
Methionine is an essential amino acid in humans. As the precursor of other amino acids such as cysteine and taurine, versatile compounds such as SAM-e, and the important antioxidant glutathione, methionine plays a critical role in the metabolism and health of many species, including humans. It is encoded by the codon AUG.

Pyrrolysine is an α-amino acid that is used in the biosynthesis of proteins in some methanogenic archaea and bacteria; it is not present in humans. It contains an α-amino group, a carboxylic acid group. Its pyrroline side-chain is similar to that of lysine in being basic and positively charged at neutral pH.

Transfer RNA is an adaptor molecule composed of RNA, typically 76 to 90 nucleotides in length, that serves as the physical link between the mRNA and the amino acid sequence of proteins. Transfer RNA (tRNA) does this by carrying an amino acid to the protein synthesizing machinery of a cell called the ribosome. Complementation of a 3-nucleotide codon in a messenger RNA (mRNA) by a 3-nucleotide anticodon of the tRNA results in protein synthesis based on the mRNA code. As such, tRNAs are a necessary component of translation, the biological synthesis of new proteins in accordance with the genetic code.
In molecular biology, biosynthesis is a multi-step, enzyme-catalyzed process where substrates are converted into more complex products in living organisms. In biosynthesis, simple compounds are modified, converted into other compounds, or joined to form macromolecules. This process often consists of metabolic pathways. Some of these biosynthetic pathways are located within a single cellular organelle, while others involve enzymes that are located within multiple cellular organelles. Examples of these biosynthetic pathways include the production of lipid membrane components and nucleotides. Biosynthesis is usually synonymous with anabolism.

Amino acid synthesis is the set of biochemical processes by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can synthesize 11 of the 20 standard amino acids. These 11 are called the non-essential amino acids).
Acetyl-CoA synthetase (ACS) or Acetate—CoA ligase is an enzyme involved in metabolism of acetate. It is in the ligase class of enzymes, meaning that it catalyzes the formation of a new chemical bond between two large molecules.

Guanosine monophosphate synthetase, also known as GMPS is an enzyme that converts xanthosine monophosphate to guanosine monophosphate.
In enzymology, an isoleucine—tRNA ligase is an enzyme that catalyzes the chemical reaction
In enzymology, a leucine—tRNA ligase is an enzyme that catalyzes the chemical reaction
In enzymology, a lysine—tRNA ligase is an enzyme that catalyzes the chemical reaction
In enzymology, a threonine-tRNA ligase is an enzyme that catalyzes the chemical reaction
In enzymology, a tryptophan-tRNA ligase is an enzyme that catalyzes the chemical reaction
Tyrosine—tRNA ligase, also known as tyrosyl-tRNA synthetase is an enzyme that is encoded by the gene YARS. Tyrosine—tRNA ligase catalyzes the chemical reaction

SARS and cytoplasmic seryl-tRNA synthetase are a human gene and its encoded enzyme product, respectively. SARS belongs to the class II amino-acyl tRNA family and is found in all humans; its encoded enzyme, seryl-tRNA synthetase, is involved in protein translation and is related to several bacterial and yeast counterparts.

An expanded genetic code is an artificially modified genetic code in which one or more specific codons have been re-allocated to encode an amino acid that is not among the 22 common naturally-encoded proteinogenic amino acids.
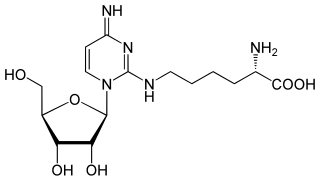
Lysidine is an uncommon nucleoside, rarely seen outside of tRNA. It is a derivative of cytidine in which the carbonyl is replaced by the amino acid lysine. The third position in the anti-codon of the Isoleucine-specific tRNA, is typically changed from a cytidine which would pair with guanosine to a lysidine which will base pair with adenosine. Uridine could not be used at this position even though it is a conventional partner for adenosine since it will also "wobble base pair" with guanosine. So lysidine allows better translation fidelity. Lysidine is denoted as L or k2C.
Amino acid activation refers to the attachment of an amino acid to its respective transfer RNA (tRNA). The reaction occurs in the cell cytosol and consists of two steps: first, the enzyme aminoacyl tRNA synthetase catalyzes the binding of adenosine triphosphate (ATP) to a corresponding amino acid, forming a reactive aminoacyl adenylate intermediate and releasing inorganic pyrophosphate (PPi). Subsequently, aminoacyl tRNA synthetase binds the AMP-amino acid to a tRNA molecule, releasing AMP and attaching the amino acid to the tRNA. The resulting aminoacyl-tRNA is said to be charged.

Agmatidine (2-agmatinylcytidine, symbol C+ or agm2C) is a modified cytidine present in the wobble position of the anticodon of several archaeal AUA decoding tRNAs. Agmatidine is essential for correct decoding of the AUA codon in many archaea and is required for aminoacylation of tRNAIle2 with isoleucine.

In biochemistry, wybutosine (yW) is a heavily modified nucleoside of phenylalanine transfer RNA that stabilizes interactions between the codons and anti-codons during protein synthesis. Ensuring accurate synthesis of protein is essential in maintaining health as defects in tRNA modifications are able to cause disease. In eukaryotic organisms, it is found only in position 37, 3'-adjacent to the anticodon, of phenylalanine tRNA. Wybutosine enables correct translation through the stabilization of the codon-anticodon base pairing during the decoding process.
TRNAMet cytidine acetyltransferase (EC 2.3.1.193, YpfI, TmcA) is an enzyme with systematic name acetyl-CoA:(elongator tRNAMet)-cytidine34 N4-acetyltransferase (ATP-hydrolysing). This enzyme catalyses the following chemical reaction










