 | |
| Names | |
|---|---|
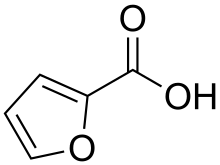
| Preferred IUPAC name Furan-2-carboxylic acid [1] | |
Other names
| |
| Identifiers | |
3D model (JSmol) | |
| 110149 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.639 |
| 3056 | |
| KEGG | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C5H4O3 | |
| Molar mass | 112.084 g·mol−1 |
| Appearance | White/ Off-White (Beige) Crystalline Powder |
| Density | 0.55 g/cm3 |
| Melting point | 128 to 132 °C (262 to 270 °F; 401 to 405 K) |
| Boiling point | 230 to 232 °C (446 to 450 °F; 503 to 505 K) |
| Easily soluble in cold and hot water, 27.1 g/L | |
| Acidity (pKa) | 3.12 at 25 °C |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards | Irritating to eyes, respiratory system and skin. |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds | 2-Thiophenecarboxylic acid, 3-Furoic acid, Furfuryl alcohol, 2,5-Furandicarboxylic acid, Furfurylamine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
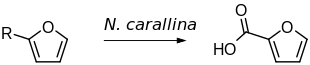
2-Furoic acid is an organic compound, consisting of a furan ring and a carboxylic acid side-group. Along with other furans, its name is derived from the Latin word furfur, meaning bran, from which these compounds were first produced. [2] The salts and esters of furoic acids are known as furoates. 2-Furoic acid is most widely encountered in food products as a preservative and a flavouring agent, where it imparts a sweet, earthy flavour. [3]