
Androgen insensitivity syndrome (AIS) is a condition involving the inability to respond to androgens, typically due to androgen receptor dysfunction.
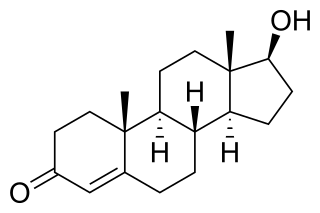
An androgen is any natural or synthetic steroid hormone that regulates the development and maintenance of male characteristics in vertebrates by binding to androgen receptors. This includes the embryological development of the primary male sex organs, and the development of male secondary sex characteristics at puberty. Androgens are synthesized in the testes, the ovaries, and the adrenal glands.

XY complete gonadal dysgenesis, also known as Swyer syndrome, is a type of defect hypogonadism in a person whose karyotype is 46,XY. Though they typically have normal vulvas, the person has underdeveloped gonads, fibrous tissue termed "streak gonads", and if left untreated, will not experience puberty. The cause is a lack or inactivation of an SRY gene which is responsible for sexual differentiation. Pregnancy is often possible in Swyer syndrome with assisted reproductive technology. The phenotype is usually similar to Turner syndrome (45,X0) due to a lack of X inactivation. The typical medical treatment is hormone replacement therapy. The syndrome was named after Gerald Swyer, an endocrinologist based in London.

Lipoid congenital adrenal hyperplasia is an endocrine disorder that is an uncommon and potentially lethal form of congenital adrenal hyperplasia (CAH). It arises from defects in the earliest stages of steroid hormone synthesis: the transport of cholesterol into the mitochondria and the conversion of cholesterol to pregnenolone—the first step in the synthesis of all steroid hormones. Lipoid CAH causes mineralocorticoid deficiency in affected infants and children. Male infants are severely undervirilized causing their external genitalia to look feminine. The adrenals are large and filled with lipid globules derived from cholesterol.

Thyroid hormone resistance (also resistance to thyroid hormone (RTH), and sometimes Refetoff syndrome) describes a rare syndrome in which the thyroid hormone levels are elevated but the thyroid stimulating hormone (TSH) level is not suppressed, or not completely suppressed as would be expected. The first report of the condition appeared in 1967. Essentially this is decreased end organ responsiveness to thyroid hormones. A new term "impaired sensitivity to thyroid hormone" has been suggested in March 2014 by Refetoff et al.
Isolated hypogonadotropic hypogonadism (IHH), also called idiopathic or congenital hypogonadotropic hypogonadism (CHH), as well as isolated or congenital gonadotropin-releasing hormone deficiency (IGD), is a condition which results in a small subset of cases of hypogonadotropic hypogonadism (HH) due to deficiency in or insensitivity to gonadotropin-releasing hormone (GnRH) where the function and anatomy of the anterior pituitary is otherwise normal and secondary causes of HH are not present.
Hypoestrogenism, or estrogen deficiency, refers to a lower than normal level of estrogen. It is an umbrella term used to describe estrogen deficiency in various conditions. Estrogen deficiency is also associated with an increased risk of cardiovascular disease, and has been linked to diseases like urinary tract infections and osteoporosis.
XX gonadal dysgenesis is a type of female hypogonadism in which the ovaries do not function to induce puberty in an otherwise normal girl whose karyotype is found to be 46,XX. With nonfunctional streak ovaries, she is low in estrogen levels (hypoestrogenic) and has high levels of FSH and LH. Estrogen and progesterone therapy is usually then commenced. Some cases are considered a severe version of premature ovarian failure where the ovaries fail before puberty.

Gonadal dysgenesis is classified as any congenital developmental disorder of the reproductive system in humans. It is atypical development of gonads in an embryo. One type of gonadal dysgenesis is the development of functionless, fibrous tissue, termed streak gonads, instead of reproductive tissue. Streak gonads are a form of aplasia, resulting in hormonal failure that manifests as sexual infantism and infertility, with no initiation of puberty and secondary sex characteristics.

Partial androgen insensitivity syndrome (PAIS) is a condition that results in the partial inability of the cell to respond to androgens. It is an X linked recessive condition. The partial unresponsiveness of the cell to the presence of androgenic hormones impairs the masculinization of male genitalia in the developing fetus, as well as the development of male secondary sexual characteristics at puberty, but does not significantly impair female genital or sexual development. As such, the insensitivity to androgens is clinically significant only when it occurs in individuals with a Y chromosome. Clinical features include ambiguous genitalia at birth and primary amenhorrhoea with clitoromegaly with inguinal masses. Müllerian structures are not present in the individual.

Estrogen insensitivity syndrome (EIS), or estrogen resistance, is a form of congenital estrogen deficiency or hypoestrogenism which is caused by a defective estrogen receptor (ER) – specifically, the estrogen receptor alpha (ERα) – that results in an inability of estrogen to mediate its biological effects in the body. Congenital estrogen deficiency can alternatively be caused by a defect in aromatase, the enzyme responsible for the biosynthesis of estrogens, a condition which is referred to as aromatase deficiency and is similar in symptomatology to EIS.

Disorders of sex development (DSDs), also known as differences in sex development or variations in sex characteristics (VSC), are congenital conditions affecting the reproductive system, in which development of chromosomal, gonadal, or anatomical sex is atypical.

The human gene SRD5A2 encodes the 3-oxo-5α-steroid 4-dehydrogenase 2 enzyme, also known as 5α-reductase type 2 (5αR2), one of three isozymes of 5α-reductase.

Aromatase deficiency is a rare condition characterized by extremely low levels or complete absence of the enzyme aromatase activity in the body. It is an autosomal recessive disease resulting from various mutations of gene CYP19 (P450arom) which can lead to ambiguous genitalia and delayed puberty in females, continued linear growth into adulthood and osteoporosis in males and virilization in pregnant mothers. As of 2020, fewer than 15 cases have been identified in genetically male individuals and at least 30 cases in genetically female individuals.
Breast development, also known as mammogenesis, is a complex biological process in primates that takes place throughout a female's life.

Mild androgen insensitivity syndrome (MAIS) is a condition that results in a mild impairment of the cell's ability to respond to androgens. The degree of impairment is sufficient to impair spermatogenesis and / or the development of secondary sexual characteristics at puberty in males, but does not affect genital differentiation or development. Female genital and sexual development is not significantly affected by the insensitivity to androgens; as such, MAIS is only diagnosed in males. The clinical phenotype associated with MAIS is a normal male habitus with mild spermatogenic defect and / or reduced secondary terminal hair.
Hypergonadotropic hypogonadism (HH), also known as primary or peripheral/gonadal hypogonadism or primary gonadal failure, is a condition which is characterized by hypogonadism which is due to an impaired response of the gonads to the gonadotropins, follicle-stimulating hormone (FSH) and luteinizing hormone (LH), and in turn a lack of sex steroid production. As compensation and the lack of negative feedback, gonadotropin levels are elevated. Individuals with HH have an intact and functioning hypothalamus and pituitary glands so they are still able to produce FSH and LH. HH may present as either congenital or acquired, but the majority of cases are of the former nature. HH can be treated with hormone replacement therapy.

Leydig cell hypoplasia (LCH), also known as Leydig cell agenesis, is a rare autosomal recessive genetic and endocrine syndrome affecting an estimated 1 in 1,000,000 genetic males. It is characterized by an inability of the body to respond to luteinizing hormone (LH), a gonadotropin which is normally responsible for signaling Leydig cells of the testicles to produce testosterone and other androgen sex hormones. The condition manifests itself as pseudohermaphroditism, hypergonadotropic hypogonadism, reduced or absent puberty, and infertility.
Hypergonadotropic hypergonadism is an endocrine situation and subtype of hypergonadism in which both gonadotropin levels and gonadal function, such as sex hormone production, are abnormally high. It can be associated with hyperandrogenism and hyperestrogenism and with gonadal cysts and tumors. It can be caused by medications such as gonadotropins, gonadotropin-releasing hormone agonists, nonsteroidal antiandrogens, and selective estrogen receptor modulators, as well as conditions like human chorionic gonadotropin-secreting tumors, complete androgen insensitivity syndrome, and estrogen insensitivity syndrome.
The pharmacology of estradiol, an estrogen medication and naturally occurring steroid hormone, concerns its pharmacodynamics, pharmacokinetics, and various routes of administration.















