
Nanopore sequencing is a third generation approach used in the sequencing of biopolymers — specifically, polynucleotides in the form of DNA or RNA.

Resonance Raman spectroscopy is a variant of Raman spectroscopy in which the incident photon energy is close in energy to an electronic transition of a compound or material under examination. This similarity in energy (resonance) leads to greatly increased intensity of the Raman scattering of certain vibrational modes, compared to ordinary Raman spectroscopy.

A nanomotor is a molecular or nanoscale device capable of converting energy into movement. It can typically generate forces on the order of piconewtons.

Cadmium selenide is an inorganic compound with the formula CdSe. It is a black to red-black solid that is classified as a II-VI semiconductor of the n-type. It is a pigment, but applications are declining because of environmental concerns.
Aqueous biphasic systems (ABS) or aqueous two-phase systems (ATPS) are clean alternatives for traditional organic-water solvent extraction systems.

Molecular motors are natural (biological) or artificial molecular machines that are the essential agents of movement in living organisms. In general terms, a motor is a device that consumes energy in one form and converts it into motion or mechanical work; for example, many protein-based molecular motors harness the chemical free energy released by the hydrolysis of ATP in order to perform mechanical work. In terms of energetic efficiency, this type of motor can be superior to currently available man-made motors. One important difference between molecular motors and macroscopic motors is that molecular motors operate in the thermal bath, an environment in which the fluctuations due to thermal noise are significant.
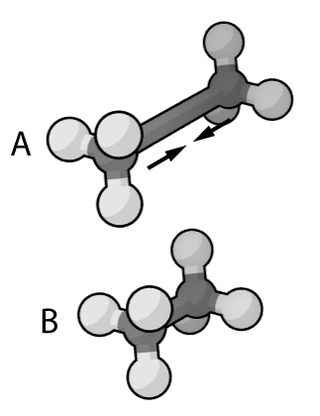
In the context of chemistry, molecular physics, physical chemistry, and molecular modelling, a force field is a computational model that is used to describe the forces between atoms within molecules or between molecules as well as in crystals. Force fields are a variety of interatomic potentials. More precisely, the force field refers to the functional form and parameter sets used to calculate the potential energy of a system on the atomistic level. Force fields are usually used in molecular dynamics or Monte Carlo simulations. The parameters for a chosen energy function may be derived from classical laboratory experiment data, calculations in quantum mechanics, or both. Force fields utilize the same concept as force fields in classical physics, with the main difference being that the force field parameters in chemistry describe the energy landscape on the atomistic level. From a force field, the acting forces on every particle are derived as a gradient of the potential energy with respect to the particle coordinates.

Methane monooxygenase (MMO) is an enzyme capable of oxidizing the C-H bond in methane as well as other alkanes. Methane monooxygenase belongs to the class of oxidoreductase enzymes.
Enzyme catalysis is the increase in the rate of a process by a biological molecule, an "enzyme". Most enzymes are proteins, and most such processes are chemical reactions. Within the enzyme, generally catalysis occurs at a localized site, called the active site.

Nanofluidics is the study of the behavior, manipulation, and control of fluids that are confined to structures of nanometer characteristic dimensions. Fluids confined in these structures exhibit physical behaviors not observed in larger structures, such as those of micrometer dimensions and above, because the characteristic physical scaling lengths of the fluid, very closely coincide with the dimensions of the nanostructure itself.
Self-assembling peptides are a category of peptides which undergo spontaneous assembling into ordered nanostructures. Originally described in 1993, these designer peptides have attracted interest in the field of nanotechnology for their potential for application in areas such as biomedical nanotechnology, tissue cell culturing, molecular electronics, and more.

Carbon nanotube chemistry involves chemical reactions, which are used to modify the properties of carbon nanotubes (CNTs). CNTs can be functionalized to attain desired properties that can be used in a wide variety of applications. The two main methods of CNT functionalization are covalent and non-covalent modifications.

A carbon nanothread is a sp3-bonded, one-dimensional carbon crystalline nanomaterial. The tetrahedral sp3-bonding of its carbon is similar to that of diamond. Nanothreads are only a few atoms across, more than 300,000 times thinner than a human hair. They consist of a stiff, strong carbon core surrounded by hydrogen atoms. Carbon nanotubes, although also one-dimensional nanomaterials, in contrast have sp2-carbon bonding as is found in graphite. The smallest carbon nanothread has a diameter of only 0.2 nanometers, much smaller than the diameter of a single-wall carbon nanotube.
Water shortages have become an increasingly pressing concern recently and with recent predictions of a high probability of the current drought turning into a megadrought occurring in the western United States, technologies involving water treatment and processing need to improve. Carbon nanotubes (CNT) have been the subject of extensive studies because they demonstrate a range of unique properties that existing technologies lack. For example, carbon nanotube membranes can demonstrate higher water flux with lower energy than current membranes. These membranes can also filter out particles that are too small for conventional systems which can lead to better water purification techniques and less waste. The largest obstacle facing CNT is processing as it is difficult to produce them in the large quantities that most of these technologies will require.
Suzanne A. Blum is an American professor of chemistry at the University of California, Irvine. Blum works on mechanistic chemistry, most recently focusing on borylation reactions and the development of single-molecule and single-particle fluorescence microscopy to study organic chemistry and catalysis. She received the American Chemical Society's Arthur C. Cope Scholar Award in 2023.

Dr. Dan Thomas Major is a Professor of Chemistry at Bar Ilan University specializing in Computational Chemistry.
An Artificial Metalloenzyme (ArM) is a designer metalloprotein, not found in nature, which can catalyze desired chemical reactions. Despite fitting into classical enzyme categories, ArMs also have potential in new-to-nature chemical reactivity like catalysing Suzuki coupling, Metathesis etc., which were never reported among natural enzymatic reactions.

Mei Hong is a Chinese-American biophysical chemist and professor of chemistry at the Massachusetts Institute of Technology. She is known for her creative development and application of solid-state nuclear magnetic resonance (ssNMR) spectroscopy to elucidate the structures and mechanisms of membrane proteins, plant cell walls, and amyloid proteins. She has received a number of recognitions for her work, including the American Chemical Society Nakanishi Prize in 2021, Günther Laukien Prize in 2014, the Protein Society Young Investigator award in 2012, and the American Chemical Society’s Pure Chemistry award in 2003.
Julia A. Kovacs is an American chemist specializing in bioinorganic chemistry. She is professor of chemistry at the University of Washington. Her research involves synthesizing small-molecule mimics of the active sites of metalloproteins, in order to investigate how cysteinates influence the function of non-heme iron enzymes, and the mechanism of the oxygen-evolving complex (OEC).
Katsumi Kaneko was born in Yokohama (Kanagawa), Japan. He graduated with a Bachelor of Engineering degree in 1969 from Yokohama National University, Yokohama. He received a master's degree in physical chemistry at The University of Tokyo, in 1971. He received Doctor of Science in solid state chemistry in 1978 for submitted thesis from The University of Tokyo, entitled “Electrical Properties and Defect Structures of Iron Hydroxide Oxide











