thayer, Ann M. (2015). "Frank H. Field & Joe L. Franklin Award For Outstanding Achievement In Mass Spectrometry". ACS Chemical and Engineering News. Retrieved 2022-06-07.
- ↑ "Purdue Chemistry: Kenttämaa Labs: Hilkka I. Kenttämaa". www.chem.purdue.edu. Retrieved 2022-06-07.
- 1 2 "Purdue University: College of Science: Chemistry professor receives distinguished honors". www.purdue.edu. Retrieved 2022-06-07.
- ↑ Feng, Erlu; Ma, Xin; Kenttämaa, Hilkka I. (2021-06-08). "Characterization of Protonated Substituted Ureas by Using Diagnostic Gas-Phase Ion-Molecule Reactions Followed by Collision-Activated Dissociation in Tandem Mass Spectrometry Experiments". Analytical Chemistry. 93 (22): 7851–7859. doi:10.1021/acs.analchem.1c00326. ISSN 0003-2700. PMID 34028247. S2CID 235168836.
- ↑ Kumar, Rashmi; Kenttämaa, Hilkka I. (2020-10-07). "Effects of Analyte Concentration on the Protonation Sites of 4-Aminobenzoic Acid upon Atmospheric Pressure Chemical Ionization As Revealed by Gas-Phase Ion–Molecule Reactions". Journal of the American Society for Mass Spectrometry. 31 (10): 2210–2217. doi:10.1021/jasms.0c00285. ISSN 1044-0305. PMID 32852952. S2CID 221359886.
- ↑ Yu, Zaikuan; Murria, Priya; Easton, Mckay W.; Degenstein, John C.; Zhu, Hanyu; Xu, Lan; Agrawal, Rakesh; Delgass, W. Nicholas; Ribeiro, Fabio H.; Kenttämaa, Hilkka I. (2019-10-24). "Exploring the Reaction Mechanisms of Fast Pyrolysis of Xylan Model Compounds via Tandem Mass Spectrometry and Quantum Chemical Calculations". The Journal of Physical Chemistry A. 123 (42): 9149–9157. Bibcode:2019JPCA..123.9149Y. doi:10.1021/acs.jpca.9b04438. ISSN 1089-5639. PMID 31545607. S2CID 202748635.
- ↑ Kiminkinen, L. K. Marjatta; Stirk, Krista G.; Kenttamaa, Hilkka I. (1992). "A remarkably stable organic radical cation: the distonic isomer of the unstable radical cation of dimethyl propyl phosphate". Journal of the American Chemical Society. 114 (6): 2027–2031. doi:10.1021/ja00032a015. ISSN 0002-7863.
- ↑ Jarrell, Tiffany M.; Owen, Benjamin C.; Riedeman, James S.; Prentice, Boone M.; Pulliam, Chris J.; Max, Joann; Kenttämaa, Hilkka I. (2017-06-01). "Laser-Induced Acoustic Desorption/Electron Ionization of Amino Acids and Small Peptides". Journal of the American Society for Mass Spectrometry. 28 (6): 1091–1098. Bibcode:2017JASMS..28.1091J. doi:10.1007/s13361-017-1684-1. ISSN 1044-0305. PMID 28500583. S2CID 41709530.
- ↑ Ma, Xin; Zhang, Yuyang; Lei, Hao-Ran; Kenttämaa, Hilkka I. (2019-05-01). "Laser-induced acoustic desorption". MRS Bulletin. 44 (5): 372–381. Bibcode:2019MRSBu..44..372M. doi:10.1557/mrs.2019.105. ISSN 1938-1425. S2CID 164846809.
- ↑ "Ulkomaiset jäsenet". Tiedeakatemia (in Finnish). Retrieved 2022-06-07.
- ↑ "Mass Spectrometry Reviews Editorial Board". Wiley Analytical Science Mass Spectrometry Reviews. Retrieved June 7, 2022.
- ↑ "ASMS News". Journal of the American Society for Mass Spectrometry. 2 (4): 350–353. 1991-08-01. doi: 10.1016/1044-0305(91)80027-5 . ISSN 1044-0305.
- ↑ "News and Views" (PDF). American Society for Mass Spectrometry. 2015. Retrieved 2022-06-07.
- ↑ "Research Awards". www.asms.org. Retrieved 2022-06-07.
- ↑ "NSF Award Search: Award # 9631701 - Oxidative Damage and Repair of Biological Macromolecules". www.nsf.gov. Retrieved 2022-06-07.
- ↑ "Hikka Kenttamaa presented 2009 Provost's Award for Outstanding Graduate Faculty Mentor - Purdue University Department of Chemistry". www.chem.purdue.edu. Retrieved 2022-06-07.
- ↑ "ACS 2015 National Award Winners". ACS Chemical & Engineering News. 2014-08-11. Retrieved 2022-06-07.
- ↑ "2015-16 Innovators Hall of Fame Inductees - Office of Technology Commercialization - Purdue University". www.prf.org. Retrieved 2022-06-07.
- ↑ "2016 Fellows | American Association for the Advancement of Science". www.aaas.org. Retrieved 2022-06-07.
- ↑ "Hilkka Kenttamaa". The Analytical Scientist. Retrieved 2022-06-07.
- ↑ "Purdue researchers part of team honored by NASA". School of Aeronautics and Astronautics - Purdue University. Retrieved 2022-06-07.
- ↑ Hoffpauir, Daniel (2020-11-25). "2020 NASA Engineering and Safety Center Honor Awards". NASA. Retrieved 2022-06-07.
- ↑ Wysocki, Vicki H.; Kenttämaa, Hilkka I.; Cooks, R.Graham (1987). "Internal energy distributions of isolated ions after activation by various methods". International Journal of Mass Spectrometry and Ion Processes. 75 (2): 181–208. Bibcode:1987IJMSI..75..181W. doi:10.1016/0168-1176(87)83054-9.
- ↑ Stirk, Krista M.; Kiminkinen, L. K. Marjatta; Kenttamaa, Hilkka I. (1992). "Ion-molecule reactions of distonic radical cations". Chemical Reviews. 92 (7): 1649–1665. doi:10.1021/cr00015a008. ISSN 0009-2665.
- ↑ Pinkston, David S.; Duan, Penggao; Gallardo, Vanessa A.; Habicht, Steven C.; Tan, Xiaoli; Qian, Kuangnan; Gray, Murray; Müllen, Klaus; Kenttämaa, Hilkka I. (2009-11-19). "Analysis of Asphaltenes and Asphaltene Model Compounds by Laser-Induced Acoustic Desorption/Fourier Transform Ion Cyclotron Resonance Mass Spectrometry". Energy & Fuels. 23 (11): 5564–5570. doi:10.1021/ef9006005. ISSN 0887-0624.
- ↑ Borton, David; Pinkston, David S.; Hurt, Matthew R.; Tan, Xiaoli; Azyat, Khalid; Scherer, Alexander; Tykwinski, Rik; Gray, Murray; Qian, Kuangnan; Kenttämaa, Hilkka I. (2010-10-21). "Molecular Structures of Asphaltenes Based on the Dissociation Reactions of Their Ions in Mass Spectrometry". Energy & Fuels. 24 (10): 5548–5559. doi:10.1021/ef1007819. ISSN 0887-0624.
- ↑ Parsell, Trenton H.; Owen, Benjamin C.; Klein, Ian; Jarrell, Tiffany M.; Marcum, Christopher L.; Haupert, Laura J.; Amundson, Lucas M.; Kenttämaa, Hilkka I.; Ribeiro, Fabio; Miller, Jeffrey T.; Abu-Omar, Mahdi M. (2013). "Cleavage and hydrodeoxygenation (HDO) of C–O bonds relevant to lignin conversion using Pd/Zn synergistic catalysis". Chem. Sci. 4 (2): 806–813. doi:10.1039/C2SC21657D. ISSN 2041-6520.
- ↑ Parsell, Trenton; Yohe, Sara; Degenstein, John; Jarrell, Tiffany; Klein, Ian; Gencer, Emre; Hewetson, Barron; Hurt, Matt; Kim, Jeong Im; Choudhari, Harshavardhan; Saha, Basudeb (2015). "A synergistic biorefinery based on catalytic conversion of lignin prior to cellulose starting from lignocellulosic biomass". Green Chemistry. 17 (3): 1492–1499. doi:10.1039/C4GC01911C. ISSN 1463-9262. OSTI 1210426.
- ↑ Luo, Hao; Klein, Ian M.; Jiang, Yuan; Zhu, Hanyu; Liu, Baoyuan; Kenttämaa, Hilkka I.; Abu-Omar, Mahdi M. (2016-04-04). "Total Utilization of Miscanthus Biomass, Lignin and Carbohydrates, Using Earth Abundant Nickel Catalyst". ACS Sustainable Chemistry & Engineering. 4 (4): 2316–2322. doi: 10.1021/acssuschemeng.5b01776 . ISSN 2168-0485.
- ↑ Fingas, Merv, ed. (2022-05-23). The Chemistry of Oil and Petroleum Products. De Gruyter. doi:10.1515/9783110694529. ISBN 978-3-11-069452-9. S2CID 249083493.
- ↑ USapplication 7619217B2 Ryan C Shea, Hilkka I Kenttamaa, Weldon E Vaughn, Steven C Habicht, James Perez/Purdue Research Foundation: "High power laser induced acoustic desorption probe" filing date 29.05.2007
- ↑ USapplication 8344319B2 Hilkka I. Kenttamaa, II David Jesse Borton, Jinshan Gao, Zhicheng Jin, Benjamin Curtis Owen/Purdue Research Foundation: "Laser-induced acoustic desorption/atmospheric pressure chemical ionization of compounds" filing date 05.04.2011
- ↑ WOapplication 2013103417A3 David J. BORTON, Nelson R. VINUEZA BENITEZ, Lucas M. AMUNDSON, Matthew R. HURT, Hilkka I. KENTTAMAA/Purdue Research Foundation: "Sample deposition chamber for laser-induced acoustic desorption (liad) foils" filing date 10.10.2012
- ↑ USapplication 9496127B2 Benjamin C. Owen, Hilkka I. Kenttamaa/Purdue Research Foundation: "Differentially pumped dual linear quadrupole ion trap mass spectrometer" filing date 15.09.2015
- ↑ "Multichannel Pulsed Valve Inlet System | Purdue OTC". inventions.prf.org. Retrieved 2022-06-07.