
Biological control or biocontrol is a method of controlling pests, whether pest animals such as insects and mites, weeds, or pathogens affecting animals or plants by using other organisms. It relies on predation, parasitism, herbivory, or other natural mechanisms, but typically also involves an active human management role. It can be an important component of integrated pest management (IPM) programs.
Dauer describes an alternative developmental stage of nematode worms, particularly rhabditids including Caenorhabditis elegans, whereby the larva goes into a type of stasis and can survive harsh conditions. Since the entrance of the dauer stage is dependent on environmental cues, it represents a classic and well studied example of polyphenism. The dauer state is given other names in the various types of nematodes such as ‘diapause’ or ‘hypobiosis’, but since the C. elegans nematode has become the most studied nematode, the term ‘dauer stage’ or 'dauer larvae' is becoming universally recognised when referring to this state in other free-living nematodes. The dauer stage is also considered to be equivalent to the infective stage of parasitic nematode larvae.

Entomopathogenic nematodes (EPN) are a group of nematodes, that cause death to insects. The term entomopathogenic has a Greek origin, with entomon, meaning insect, and pathogenic, which means causing disease. They are animals that occupy a bio control middle ground between microbial pathogens and predator/parasitoids. Although many other parasitic thread worms cause diseases in living organisms, entomopathogenic nematodes are specific in only infecting insects. Entomopathogenic nematodes (EPNs) live parasitically inside the infected insect host, and so they are termed as endoparasitic. They infect many different types of insects living in the soil like the larval forms of moths, butterflies, flies and beetles as well as adult forms of beetles, grasshoppers and crickets. EPNs have been found all over the world in a range of ecologically diverse habitats. They are highly diverse, complex and specialized. The most commonly studied entomopathogenic nematodes are those that can be used in the biological control of harmful insects, the members of Steinernematidae and Heterorhabditidae. They are the only insect-parasitic nematodes possessing an optimal balance of biological control attributes.

Deroceras reticulatum, common names the "grey field slug", "grey garden slug", and "milky slug", is a species of small air-breathing land slug, a terrestrial pulmonate gastropod mollusc in the family Agriolimacidae. This species is an important agricultural pest.

Angiostrongylus cantonensis is a nematode (roundworm) parasite that causes angiostrongyliasis, an infection that is the most common cause of eosinophilic meningitis in Southeast Asia and the Pacific Basin. The nematode commonly resides in the pulmonary arteries of rats, giving it the common name rat lungworm. Snails are the primary intermediate hosts, where larvae develop until they are infectious.
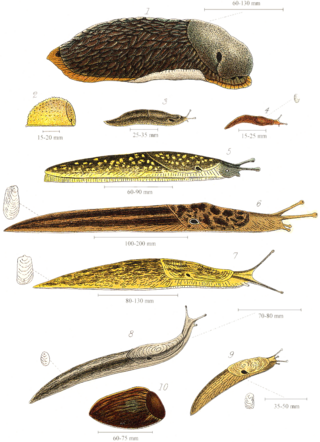
Slug, or land slug, is a common name for any apparently shell-less terrestrial gastropod mollusc. The word slug is also often used as part of the common name of any gastropod mollusc that has no shell, a very reduced shell, or only a small internal shell, particularly sea slugs and semi-slugs.

The nematodes, roundworms or eelworms constitute the phylum Nematoda. They are a diverse animal phylum inhabiting a broad range of environments. Most species are free-living, feeding on microorganisms, but there are many that are parasitic. The parasitic worms (helminths) are the cause of soil-transmitted helminthiases.

Angiostrongylus vasorum, also known as French heartworm, is a species of parasitic nematode in the family Metastrongylidae. It causes the disease canine angiostrongylosis in dogs. It is not zoonotic, that is, it cannot be transmitted to humans.
Moraxella osloensis is a Gram-negative oxidase-positive, aerobic bacterium within the family Moraxellaceae in the gamma subdivision of the purple bacteria.
Xenorhabdus is a genus of motile, gram-negative bacteria from the family of the Morganellaceae. All the species of the genus are only known to live in symbiosis with soil entomopathogenic nematodes from the genus Steinernema.

Purpureocillium lilacinum is a species of filamentous fungus in the family Ophiocordycipitaceae. It has been isolated from a wide range of habitats, including cultivated and uncultivated soils, forests, grassland, deserts, estuarine sediments and sewage sludge, and insects. It has also been found in nematode eggs, and occasionally from females of root-knot and cyst nematodes. In addition, it has frequently been detected in the rhizosphere of many crops. The species can grow at a wide range of temperatures – from 8 to 38 °C for a few isolates, with optimal growth in the range 26 to 30 °C. It also has a wide pH tolerance and can grow on a variety of substrates. P. lilacinum has shown promising results for use as a biocontrol agent to control the growth of destructive root-knot nematodes.

Deroceras invadens is a species of air-breathing land slug, a terrestrial pulmonate gastropod mollusc in the family Agriolimacidae. Until 2011, this widely distributed species was known as Deroceras panormitanum, and earlier as Deroceras caruanae or Agriolimax caruanae, but Reise et al. (2011) showed that these names refer to a distinct species of similar external appearance known at that time only from Sicily and Malta. Consequently, although the more widespread species was already well known, it then had to be redescribed under the new name of D. invadens. Genetic evidence has indicated that D. invadens is native in southern Italy, including parts of Sicily, and possibly parts of central Italy. Elsewhere it has been introduced, predominantly within the last 100 years, but its spread has been constrained by cold winter temperatures.
Pasteuria is a genus of mycelial and endospore-forming, nonmotile gram-positive bacteria that are obligate parasites of some nematodes and crustaceans. The genus of Pasteuria was previously classified within the family Alicyclobacillaceae, but has since been moved to the family Pasteuriaceae.

Heterorhabditis megidis is a species of nematodes in the genus Heterorhabditis. All species of this genus are obligate parasites of insects, and some are used as biological control agents for the control of pest insects.
Skrjabingylus nasicola is a species of parasitic nematode in the family Metastrongylidae. Its lifecycle includes an intermediary mollusc host and a paratenic host, a shrew or small rodent. Adult worms are found in the nasal sinuses of mustelids.
Phasmarhabditis californica is a nematode in the family Rhabditidae. It is a lethal facultative parasite of terrestrial gastropods.
Phasmarhabditis (Greek: Phasma = (φάσμα ; rhabditis = is a genus of bacterial-feeding nematodes which are facultative parasites whose primary hosts are terrestrial gastropods. The name comes from Greek: Phasma- (φάσμα ; rhabditis = rod-like (ῥάβδος. The genus is made up of 18 species including P. hermaphrodita, P. californica, P. neopapillosa, P. papillosa, P. apuliae, P. bohemica, P. bonaquaense, P. huizhouensis, P. nidrosiensis, P. valida and P. tawfiki.
Phasmarhabditis neopapillosa is a nematode in the family Rhabditidae. It is a lethal facultative parasite of the terrestrial gastropods.

Steinernema carpocapsae is an entomopathogenic nematode and a member of the family Steinernematidae. It is a parasitic roundworm that has evolved an insect-killing symbiosis with bacteria, and kills its hosts within a few days of infection. This parasite releases its bacterial symbiont along with a variety of proteins into the host after infection, and together the bacteria and nematode overcome host immunity and kill the host quickly. As a consequence, S. carpocapsae has been widely adapted for use as a biological control agent in agriculture and pest control. S. carpocapsae is considered a generalist parasite and has been effectively used to control a variety of insects including: Webworms, cutworms, armyworms, girdlers, some weevils, and wood-borers. This species is an example of an "ambush" forager, standing on its tail in an upright position near the soil surface and attaching to passing hosts, even capable of jumping. As an ambush forager, S. carpocapsae is thought to be especially effective when applied against highly mobile surface-adapted insects. S. carpocapsae can sense carbon dioxide production, making the spiracles a key portal of entry into its insect hosts. It is most effective at temperatures ranging from 22–28 °C (72–82 °F).
Necromeny is a symbiotic relationship where an animal infects a host and waits inside its body until its death, at which point it develops and completes its life-cycle on the cadaver, feeding on the decaying matter and the subsequent bacterial growth. As the necromenic animal benefits from the relationship while the host is unharmed, it is an example of commensalism.














