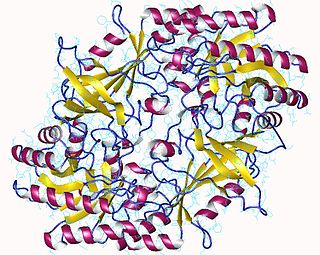
The enzyme ornithine decarboxylase catalyzes the decarboxylation of ornithine to form putrescine. This reaction is the committed step in polyamine synthesis. In humans, this protein has 461 amino acids and forms a homodimer.
Spermine is a polyamine involved in cellular metabolism that is found in all eukaryotic cells. The precursor for synthesis of spermine is the amino acid ornithine. It is an essential growth factor in some bacteria as well. It is found as a polycation at physiological pH. Spermine is associated with nucleic acids and is thought to stabilize helical structure, particularly in viruses. It functions as an intracellular free radical scavenger to protect DNA from free radical attack. Spermine is the chemical primarily responsible for the characteristic odor of semen.

Spermidine is a polyamine compound found in ribosomes and living tissues and having various metabolic functions within organisms. It was originally isolated from semen.

Spermidine synthase is an enzyme that catalyzes the transfer of the propylamine group from S-adenosylmethioninamine to putrescine in the biosynthesis of spermidine. The systematic name is S-adenosyl 3-(methylthio)propylamine:putrescine 3-aminopropyltransferase and it belongs to the group of aminopropyl transferases. It does not need any cofactors. Most spermidine synthases exist in solution as dimers.
Pantothenate kinase (EC 2.7.1.33, PanK; CoaA) is the first enzyme in the Coenzyme A (CoA) biosynthetic pathway. It phosphorylates pantothenate (vitamin B5) to form 4'-phosphopantothenate at the expense of a molecule of adenosine triphosphate (ATP). It is the rate-limiting step in the biosynthesis of CoA.
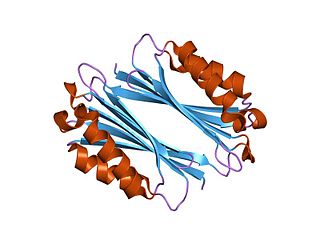
The enzyme adenosylmethionine decarboxylase catalyzes the conversion of S-adenosyl methionine to S-adenosylmethioninamine. Polyamines such as spermidine and spermine are essential for cellular growth under most conditions, being implicated in many cellular processes including DNA, RNA and protein synthesis. S-adenosylmethionine decarboxylase (AdoMetDC) plays an essential regulatory role in the polyamine biosynthetic pathway by generating the n-propylamine residue required for the synthesis of spermidine and spermine from putrescein. Unlike many amino acid decarboxylases AdoMetDC uses a covalently bound pyruvate residue as a cofactor rather than the more common pyridoxal 5'-phosphate. These proteins can be divided into two main groups which show little sequence similarity either to each other, or to other pyruvoyl-dependent amino acid decarboxylases: class I enzymes found in bacteria and archaea, and class II enzymes found in eukaryotes. In both groups the active enzyme is generated by the post-translational autocatalytic cleavage of a precursor protein. This cleavage generates the pyruvate precursor from an internal serine residue and results in the formation of two non-identical subunits termed alpha and beta which form the active enzyme.

In enzymology, a trypanothione synthase (EC 6.3.1.9) is an enzyme that catalyzes the chemical reaction

In enzymology, a diamine N-acetyltransferase is an enzyme that catalyzes the chemical reaction

Diamine acetyltransferase 1 is an enzyme that in humans is encoded by the SAT1 gene found on the X chromosome.

Polyamine-modulated factor 1 is a protein that in humans is encoded by the PMF1 gene.

Spermine oxidase is an enzyme that in humans is encoded by the SMOX gene.

Diamine acetyltransferase 2 is an enzyme that in humans is encoded by the SAT2 gene. SAT2 maintains a key metabolic glutamine/glutamate balance underpinning retrograde signaling by dendritic release of the neurotransmitter glutamate.

S-Adenosylmethioninamine is a substrate that is required for the biosynthesis of polyamines including spermidine, spermine, and thermospermine. It is produced by decarboxylation of S-adenosyl methionine.
A polyamine is an organic compound having more than two amino groups. Alkyl polyamines occur naturally, but some are synthetic. Alkylpolyamines are colorless, hygroscopic, and water soluble. Near neutral pH, they exist as the ammonium derivatives. Most aromatic polyamines are crystalline solids at room temperature.
N1-acetylpolyamine oxidase (EC 1.5.3.13, hPAO-1, mPAO, hPAO) is an enzyme with systematic name N1-acetylpolyamine:oxygen oxidoreductase (3-acetamidopropanal-forming). This enzyme catalyses the following chemical reaction
Spermine oxidase (EC 1.5.3.16, PAOh1/SMO, AtPAO1, AtPAO4, SMO) is an enzyme with systematic name spermidine:oxygen oxidoreductase (spermidine-forming). This enzyme catalyses the following chemical reaction
Non-specific polyamine oxidase (EC 1.5.3.17, polyamine oxidase, Fms1, AtPAO3) is an enzyme with systematic name polyamine:oxygen oxidoreductase (3-aminopropanal or 3-acetamidopropanal-forming). This enzyme catalyses the following chemical reaction
Homospermidine synthase (EC 2.5.1.44) is an enzyme with systematic name putrescine:putrescine 4-aminobutyltransferase (ammonia-forming). This enzyme catalyses the following chemical reaction
Deoxyhypusine synthase (EC 2.5.1.46, spermidine:eIF5A-lysine 4-aminobutyltransferase (propane-1,3-diamine-forming)) is an enzyme with systematic name (eIF5A-precursor)-lysine:spermidine 4-aminobutyltransferase (propane-1,3-diamine-forming). This enzyme catalyses the following chemical reaction
BpsA is a single-module non-ribosomal peptide synthase (NRPS) located in the cytoplasm responsible for the process of creating branched-chain polyamines, and producing spermidine and spermine. It has a singular ligand in its structure involved with Fe3+ and PLIP interactions. As seen by its EC number, it is a transferase (2) that transfers an alkyl or aryl group other than methyl groups (5) (2.5.1). BpsA was first discovered in the archaea Methanococcus jannaschii and thermophile Thermococcus kodakarensis and since then has been used in a variety of applications such as being used as a reporter, researching phosphopantetheinyl transferase (PPTase), and for NRPS domain recombination experiments it can be used as a model. Both (hyper)thermophilic bacteria and euryarchaeotal archaea seem to conserve BpsA and orthologs as branches chains polyamines are crucial for survival. There is also a second type of BpsA also known as Blue-pigment indigoidine synthetase that produces the pigment indigoidine and is found in organisms like Erwinia chrysanthemi. However, not much seems to be known about this variant except that it is a synthase, and it does not yet appear to be classified under an EC number.











