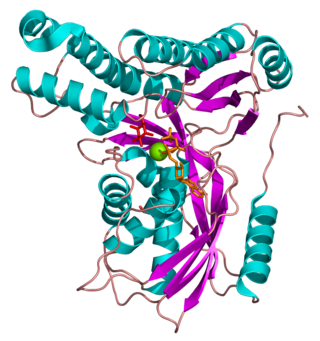
Galactokinase is an enzyme (phosphotransferase) that facilitates the phosphorylation of α-D-galactose to galactose 1-phosphate at the expense of one molecule of ATP. Galactokinase catalyzes the second step of the Leloir pathway, a metabolic pathway found in most organisms for the catabolism of α-D-galactose to glucose 1-phosphate. First isolated from mammalian liver, galactokinase has been studied extensively in yeast, archaea, plants, and humans.

Polymyxins are antibiotics. Polymyxins B and E are used in the treatment of Gram-negative bacterial infections. They work mostly by breaking up the bacterial cell membrane. They are part of a broader class of molecules called nonribosomal peptides.

The bacterial outer membrane is found in gram-negative bacteria. Gram-negative bacteria form two lipid bilayers in their cell envelopes - an inner membrane (IM) that encapsulates the cytoplasm, and an outer membrane (OM) that encapsulates the periplasm.

The enzyme UDP-glucose 4-epimerase, also known as UDP-galactose 4-epimerase or GALE, is a homodimeric epimerase found in bacterial, fungal, plant, and mammalian cells. This enzyme performs the final step in the Leloir pathway of galactose metabolism, catalyzing the reversible conversion of UDP-galactose to UDP-glucose. GALE tightly binds nicotinamide adenine dinucleotide (NAD+), a co-factor required for catalytic activity.
4-amino-4-deoxychorismate lyase is an enzyme that participates in folate biosynthesis by catalyzing the production of PABA by the following reaction

In enzymology, N-acetylglucosamine-6-phosphate deacetylase (EC 3.5.1.25), also known as GlcNAc-6-phosphate deacetylase or NagA, is an enzyme that catalyzes the deacetylation of N-acetylglucosamine-6-phosphate (GlcNAc-6-P) to glucosamine-6-phosphate (GlcN-6-P):
In enzymology, a lipid-A-disaccharide synthase is an enzyme that catalyzes the chemical reaction
Tetraacyldisaccharide 4'-kinase is an enzyme that phosphorylates the 4'-position of a tetraacyldisaccharide 1-phosphate precursor (DS-1-P) of lipopolysaccharide lipid A. This lipid forms outer membranes of Gram-negative bacteria. This enzyme catalyzes the chemical reaction
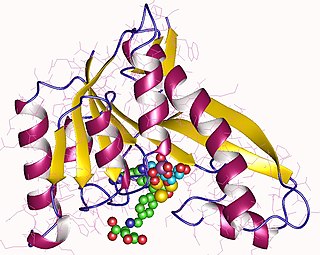
Phosphoribosylglycinamide formyltransferase (EC 2.1.2.2), also known as glycinamide ribonucleotide transformylase (GAR Tfase), is an enzyme with systematic name 10-formyltetrahydrofolate:5'-phosphoribosylglycinamide N-formyltransferase. This enzyme catalyses the following chemical reaction

The haloacid dehydrogenase superfamily is a superfamily of enzymes that include phosphatases, phosphonatases, P-type ATPases, beta-phosphoglucomutases, phosphomannomutases, and dehalogenases, and are involved in a variety of cellular processes ranging from amino acid biosynthesis to detoxification.
In molecular biology, the lipopolysaccharide kinase (Kdo/WaaP) family is a family of lipopolysaccharide kinases that includes lipopolysaccharide core heptose(I) kinase rfaP. Lipopolysaccharide core heptose(I) kinase rfaP is required for the addition of phosphate to O-4 of the first heptose residue of the lipopolysaccharide (LPS) inner core region. It has previously been shown that it is necessary for resistance to hydrophobic and polycationic antimicrobials in E. coli and that it is required for virulence in invasive strains of Salmonella enterica. The family also includes 3-deoxy-D-manno-octulosonic acid kinase from Haemophilus influenzae, which phosphorylates Kdo-lipid IV(A), a lipopolysaccharide precursor, and is involved in virulence.
UDP-glucuronic acid dehydrogenase (UDP-4-keto-hexauronic acid decarboxylating) (EC 1.1.1.305, UDP-GlcUA decarboxylase, ArnADH) is an enzyme with systematic name UDP-glucuronate:NAD+ oxidoreductase (decarboxylating). This enzyme catalyses the following chemical reaction
UDP-3-O-(3-hydroxymyristoyl)glucosamine N-acyltransferase is an enzyme with systematic name (3R)-3-hydroxymyristoyl-(acyl-carrier protein):UDP-3-O-( -3-hydroxymyristoyl)-alpha-D-glucosamine N-acetyltransferase. This enzyme catalyses the following chemical reaction
Lipid IVA 4-amino-4-deoxy-L-arabinosyltransferase is an enzyme with systematic name 4-amino-4-deoxy-alpha-L-arabinopyranosyl ditrans, octacis-undecaprenyl phosphate:lipid IVA 4-amino-4-deoxy-L-arabinopyranosyltransferase. This enzyme catalyses the following chemical reaction
Lipid IVA 3-deoxy-D-manno-octulosonic acid transferase is an enzyme with systematic name CMP-3-deoxy-D-manno-oct-2-ulosonate:lipid IVA 3-deoxy-D-manno-oct-2-ulosonate transferase. This enzyme catalyses the following chemical reaction
UDP-4-amino-4-deoxy-L-arabinose aminotransferase is an enzyme with systematic name UDP-4-amino-4-deoxy-beta-L-arabinose:2-oxoglutarate aminotransferase. This enzyme catalyses the following chemical reaction
3-deoxy-D-manno-octulosonic acid kinase is an enzyme with systematic name ATP:(KDO)-lipid IVA 3-deoxy-alpha-D-manno-oct-2-ulopyranose 4-phosphotransferase. This enzyme catalyses the following chemical reaction
Undecaprenyl-phosphate 4-deoxy-4-formamido-L-arabinose transferase is an enzyme with systematic name UDP-4-amino-4-deoxy-alpha-L-arabinose:ditrans,octacis-undecaprenyl phosphate 4-amino-4-deoxy-alpha-L-arabinosyltransferase. This enzyme catalyses the following chemical reaction
UDP-2,3-diacylglucosamine diphosphatase (EC 3.6.1.54, UDP-2,3-diacylglucosamine hydrolase, UDP-2,3-diacylglucosamine pyrophosphatase, ybbF (gene), lpxH (gene)) is an enzyme with systematic name UDP-2,3-bis((3R)-3-hydroxymyristoyl)-alpha-D-glucosamine 2,3-bis((3R)-3-hydroxymyristoyl)-beta-D-glucosaminyl 1-phosphate phosphohydrolase. This enzyme catalyses the following chemical reaction
N-glycosyltransferase is an enzyme in prokaryotes which transfers individual hexoses onto asparagine sidechains in substrate proteins, using a nucleotide-bound intermediary, within the cytoplasm. They are distinct from regular N-glycosylating enzymes, which are oligosaccharyltransferases that transfer pre-assembled oligosaccharides. Both enzyme families however target a shared amino acid sequence asparagine—-any amino acid except proline—serine or threonine (N–x–S/T), with some variations.







