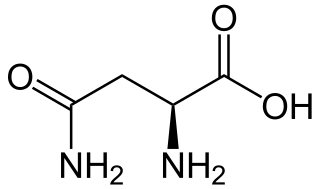
Asparagine is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group, an α-carboxylic acid group, and a side chain carboxamide, classifying it as a polar, aliphatic amino acid. It is non-essential in humans, meaning the body can synthesize it. It is encoded by the codons AAU and AAC.
In molecular biology, biosynthesis is a multi-step, enzyme-catalyzed process where substrates are converted into more complex products in living organisms. In biosynthesis, simple compounds are modified, converted into other compounds, or joined to form macromolecules. This process often consists of metabolic pathways. Some of these biosynthetic pathways are located within a single cellular organelle, while others involve enzymes that are located within multiple cellular organelles. Examples of these biosynthetic pathways include the production of lipid membrane components and nucleotides. Biosynthesis is usually synonymous with anabolism.
Glutamine synthetase (GS) is an enzyme that plays an essential role in the metabolism of nitrogen by catalyzing the condensation of glutamate and ammonia to form glutamine:

Asparaginase is an enzyme that is used as a medication and in food manufacturing. As a medication, L-asparaginase is used to treat acute lymphoblastic leukemia (ALL) and lymphoblastic lymphoma (LBL). It is given by injection into a vein, muscle, or under the skin. A pegylated version is also available. In food manufacturing it is used to decrease acrylamide.

Argininosuccinate synthase or synthetase is an enzyme that catalyzes the synthesis of argininosuccinate from citrulline and aspartate. In humans, argininosuccinate synthase is encoded by the ASS gene located on chromosome 9.

Amino acid synthesis is the set of biochemical processes by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can synthesize 11 of the 20 standard amino acids. These 11 are called the non-essential amino acids).

Indoleamine-pyrrole 2,3-dioxygenase (IDO or INDO EC 1.13.11.52) is a heme-containing enzyme physiologically expressed in a number of tissues and cells, such as the small intestine, lungs, female genital tract or placenta. In humans is encoded by the IDO1 gene. IDO is involved in tryptophan metabolism. It is one of three enzymes that catalyze the first and rate-limiting step in the kynurenine pathway, the O2-dependent oxidation of L-tryptophan to N-formylkynurenine, the others being indolamine-2,3-dioxygenase 2 (IDO2) and tryptophan 2,3-dioxygenase (TDO). IDO is an important part of the immune system and plays a part in natural defense against various pathogens. It is produced by the cells in response to inflammation and has an immunosuppressive function because of its ability to limit T-cell function and engage mechanisms of immune tolerance. Emerging evidence suggests that IDO becomes activated during tumor development, helping malignant cells escape eradication by the immune system. Expression of IDO has been described in a number of types of cancer, such as acute myeloid leukemia, ovarian cancer or colorectal cancer. IDO is part of the malignant transformation process and plays a key role in suppressing the anti-tumor immune response in the body, so inhibiting it could increase the effect of chemotherapy as well as other immunotherapeutic protocols. Furthermore, there is data implicating a role for IDO1 in the modulation of vascular tone in conditions of inflammation via a novel pathway involving singlet oxygen.

CAD protein is a trifunctional multi-domain enzyme involved in the first three steps of pyrimidine biosynthesis. De-novo synthesis starts with cytosolic carbamoylphosphate synthetase II which uses glutamine, carbon dioxide and ATP. This enzyme is inhibited by uridine triphosphate.
CTP synthase is an enzyme involved in pyrimidine biosynthesis that interconverts UTP and CTP.
In enzymology, an asparaginyl-tRNA synthase (glutamine-hydrolysing) is an enzyme that catalyzes the chemical reaction
In enzymology, an aspartate—ammonia ligase (ADP-forming) (EC 6.3.1.4) is an enzyme that catalyzes the chemical reaction

In enzymology, an omega-amidase (EC 3.5.1.3) is an enzyme that catalyzes the chemical reaction

Folylpolyglutamate synthase, mitochondrial is an enzyme that in humans is encoded by the FPGS gene.
Glutaminolysis (glutamine + -lysis) is a series of biochemical reactions by which the amino acid glutamine is lysed to glutamate, aspartate, CO2, pyruvate, lactate, alanine and citrate.

6-Diazo-5-oxo-L-norleucine (DON) is a glutamine antagonist, which was isolated originally from Streptomyces in a sample of Peruvian soil. This diazo compound is biosynthesized from lysine by three enzymes in bacteria. It is one of the most famous non-proteinogenic amino acid and was characterized in 1956 by Henry W Dion et al., who suggested a possible use in cancer therapy. This antitumoral efficacy was confirmed in different animal models. DON was tested as chemotherapeutic agent in different clinical studies, but was never approved. In 2019, DON was shown to kill tumor cells while reversing disease symptoms and improve overall survival in late-stage experimental glioblastoma in mice, when combined with calorie-restricted ketogenic diet.

CTP synthase 1 is an enzyme that is encoded by the CTPS1 gene in humans. CTP synthase 1 is an enzyme in the de novo pyrimidine synthesis pathway that catalyses the conversion of uridine triphosphate (UTP) to cytidine triphosphate (CTP). CTP is a key building block for the production of DNA, RNA and some phospholipids.
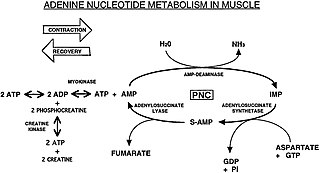
The Purine Nucleotide Cycle is a metabolic pathway in protein metabolism requiring the amino acids aspartate and glutamate. The cycle is used to regulate the levels of adenine nucleotides, in which ammonia and fumarate are generated. AMP converts into IMP and the byproduct ammonia. IMP converts to S-AMP (adenylosuccinate), which then converts to AMP and the byproduct fumarate. The fumarate goes on to produce ATP (energy) via oxidative phosphorylation as it enters the Krebs cycle and then the electron transport chain. Lowenstein first described this pathway and outlined its importance in processes including amino acid catabolism and regulation of flux through glycolysis and the Krebs cycle.
Asparagine synthase (glutamine-hydrolysing) (EC 6.3.5.4, asparagine synthetase (glutamine-hydrolysing), glutamine-dependent asparagine synthetase, asparagine synthetase B, AS, AS-B) is an enzyme with systematic name L-aspartate:L-glutamine amido-ligase (AMP-forming). This enzyme catalyses the following chemical reaction
The glnALG operon is an operon that regulates the nitrogen content of a cell. It codes for the structural gene glnA the two regulatory genes glnL and glnG. glnA encodes glutamine synthetase, an enzyme which catalyzes the conversion of glutamate and ammonia to glutamine, thereby controlling the nitrogen level in the cell. glnG encodes NRI which regulates the expression of the glnALG operon at three promoters, which are glnAp1, glnAp2 located upstream of glnA) and glnLp. glnL encodes NRII which regulates the activity of NRI. No significant homology is found in Eukaryotes.
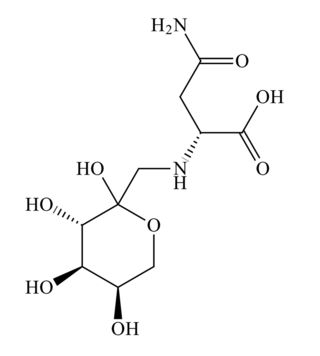
Fructose-asparagine (F-Asn) is a glycosylamine compound that is most notably used by Salmonella during Salmonella-mediated inflammation of the intestine. In addition to Salmonella, several other species of bacteria may utilize fructose-asparagine as a nutrient. The name of the genetic locus that encodes the uptake capability in Salmonella is fra. This fra locus has five genes: fraR, fraB a fructose-asparagine deglycase, fraD a sugar kinase, fraA a fructose-asparagine transporter, and fraE a L-asparaginase. Notably, mutations in fraB cause the buildup of the toxic intermediate 6-phosphofuctose-aspartate (6-P-F-Asp). The buildup of 6-P-F-Asp has a bacteriostatic effect on fraB mutant cells, making FraB a potential drug target.