Related Research Articles

Chemotherapy is the type of cancer treatment that uses one or more anti-cancer drugs in a standard regimen. Chemotherapy may be given with a curative intent, or it may aim only to prolong life or to reduce symptoms. Chemotherapy is one of the major categories of the medical discipline specifically devoted to pharmacotherapy for cancer, which is called medical oncology.
Angiogenesis is the physiological process through which new blood vessels form from pre-existing vessels, formed in the earlier stage of vasculogenesis. Angiogenesis continues the growth of the vasculature mainly by processes of sprouting and splitting, but processes such as coalescent angiogenesis, vessel elongation and vessel cooption also play a role. Vasculogenesis is the embryonic formation of endothelial cells from mesoderm cell precursors, and from neovascularization, although discussions are not always precise. The first vessels in the developing embryo form through vasculogenesis, after which angiogenesis is responsible for most, if not all, blood vessel growth during development and in disease.
An angiogenesis inhibitor is a substance that inhibits the growth of new blood vessels (angiogenesis). Some angiogenesis inhibitors are endogenous and a normal part of the body's control and others are obtained exogenously through pharmaceutical drugs or diet.
Moses Judah Folkman was an American biologist and pediatric surgeon best known for his research on tumor angiogenesis, the process by which a tumor attracts blood vessels to nourish itself and sustain its existence. He founded the field of angiogenesis research, which has led to the discovery of a number of therapies based on inhibiting or stimulating neovascularization.

Endostatin is a naturally occurring, 20-kDa C-terminal fragment derived from type XVIII collagen. It is reported to serve as an anti-angiogenic agent, similar to angiostatin and thrombospondin.

The enhanced permeability and retention (EPR) effect is a controversial concept by which molecules of certain sizes tend to accumulate in tumor tissue much more than they do in normal tissues. The general explanation that is given for this phenomenon is that, in order for tumor cells to grow quickly, they must stimulate the production of blood vessels. VEGF and other growth factors are involved in cancer angiogenesis. Tumor cell aggregates as small as 150–200 μm, start to become dependent on blood supply carried out by neovasculature for their nutritional and oxygen supply. These newly formed tumor vessels are usually abnormal in form and architecture. They are poorly aligned defective endothelial cells with wide fenestrations, lacking a smooth muscle layer, or innervation with a wider lumen, and impaired functional receptors for angiotensin II. Furthermore, tumor tissues usually lack effective lymphatic drainage. All of these factors lead to abnormal molecular and fluid transport dynamics, especially for macromolecular drugs. This phenomenon is referred to as the "enhanced permeability and retention (EPR) effect" of macromolecules and lipids in solid tumors. The EPR effect is further enhanced by many pathophysiological factors involved in enhancement of the extravasation of macromolecules in solid tumor tissues. For instance, bradykinin, nitric oxide / peroxynitrite, prostaglandins, vascular permeability factor, tumor necrosis factor and others. One factor that leads to the increased retention is the lack of lymphatics around the tumor region which would filter out such particles under normal conditions.

Midkine, also known as neurite growth-promoting factor 2 (NEGF2), is a protein that in humans is encoded by the MDK gene.

A mitotic inhibitor, microtubule inhibitor, or tubulin inhibitor, is a drug that inhibits mitosis, or cell division, and is used in treating cancer, gout, and nail fungus. These drugs disrupt microtubules, which are structures that pull the chromosomes apart when a cell divides. Mitotic inhibitors are used in cancer treatment, because cancer cells are able to grow through continuous division that eventually spread through the body (metastasize). Thus, cancer cells are more sensitive to inhibition of mitosis than normal cells. Mitotic inhibitors are also used in cytogenetics, where they stop cell division at a stage where chromosomes can be easily examined.

Arginylglycylaspartic acid (RGD) is the most common peptide motif responsible for cell adhesion to the extracellular matrix (ECM), found in species ranging from Drosophila to humans. Cell adhesion proteins called integrins recognize and bind to this sequence, which is found within many matrix proteins, including fibronectin, fibrinogen, vitronectin, osteopontin, and several other adhesive extracellular matrix proteins. The discovery of RGD and elucidation of how RGD binds to integrins has led to the development of a number of drugs and diagnostics, while the peptide itself is used ubiquitously in bioengineering. Depending on the application and the integrin targeted, RGD can be chemically modified or replaced by a similar peptide which promotes cell adhesion.
Angioprevention is the concept of preventing disease development or progression through the inhibition of angiogenesis, the process of forming blood vessels. The concept of angioprevention has been developed by Adriana Albini and co-workers who showed that several drugs and natural compounds for cancer chemo-prevention actually prevent tumor blood vessel formation. This concept has been furthered through the identification of many other "angiopreventive" compounds.

Inspyr Therapeutics, Inc. is a development-stage pharmaceutical company based in San Antonio, Texas. The company is focused on therapeutics that deliver a cancer-destroying drug directly to the tumor or its supporting environment, the tumor vasculature.
Tumor-associated macrophages (TAMs) are a class of immune cells present in high numbers in the microenvironment of solid tumors. They are heavily involved in cancer-related inflammation. Macrophages are known to originate from bone marrow-derived blood monocytes or yolk sac progenitors, but the exact origin of TAMs in human tumors remains to be elucidated. The composition of monocyte-derived macrophages and tissue-resident macrophages in the tumor microenvironment depends on the tumor type, stage, size, and location, thus it has been proposed that TAM identity and heterogeneity is the outcome of interactions between tumor-derived, tissue-specific, and developmental signals.

A nanocarrier is nanomaterial being used as a transport module for another substance, such as a drug. Commonly used nanocarriers include micelles, polymers, carbon-based materials, liposomes and other substances. Nanocarriers are currently being studied for their use in drug delivery and their unique characteristics demonstrate potential use in chemotherapy. This class of materials was first reported by a team of researchers of University of Évora, Alentejo in early 1960's, and grew exponentially in relevance since then.
Directed enzyme prodrug therapy (DEPT) uses enzymes artificially introduced into the body to convert prodrugs, which have no or poor biologically activity, to the active form in the desired location within the body. Many chemotherapy drugs for cancer lack tumour specificity and the doses required to reach therapeutic levels in the tumour are often toxic to other tissues. DEPT strategies are an experimental method of reducing the systemic toxicity of a drug, by achieving high levels of the active drug only at the desired site. This article describes the variations of DEPT technology.

Vasculogenic mimicry (VM) is a strategy used by tumors to ensure sufficient blood supply is brought to its cells through establishing new tumor vascularization. This process is similar to tumor angiogenesis; on the other hand vascular mimicry is unique in that this process occurs independent of endothelial cells. Vasculature is instead developed de novo by cancer cells, which under stress conditions such as hypoxia, express similar properties to stem cells, capable of differentiating to mimic the function of endothelial cells and form vasculature-like structures. The ability of tumors to develop and harness nearby vasculature is considered one of the hallmarks of cancer disease development and is thought to be closely linked to tumor invasion and metastasis. Vascular mimicry has been observed predominantly in aggressive and metastatic cancers and has been associated with negative tumor characteristics such as increased metastasis, increased tissue invasion, and overall poor outcomes for patient survival. Vascular mimicry poses a serious problem for current therapeutic strategies due to its ability to function in the presence of Anti-angiogenic therapeutic agents. In fact, such therapeutics have been found to actually drive VM formation in tumors, causing more aggressive and difficult to treat tumors to develop.
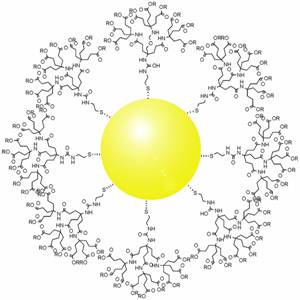
Gold nanoparticles in chemotherapy and radiotherapy is the use of colloidal gold in therapeutic treatments, often for cancer or arthritis. Gold nanoparticle technology shows promise in the advancement of cancer treatments. Some of the properties that gold nanoparticles possess, such as small size, non-toxicity and non-immunogenicity make these molecules useful candidates for targeted drug delivery systems. With tumor-targeting delivery vectors becoming smaller, the ability to by-pass the natural barriers and obstacles of the body becomes more probable. To increase specificity and likelihood of drug delivery, tumor specific ligands may be grafted onto the particles along with the chemotherapeutic drug molecules, to allow these molecules to circulate throughout the tumor without being redistributed into the body.

Tumor-associated endothelial cells or tumor endothelial cells (TECs) refers to cells lining the tumor-associated blood vessels that control the passage of nutrients into surrounding tumor tissue. Across different cancer types, tumor-associated blood vessels have been discovered to differ significantly from normal blood vessels in morphology, gene expression, and functionality in ways that promote cancer progression. There has been notable interest in developing cancer therapeutics that capitalize on these abnormalities of the tumor-associated endothelium to destroy tumors.
The host response to cancer therapy is defined as a physiological response of the non-malignant cells of the body to a specific cancer therapy. The response is therapy-specific, occurring independently of cancer type or stage.
Metronomic therapy is a new type of chemotherapy in which anti-cancer drugs are administered in a lower dose than the maximum tolerated dose repetitively over a long period to treat cancers with fewer side effects. Metronomic therapy is shown to affect both tumor microenvironment and tumor cells to achieve its therapeutic effects. Metronomic therapy is also cost-effective as a lower dose is used compared to conventional chemotherapy. The use of metronomic therapy has been extensively investigated and can be advantageous in selected group of patients. Yet, more clinical trials are necessary to generalize the method.

Michele 'Miki' De Palma is an Italian biologist and a professor at EPFL. He is known for his work on the role of macrophages in cancer progression and the discovery of Tie2-expressing angiogenic monocytes.
References
This article includes a list of references, related reading, or external links, but its sources remain unclear because it lacks inline citations .(April 2009) |
- Nanocell targets cancer
- Nanocell's double hit on cancer
- MIT engineers an anti-cancer smart bomb
- Sengupta S, Eavarone D, Capila I, Zhao G, Watson N, Kiziltepe T, Sasisekharan R. Temporal targeting of tumour cells and neovasculature with a nanoscale delivery system. Nature. 2005 Jul 28;436(7050):568-72.